Scalable Synthesis of Chiral Spiro Indolones via Organocatalytic Asymmetric Oxidative Coupling
The pharmaceutical industry continuously seeks robust and efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive natural products and drug candidates. Patent CN113980028A introduces a groundbreaking preparation method for chiral spiro indolone compounds, utilizing an intramolecular carbonyl α-asymmetric oxidative coupling reaction. This technology represents a significant leap forward in fine chemical synthesis, employing a cinchona alkaloid-derived quaternary ammonium iodide salt as a chiral phase-transfer catalyst and peroxide as a green oxidant. Unlike traditional methods that often rely on harsh conditions or expensive transition metals, this protocol operates under remarkably mild conditions, typically at 20°C, ensuring the integrity of sensitive functional groups while delivering high yields and excellent stereoselectivity. The versatility of this approach allows for the modification of various substituents on the indolone core, making it a powerful tool for generating diverse libraries of pharmaceutical intermediates.
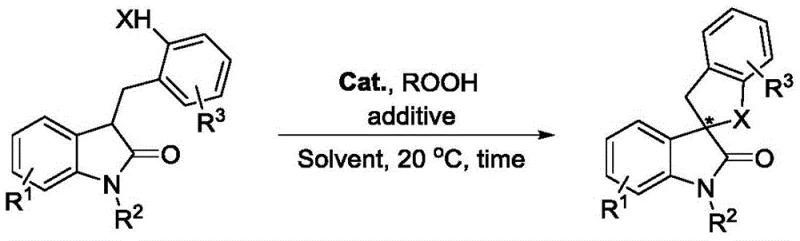
The strategic value of this patent lies in its ability to access chiral spiro indolone structures, which are privileged motifs in medicinal chemistry known for their anticancer, anti-HIV, and anti-malarial activities. By leveraging a metal-free organocatalytic system, the process aligns perfectly with modern green chemistry principles, reducing the environmental footprint associated with heavy metal waste disposal. For R&D teams, this offers a reliable pathway to synthesize complex spirocyclic cores that were previously difficult to access with high optical purity. The reaction tolerates a wide range of functional groups, including halogens, alkyls, and alkoxy groups, providing the flexibility needed for late-stage functionalization in drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral spiro indolone skeletons has relied heavily on Lewis acid-catalyzed 1,3-dipolar cycloadditions or nucleophilic phosphine-catalyzed cyclizations. While these methods have achieved certain successes, they often suffer from significant drawbacks that hinder their industrial application. Lewis acid catalysts frequently require strict anhydrous conditions and low temperatures to maintain selectivity, leading to high energy consumption and operational complexity. Furthermore, the removal of residual metal contaminants from the final API intermediate is a costly and time-consuming purification step that can drastically impact overall process efficiency. Phosphine catalysis, on the other hand, may involve expensive reagents and can sometimes exhibit limited substrate scope or lower atom economy. These conventional approaches often struggle to balance high stereoselectivity with practical scalability, creating a bottleneck for the commercial production of spiro indolone-based therapeutics.
The Novel Approach
The methodology described in CN113980028A overcomes these hurdles by introducing a chiral iodide salt catalytic system combined with peroxide oxidation. This novel approach eliminates the need for transition metals entirely, replacing them with inexpensive and readily available cinchona alkaloid derivatives. The reaction proceeds via an intramolecular oxidative coupling mechanism that is inherently more atom-economical than cycloaddition strategies. By operating at ambient temperature (20°C) in common organic solvents like n-propyl acetate, the process significantly reduces energy requirements and safety risks associated with exothermic reactions. The inclusion of molecular sieves as an additive further enhances the reaction efficiency by sequestering water, a byproduct that can otherwise inhibit the catalytic cycle. This combination of mild conditions, metal-free catalysis, and high efficiency makes the new method superior for both laboratory-scale discovery and kilogram-scale manufacturing.
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Oxidative Coupling
Understanding the catalytic cycle is crucial for optimizing this reaction for specific substrates. The mechanism begins with the oxidation of the quaternary ammonium iodide salt catalyst by the peroxide oxidant, generating a reactive hypoiodite or hypoiodous acid species in situ. This active iodine species then oxidizes the indolone substrate to form a key N-I intermediate. Following this oxidation step, the reaction proceeds through an intramolecular SN2-type cyclization, where the nucleophilic attack is stereochemically controlled by the chiral catalyst. The rigid structure of the cinchona alkaloid framework ensures that the substrate approaches the reactive center from a specific trajectory, resulting in high enantioselectivity. Additionally, non-covalent interactions, such as tight ion pairing between the quaternary ammonium cation and the indolone enol anion, play a vital role in stabilizing the transition state. Hydrogen bonding between the catalyst's secondary alcohol moiety and the substrate's sulfonyl group further reinforces this chiral induction, locking the conformation necessary for asymmetric bond formation.
