Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-Ones for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-Ones for Scalable Pharmaceutical Manufacturing
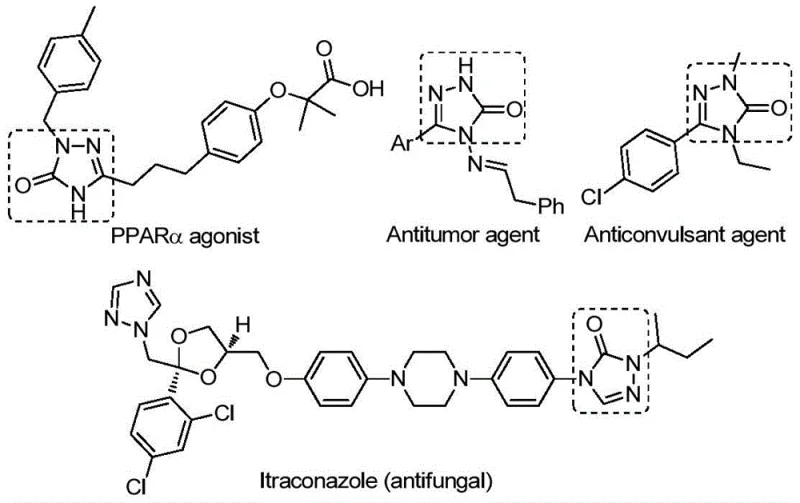
The pharmaceutical industry continuously seeks robust synthetic methodologies to access privileged scaffolds that serve as the backbone for next-generation therapeutics. Among these, the 1,2,4-triazole-3-one motif stands out due to its pervasive presence in bioactive molecules exhibiting antifungal, anti-inflammatory, antitumor, and anticonvulsant properties. A recent technological breakthrough documented in patent CN112538054B introduces a highly efficient preparation method for these valuable heterocycles. This innovation leverages a transition metal palladium-catalyzed carbonylation tandem cyclization strategy, transforming simple chlorohydrazones and sodium azide into complex triazole ketones. For R&D directors and procurement specialists alike, this patent represents a significant leap forward, offering a pathway to high-purity intermediates with streamlined operations and reduced dependency on hazardous gaseous reagents.
The significance of this development cannot be overstated in the context of modern drug discovery, where speed and purity are paramount. The disclosed method not only simplifies the synthetic route but also expands the chemical space accessible to medicinal chemists by tolerating a wide array of functional groups. By utilizing a solid carbon monoxide substitute, the process mitigates the safety risks associated with handling toxic CO gas, making it an attractive candidate for both laboratory-scale optimization and eventual commercial production. As we delve deeper into the technical specifics, it becomes clear that this methodology addresses several critical bottlenecks currently facing the synthesis of nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazole-3-one core has been fraught with synthetic challenges that hinder efficient large-scale manufacturing. Traditional literature reports describe several classical approaches, such as the cyclization of benzoyl hydrazide with urea under basic conditions or the tandem reaction of hydrazides with isocyanates. These legacy methods often suffer from severe drawbacks, including the requirement for harsh reaction conditions that can degrade sensitive functional groups. Furthermore, many of these routes necessitate the pre-activation of substrates, adding extra synthetic steps that reduce overall atom economy and increase waste generation. The narrow substrate scope of these conventional techniques frequently results in low yields when attempting to introduce diverse substituents, limiting their utility in the rapid iteration required for lead optimization in drug discovery programs.
