Advanced Synthesis of Chiral Spiro Indolone Compounds for Pharmaceutical Applications
Advanced Synthesis of Chiral Spiro Indolone Compounds for Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, stereoselective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in patent CN113980028A, which discloses a novel preparation method for chiral spiro indolone compounds. These structural motifs are ubiquitous in bioactive natural products and drug candidates, exhibiting potent anticancer, anti-HIV, and antimalarial activities. The patented technology leverages an intramolecular carbonyl α-asymmetric oxidative coupling reaction, utilizing a cinchona alkaloid-derived quaternary ammonium iodide salt as a phase-transfer catalyst and peroxide as a green oxidant. This approach represents a paradigm shift from traditional transition-metal catalysis, offering a robust pathway for reliable pharmaceutical intermediate supplier networks to access high-value chiral scaffolds with exceptional stereocontrol.
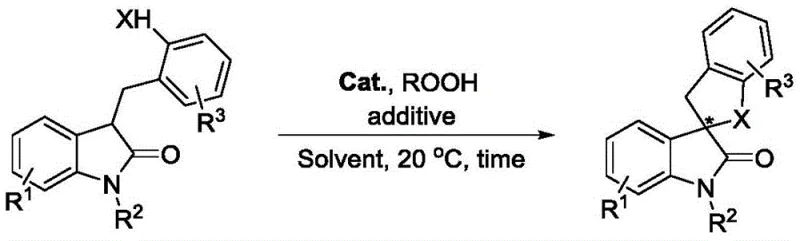
The core innovation lies in the ability to construct the challenging spirocyclic framework directly from indolone derivatives containing active hydrogen functional groups. Unlike conventional methods that often require pre-functionalized substrates or stoichiometric amounts of hazardous reagents, this catalytic system operates under remarkably mild conditions. The reaction proceeds efficiently in organic solvents such as n-propyl acetate at ambient temperature (20 °C), minimizing energy consumption and thermal degradation risks. For procurement managers and supply chain heads, this translates to a streamlined process that reduces operational complexity while ensuring the consistent delivery of high-purity chiral spiro indolone derivatives essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral spiro indolone skeletons has relied heavily on strategies such as Lewis acid-catalyzed 1,3-dipolar cycloadditions or nucleophilic phosphine-catalyzed cyclizations. While these methods have yielded valuable insights, they are fraught with significant limitations that hinder their application in cost reduction in pharmaceutical intermediates manufacturing. Lewis acid catalysts often require strict anhydrous conditions, low temperatures, and expensive ligands to achieve acceptable enantioselectivity. Furthermore, the removal of residual metal contaminants from the final API intermediate can be a costly and time-consuming purification bottleneck. Additionally, many traditional oxidative coupling protocols suffer from poor atom economy, generating substantial waste streams and requiring hazardous oxidants that pose safety risks during commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology described in CN113980028A overcomes these hurdles by employing an organocatalytic strategy centered on chiral iodide salts. By utilizing a cinchona alkaloid-derived quaternary ammonium iodide, the reaction achieves high stereoselectivity through a well-defined chiral environment without the need for toxic heavy metals. The use of cumene hydroperoxide (CHP) as the terminal oxidant is particularly advantageous; it is a stable, commercially available reagent that generates benign byproducts. The inclusion of 5A molecular sieves as an additive further distinguishes this process, as it actively removes water produced during the oxidation, shifting the equilibrium towards product formation. This results in a simplified workflow that enhances overall yield and purity, addressing the critical pain points of reducing lead time for high-purity pharmaceutical intermediates while maintaining rigorous quality standards.
Mechanistic Insights into Cinchona-Catalyzed Asymmetric Oxidative Coupling
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for process development. The reaction initiates with the oxidation of the iodide anion of the quaternary ammonium salt by the peroxide oxidant, generating a reactive hypoiodite or hypoiodate species in situ. This electrophilic iodine species then interacts with the enolizable carbonyl of the indolone substrate to form a transient N-I intermediate. This activation step is pivotal, as it renders the α-carbon susceptible to nucleophilic attack. Subsequently, an intramolecular SN2-type cyclization occurs, where the pendant nucleophile (such as a protected amine or phenol) attacks the activated center, closing the spiro ring with inversion of configuration. The rigid structure of the cinchona catalyst ensures that this cyclization occurs within a specific chiral pocket, dictating the absolute stereochemistry of the newly formed quaternary center.
