Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access privileged scaffolds that serve as the backbone for numerous bioactive molecules. As detailed in patent CN112538054B, a significant technological breakthrough has been achieved in the preparation of 1,2,4-triazole-3-one compounds, a class of five-membered nitrogen-containing heterocycles renowned for their diverse pharmacological profiles. These structures are integral to the development of agents exhibiting antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant activities, making them highly sought-after targets for drug discovery programs globally. The disclosed method represents a paradigm shift from conventional multi-step syntheses to a streamlined, transition metal-catalyzed carbonylation tandem cyclization process. By leveraging inexpensive starting materials such as chlorohydrazones and sodium azide, this innovation addresses critical pain points in pharmaceutical intermediates manufacturing, offering a route that is not only operationally simple but also economically viable for large-scale production.
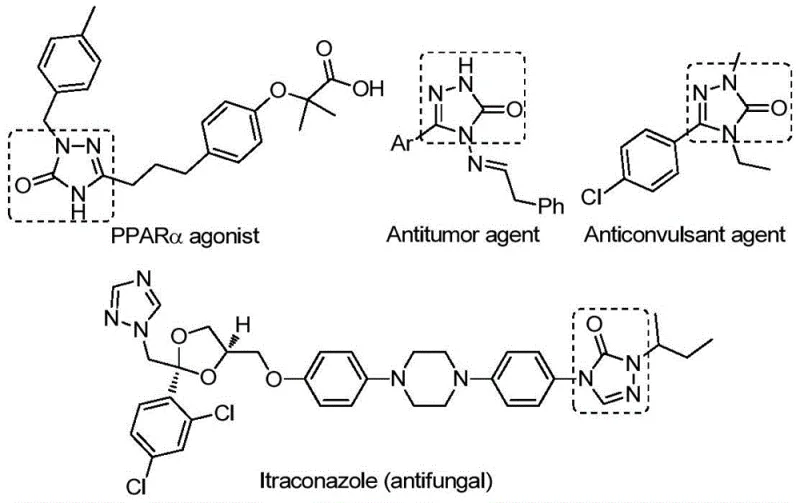
The strategic importance of the 1,2,4-triazole-3-one core cannot be overstated, as evidenced by its presence in a wide array of therapeutic agents ranging from tyrosinase inhibitors to angiotensin AT1 receptor antagonists. The ability to rapidly construct this heterocyclic system with high fidelity and purity is a key determinant in accelerating the timeline from lead identification to clinical candidate selection. For R&D Directors overseeing pipeline development, the availability of a robust synthetic method that tolerates a broad spectrum of functional groups is invaluable. It allows for the rapid generation of structural analogs to optimize potency and selectivity without being hindered by complex protection-deprotection sequences or incompatible reaction conditions. Furthermore, the method's compatibility with various substituents ensures that medicinal chemists can explore chemical space more freely, thereby enhancing the probability of identifying a successful drug candidate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one derivatives has been plagued by significant inefficiencies that hinder their widespread adoption in industrial settings. Traditional protocols often rely on the cyclization of benzoyl hydrazides with urea under strongly basic conditions, or the tandem reaction of hydrazides with isocyanates, which necessitates the handling of toxic and volatile reagents. Other reported methods involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines. These legacy approaches are frequently characterized by harsh reaction environments that can degrade sensitive functional groups, leading to poor yields and difficult purification processes. Moreover, the requirement for pre-activated substrates adds extra synthetic steps, increasing both the material cost and the environmental footprint of the manufacturing process. Such limitations create bottlenecks in the supply chain, resulting in longer lead times and higher costs for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the novel palladium-catalyzed carbonylation method introduced in the patent data offers a remarkably elegant solution. This approach utilizes a tandem cyclization reaction that efficiently constructs the triazolone ring in a single operational step from readily accessible chlorohydrazones and sodium azide. The use of a solid carbon monoxide substitute, specifically TFBen (1,3,5-tricarboxylic acid phenol ester), eliminates the need for handling hazardous CO gas, thereby significantly enhancing workplace safety and simplifying reactor requirements. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane at moderate temperatures, demonstrating exceptional substrate compatibility. This streamlined process not only reduces the number of unit operations but also minimizes waste generation, aligning perfectly with the principles of green chemistry. For procurement managers, this translates to a more reliable sourcing strategy with reduced dependency on specialized, high-cost reagents.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
The success of this synthetic methodology lies in its sophisticated yet robust catalytic cycle, which orchestrates the formation of multiple bonds with high precision. The reaction is initiated by the oxidative addition of the palladium catalyst, typically Pd2(dba)3 coordinated with a bulky phosphine ligand like Xantphos, into the carbon-chlorine bond of the chlorohydrazone substrate. This step generates a reactive divalent palladium intermediate that serves as the cornerstone for the subsequent transformations. Under the thermal conditions of the reaction (100-120°C), the solid CO surrogate TFBen decomposes to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond to form an acyl-palladium species. The introduction of sodium azide into the system triggers a nucleophilic attack on the acyl-palladium intermediate, leading to the formation of an acyl azide compound. This unstable intermediate spontaneously undergoes a Curtius rearrangement, releasing nitrogen gas and generating a highly reactive isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs where the nitrogen atom of the hydrazone moiety attacks the electrophilic carbon of the isocyanate group, closing the ring to yield the final 1,2,4-triazole-3-one product. This mechanistic pathway ensures high atom economy and minimizes the formation of side products.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-base mediated cyclizations. The mild nature of the palladium-catalyzed conditions prevents the degradation of sensitive functional groups that might otherwise decompose under strong acidic or basic environments. The specificity of the catalytic cycle ensures that the reaction proceeds through a well-defined pathway, reducing the likelihood of forming regioisomers or polymeric byproducts that are common in non-catalytic thermal reactions. Furthermore, the use of a stoichiometric amount of sodium azide, which is inexpensive and acts as both a nitrogen source and a driving force for the rearrangement, helps push the reaction to completion. The resulting crude reaction mixtures are generally cleaner, facilitating easier downstream processing. For quality control teams, this means that achieving stringent purity specifications becomes more straightforward, requiring less aggressive purification techniques that could otherwise impact overall yield.