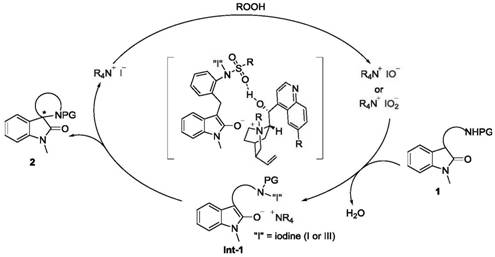
A critical aspect of this mechanism is the management of water, which is produced as a stoichiometric byproduct during the oxidation and cyclization steps. As water accumulates in the reaction medium, it can shift the equilibrium backward or deactivate the active iodine species, leading to reduced yields and slower reaction rates. The patent explicitly addresses this challenge by incorporating molecular sieves (5Å) into the reaction mixture. These solid additives act as a desiccant, continuously absorbing the generated water molecules and driving the equilibrium towards product formation according to Le Chatelier's principle. Moreover, the solid nature of the molecular sieves ensures they settle at the bottom of the reactor, preventing interference with the liquid-phase reaction dynamics while maintaining a dry environment. This clever engineering of the reaction conditions ensures consistent high performance and reproducibility, which is essential for scaling up the synthesis of high-purity pharmaceutical intermediates.
How to Synthesize Chiral Spiro Indolone Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential. The process involves dissolving the indolone derivative substrate in a suitable organic solvent, such as n-propyl acetate, which has been identified as the optimal medium for balancing solubility and reaction rate. The cinchona alkaloid-derived catalyst, typically used at a loading of 3 mol%, is added along with activated 5Å molecular sieves. The reaction is initiated by the slow addition of cumene hydroperoxide (CHP), which serves as the terminal oxidant. Maintaining the temperature at 20°C is critical; while the reaction is exothermic, the mild conditions prevent thermal degradation of the chiral catalyst or the product. After stirring for approximately 12 hours, the reaction is quenched with a saturated sodium thiosulfate solution to reduce any remaining oxidant and iodine species. The detailed standardized synthesis steps for specific substrates are outlined in the guide below.
- Prepare the reaction mixture by combining the indolone-derived substrate containing active hydrogen functional groups with a cinchona alkaloid-derived quaternary ammonium iodide salt catalyst in an organic solvent such as n-propyl acetate.
- Add molecular sieves (5Å) as an additive to absorb water generated during the reaction, followed by the addition of a peroxide oxidant like cumene hydroperoxide.
- Stir the reaction mixture at a mild temperature of 20°C for approximately 12 hours, then quench with sodium thiosulfate and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers substantial strategic benefits beyond mere technical feasibility. The shift from metal-catalyzed processes to organocatalysis fundamentally alters the cost structure and risk profile of the supply chain. By removing the dependency on precious metals like palladium, rhodium, or iridium, manufacturers can insulate themselves from the volatile pricing and geopolitical supply risks associated with these critical raw materials. Furthermore, the simplified purification workflow reduces the number of processing steps, directly translating to lower operational expenditures and faster throughput times. The use of commodity chemicals such as cumene hydroperoxide and n-propyl acetate ensures that the supply chain remains robust and resilient, with multiple qualified vendors available globally to mitigate disruption risks.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts results in significant raw material cost savings. Additionally, the absence of heavy metals removes the need for specialized scavenging resins or complex purification protocols required to meet stringent regulatory limits for metal residues in APIs. This streamlining of the downstream processing significantly lowers the cost of goods sold (COGS). The mild reaction conditions also reduce energy consumption for heating or cryogenic cooling, contributing to further operational cost reductions. Overall, the process economics are improved through higher atom economy and reduced waste generation.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including indolone derivatives and cinchona alkaloids, are widely available industrial commodities with established global supply networks. This abundance ensures a stable supply of raw materials, reducing the lead time for high-purity pharmaceutical intermediates. The robustness of the reaction against minor variations in conditions means that batch-to-batch consistency is easier to maintain, minimizing the risk of production delays due to failed batches. Suppliers can therefore offer more reliable delivery schedules and greater flexibility in responding to fluctuating market demands.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from milligram to multi-gram scales without loss of selectivity. The use of greener reagents and the generation of less hazardous waste align with increasingly strict environmental regulations, facilitating smoother regulatory approvals. The simplicity of the workup procedure, involving standard extraction and crystallization, allows for easy adaptation to large-scale reactors. This scalability ensures that the technology can support commercial production volumes ranging from pilot plant trials to full-scale manufacturing without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric oxidative coupling technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into existing production pipelines.
Q: What is the primary advantage of this oxidative coupling method over traditional Lewis acid catalysis?
A: This method utilizes a metal-free organocatalytic system based on cinchona alkaloid iodide salts, eliminating the need for expensive and potentially toxic transition metal catalysts. It operates under significantly milder conditions (20°C) compared to many traditional methods, reducing energy costs and simplifying safety protocols.
Q: How does the process ensure high enantioselectivity in the spiro indolone products?
A: High stereoselectivity is achieved through the chiral environment provided by the rigid quaternary ammonium unit of the cinchona alkaloid catalyst. This creates tight ion pairing with the indolone enol anion and facilitates specific hydrogen bonding interactions that direct the intramolecular SN2 reaction towards a single enantiomer.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its use of commercially available starting materials, mild reaction temperatures that do not require specialized cooling or heating infrastructure, and a simple workup procedure involving standard extraction and chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the organocatalytic oxidative coupling technology described in CN113980028A for the synthesis of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe handling of peroxides and the precise temperature control needed for optimal stereoselectivity. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral spiro indolone meets the highest quality standards required by global regulatory agencies.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your next drug development project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target timelines. Please contact our technical procurement team today to request specific COA data for our reference standards and to discuss comprehensive route feasibility assessments. Together, we can accelerate the delivery of life-saving medicines to patients while optimizing your supply chain efficiency.