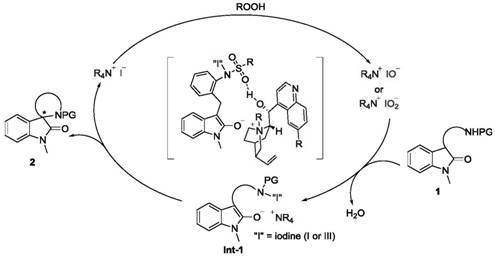
Furthermore, the role of non-covalent interactions cannot be overstated in achieving high enantiomeric ratios. The bulky quaternary ammonium cation forms a tight ion pair with the indolone enolate, while hydrogen bonding between the catalyst's secondary hydroxyl group and the substrate's sulfonyl protecting group provides additional stereochemical guidance. As the reaction progresses, water is generated as a byproduct, which can potentially hydrolyze the active iodine species or inhibit the catalyst. The strategic addition of molecular sieves mitigates this issue by sequestering water molecules, thereby maintaining the activity of the catalytic cycle and preventing side reactions. This mechanistic elegance allows for the synthesis of diverse spiro indolone derivatives with excellent yields and enantioselectivity, providing a versatile platform for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Chiral Spiro Indolone Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory optimization and industrial production. The protocol involves dissolving the indolone substrate in n-propyl acetate, adding the chiral catalyst and molecular sieves, and initiating the reaction with the dropwise addition of cumene hydroperoxide at room temperature. The simplicity of the workup procedure—quenching with sodium thiosulfate followed by standard extraction and chromatography—further underscores its utility. For detailed procedural specifics regarding reagent grades, stirring rates, and purification parameters, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining the indolone-derived substrate containing active hydrogen functional groups with a cinchona alkaloid-derived quaternary ammonium iodide salt catalyst (3 mol%) in an organic solvent such as n-propyl acetate.
- Add 5A molecular sieves (0.4 g/mmol) as an additive to absorb water generated during the reaction, followed by the addition of cumene hydroperoxide (3 equivalents) as the oxidant.
- Stir the reaction mixture at 20 °C for 12 hours, then quench with saturated sodium thiosulfate, extract with ethyl acetate, and purify the crude product via column chromatography to obtain the chiral spiro indolone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that align with the strategic goals of modern chemical procurement and supply chain management. The shift from precious metal catalysis to organocatalysis fundamentally alters the cost structure of production. By eliminating the need for expensive palladium, rhodium, or iridium complexes, manufacturers can achieve substantial cost savings in raw material acquisition. Moreover, the absence of heavy metals simplifies the regulatory compliance landscape, reducing the burden of extensive metal residue testing and specialized waste disposal protocols. This streamlined approach not only lowers the direct cost of goods sold but also accelerates the time-to-market for new drug candidates by removing potential regulatory bottlenecks associated with metal contamination.
- Cost Reduction in Manufacturing: The utilization of inexpensive, commodity-grade reagents such as cumene hydroperoxide and n-propyl acetate significantly drives down operational expenditures. The catalyst, derived from abundant cinchona alkaloids, is not only cost-effective but also reusable in certain iterations, further enhancing economic efficiency. The mild reaction conditions (20 °C) eliminate the need for energy-intensive heating or cryogenic cooling systems, resulting in significantly reduced utility costs. Additionally, the high yields reported in the patent minimize material loss, ensuring that every kilogram of starting material is converted into valuable product with maximal efficiency.
- Enhanced Supply Chain Reliability: One of the most critical factors for supply chain heads is the availability of raw materials. All key components of this synthesis, including the indolone substrates, oxidants, and solvents, are widely available industrial commodities with robust global supply chains. This diversity of supply sources mitigates the risk of shortages or price volatility that often plagues specialized reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality or environmental factors, ensuring consistent production continuity and reliable delivery schedules for downstream partners.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to proceed efficiently without the exothermic runaway risks associated with some traditional oxidation methods. The use of greener solvents and the generation of minimal hazardous waste align with increasingly stringent environmental regulations. The solid molecular sieve additive can be easily filtered and regenerated or disposed of safely, simplifying waste management. This environmental compatibility facilitates easier permitting for new manufacturing facilities and supports corporate sustainability goals, making it an attractive option for long-term strategic sourcing partnerships.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for process chemists and engineering teams.
Q: What is the key advantage of using cinchona alkaloid-derived catalysts for spiro indolone synthesis?
A: The use of cinchona alkaloid-derived quaternary ammonium iodide salts enables highly enantioselective intramolecular oxidative coupling under mild conditions (20 °C), avoiding the harsh Lewis acids or complex metal catalysts often required in traditional methods.
Q: Why are molecular sieves critical in this oxidative coupling reaction?
A: Water is a byproduct of the oxidative coupling reaction which can slow down the process or shift equilibrium. 5A molecular sieves effectively absorb this water, driving the reaction forward and significantly improving both yield and reaction efficiency.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the method operates at room temperature (20 °C) using commercially available solvents like n-propyl acetate and inexpensive oxidants, making it highly scalable and cost-effective for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN113980028A for the next generation of therapeutic agents. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from pilot plant to full-scale manufacturing. We maintain stringent purity specifications across all our operations, supported by state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to verify enantiomeric excess and impurity profiles.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your drug development pipeline. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data for similar spirocyclic scaffolds and discuss route feasibility assessments for your target molecules. Together, we can accelerate the delivery of life-saving medicines to patients worldwide.