How to Synthesize 1,2,4-Triazole-3-One Efficiently
The practical implementation of this synthesis is designed to be user-friendly for process chemists aiming to translate laboratory success to pilot plant operations. The protocol involves charging a reaction vessel with the palladium catalyst system, the ligand, the CO surrogate, the chlorohydrazone substrate, and sodium azide in a suitable solvent. The mixture is then heated to the optimal temperature range to drive the tandem cyclization to completion. Following the reaction, a simple workup procedure involving filtration and silica gel treatment allows for the isolation of the target compound. This operational simplicity is a key factor in reducing the technical barrier for adoption, making it an attractive option for contract development and manufacturing organizations (CDMOs) looking to optimize their service offerings. The detailed standardized synthesis steps for this process are outlined below to guide your technical team in replicating these results.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), carbon monoxide substitute (TFBen), chlorohydrazone, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 100°C and 120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product using column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic viability and continuity of the supply chain, the adoption of this novel synthetic route presents compelling advantages that directly impact the bottom line. The shift away from complex, multi-step traditional syntheses to a concise, one-pot catalytic process fundamentally alters the cost structure of manufacturing these valuable intermediates. By utilizing commodity chemicals like sodium azide and easily synthesized chlorohydrazones as starting materials, the reliance on expensive, custom-synthesized precursors is drastically reduced. This accessibility of raw materials mitigates supply chain risks associated with sole-source vendors or geopolitical instability affecting specialized chemical supplies. Additionally, the elimination of hazardous gas handling equipment lowers the capital expenditure required for setting up production lines, making it feasible for a wider range of manufacturing partners to produce these compounds competitively.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the use of cost-effective reagents. By replacing hazardous gaseous carbon monoxide with a solid surrogate, the process eliminates the need for specialized high-pressure reactors and gas scrubbing systems, which are capital-intensive and require rigorous maintenance. The high reaction efficiency and yield reported in the patent data mean that less raw material is wasted, directly lowering the cost of goods sold (COGS). Furthermore, the reduced number of purification steps translates to lower solvent consumption and waste disposal costs, contributing to substantial overall cost savings in API manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available starting materials. Chlorohydrazones can be rapidly synthesized from common acid chlorides and hydrazines, ensuring a steady flow of inputs even during market fluctuations. The robustness of the reaction conditions, which tolerate a variety of functional groups, reduces the risk of batch failures due to minor variations in raw material quality. This reliability allows supply chain heads to plan production schedules with greater confidence, reducing the need for excessive safety stock and minimizing the risk of stockouts that could delay downstream drug development projects. The scalability of the method ensures that supply can be ramped up quickly to meet surging demand.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this method offers a greener alternative to traditional synthesis. The avoidance of toxic isocyanates and the use of a solid CO source minimize the release of volatile organic compounds (VOCs) and hazardous emissions. The simpler workup procedure reduces the volume of aqueous and organic waste generated per kilogram of product. This alignment with green chemistry principles not only simplifies compliance with environmental regulations but also enhances the corporate social responsibility profile of the manufacturing entity. The process has been demonstrated to be scalable, providing a clear pathway from gram-scale laboratory optimization to commercial scale-up of complex pharmaceutical intermediates in multi-ton quantities.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled a set of frequently asked questions based on the patent specifications and industry standards. These insights are intended to address common concerns regarding process safety, scalability, and product quality. Understanding these details is crucial for making informed decisions about integrating this synthetic route into your existing manufacturing portfolio or procurement strategy. The following answers provide a deeper dive into the operational realities of implementing this Pd-catalyzed carbonylation method.
Q: What are the primary advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that often require harsh conditions, pre-activated substrates, or hazardous gases, this novel approach utilizes cheap and readily available chlorohydrazones and sodium azide. It operates under relatively mild thermal conditions with a solid CO surrogate, significantly improving safety and operational simplicity while delivering high yields.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up. The reaction uses standard organic solvents like 1,4-dioxane and avoids the need for high-pressure gas equipment since it employs a solid carbon monoxide substitute. The simple post-treatment involving filtration and chromatography facilitates efficient isolation of the product on a commercial scale.
Q: What is the substrate scope for this 1,2,4-triazole-3-one synthesis?
A: The method demonstrates excellent functional group tolerance. It successfully accommodates various substituents on both the R1 and R2 positions, including alkyl groups (such as tert-butyl and isopropyl), aryl groups (phenyl, naphthyl), and heteroaryl groups (furyl), allowing for the diverse design of biologically active molecules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the success of modern drug development. Our team of expert process chemists has thoroughly analyzed the potential of the Pd-catalyzed carbonylation route described in patent CN112538054B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one intermediate we deliver meets the highest international standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall project efficiency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic feasibility of switching to this new route or need specific COA data to verify product quality, we are here to assist. Please reach out to us today to request route feasibility assessments and discover how our expertise in pharmaceutical intermediates can accelerate your timeline to market while optimizing your manufacturing costs. Let us help you turn this innovative chemistry into a competitive advantage for your business.