The Novel Approach
In stark contrast, the method disclosed in patent CN112538054B offers a streamlined, one-pot solution that elegantly bypasses these historical limitations. The core of this innovation lies in the use of readily available chlorohydrazones as starting materials, which react with sodium azide in the presence of a palladium catalyst and a ligand system. Crucially, the reaction employs TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate, which releases CO in situ under heating conditions. This eliminates the need for high-pressure CO gas cylinders, drastically improving operational safety and simplifying the reactor setup. The reaction proceeds smoothly at 100°C in solvents like 1,4-dioxane, delivering the target 1,2,4-triazole-3-one compounds with impressive efficiency and broad substrate compatibility.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D teams aiming to adapt this chemistry for specific API intermediates. The proposed catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the thermal decomposition of TFBen releases carbon monoxide, which subsequently inserts into the carbon-palladium bond to form an acyl-palladium species. This key intermediate then reacts with sodium azide to generate an acyl azide compound in situ. The process continues via a Curtius rearrangement, converting the acyl azide into a highly reactive isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs, closing the ring to furnish the final 1,2,4-triazole-3-one product while regenerating the active palladium catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. The tandem nature of the reaction ensures that the reactive isocyanate intermediate is immediately consumed by the proximal nucleophile within the same molecule, minimizing side reactions such as hydrolysis or polymerization that often plague free isocyanate chemistry. The use of Xantphos as a bidentate ligand further stabilizes the palladium center, preventing catalyst aggregation and ensuring consistent turnover numbers throughout the reaction duration of 16 to 30 hours. This high level of control translates directly to cleaner crude reaction profiles, reducing the burden on downstream purification processes and ultimately yielding a product with a superior impurity profile suitable for stringent pharmaceutical specifications.
How to Synthesize 1,2,4-Triazole-3-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, CO source, chlorohydrazone, and sodium azide in an aprotic solvent. The mixture is then heated to facilitate the tandem cyclization. While the general procedure is robust, attention to stoichiometry and solvent choice is critical; for instance, 1,4-dioxane has been identified as the optimal solvent to ensure high conversion rates compared to polar aprotic alternatives like DMF or DMSO. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to guide process chemists in replicating these results effectively.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), carbon monoxide substitute (TFBen), chlorohydrazone, and sodium azide in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional multi-step syntheses to this novel palladium-catalyzed route presents compelling economic and logistical benefits. The primary driver for cost reduction lies in the simplicity of the raw material supply chain. Chlorohydrazones and sodium azide are commodity chemicals that are inexpensive and widely available from multiple global suppliers, reducing the risk of single-source dependency. Furthermore, the replacement of gaseous carbon monoxide with a solid surrogate like TFBen removes the need for specialized gas handling infrastructure and safety protocols, which significantly lowers the capital expenditure required for setting up production lines. This simplification of the process flow directly contributes to a more lean and cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of pre-activation steps and the use of a one-pot tandem reaction significantly reduce the total processing time and labor costs associated with intermediate isolation. By avoiding the use of hazardous gases and complex high-pressure equipment, the facility overheads related to safety compliance and maintenance are substantially decreased. Additionally, the high reaction efficiency and yields reported (up to 96% for certain substrates) mean less raw material is wasted, optimizing the cost per kilogram of the final API intermediate and enhancing the overall profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents rather than pressurized gases enhances the resilience of the supply chain. Solid reagents like TFBen and sodium azide are easier to transport, store, and handle than toxic CO gas, minimizing logistical bottlenecks and regulatory hurdles associated with hazardous material shipping. This stability ensures a continuous flow of materials to the production floor, reducing the likelihood of unplanned downtime due to supply shortages or delivery delays, which is critical for maintaining just-in-time manufacturing schedules for key pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are mild (100°C) and utilize standard organic solvents, making the process highly amenable to scale-up from gram to tonnage quantities without requiring exotic reactor designs. The improved atom economy and reduced waste generation align with green chemistry principles, simplifying waste treatment and disposal procedures. This environmental compatibility not only reduces the cost of waste management but also ensures compliance with increasingly stringent global environmental regulations, safeguarding the long-term viability of the production site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this method for industrial partners seeking to optimize their intermediate supply chains.
Q: What are the advantages of using TFBen over gaseous CO in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide surrogate, eliminating the need for high-pressure gas cylinders and specialized equipment, thereby significantly enhancing operational safety and simplifying the reaction setup for scale-up.
Q: What is the substrate scope of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully synthesizing derivatives with various substituents including alkyl groups (t-Bu, n-Bu), aryl groups (phenyl, naphthyl), and halogens (F, Br), with isolated yields reaching up to 96%.
Q: How does this method compare to traditional cyclization routes?
A: Unlike traditional methods requiring harsh conditions, pre-activated substrates, or multiple steps, this one-pot tandem reaction utilizes cheap, readily available chlorohydrazones and sodium azide under mild conditions (100°C), offering superior efficiency and atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
The technological potential of the palladium-catalyzed carbonylation route described in CN112538054B is immense, offering a clear path to high-quality pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one derivative adheres to the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your drug development timeline with reliable, cost-effective solutions.
