Advanced Asymmetric Oxidative Coupling for Commercial Scale Chiral Spiro Indolone Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive natural products and drug candidates. A significant breakthrough in this domain is documented in patent CN113980028A, which details a novel preparation method for chiral spiro indolone compounds. This technology leverages an intramolecular carbonyl α-asymmetric oxidative coupling reaction, utilizing cinchona alkaloid-derived quaternary ammonium iodide salts as organocatalysts and peroxides as oxidants. For R&D Directors and Process Chemists, this represents a paradigm shift from traditional transition-metal catalysis to a more sustainable, organocatalytic approach that operates under exceptionally mild conditions. The method addresses critical pain points in modern synthesis, including the removal of toxic metal residues and the simplification of reaction conditions, thereby offering a robust platform for the reliable pharmaceutical intermediate supplier networks aiming to deliver high-purity materials.
From a commercial perspective, the implications of this technology extend far beyond the laboratory bench. By enabling the construction of chiral spiro frameworks through a direct oxidative coupling strategy, manufacturers can achieve cost reduction in chiral API manufacturing through streamlined processing and reduced waste treatment costs associated with heavy metals. The process demonstrates high stereoselectivity and yield, which are paramount for maintaining stringent quality control standards in the production of high-purity spiro indolone derivatives. Furthermore, the scalability of this reaction, evidenced by its operation at ambient temperature and use of common solvents, ensures that commercial scale-up of complex heterocycles can be achieved with minimal capital expenditure on specialized reactor infrastructure, securing the supply chain continuity for downstream drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral spiro indolone skeletons has relied heavily on methodologies such as Lewis acid-catalyzed 1,3-dipolar cycloadditions or nucleophilic phosphine-catalyzed cyclizations. While these methods have provided access to diverse chemical spaces, they are often plagued by significant operational drawbacks that hinder industrial adoption. Traditional routes frequently necessitate the use of sensitive reagents, strictly anhydrous conditions, or expensive chiral ligands that drive up the cost of goods sold (COGS). Moreover, reactions involving transition metals often require rigorous downstream purification steps to reduce metal content to parts-per-million (ppm) levels, a requirement mandated by regulatory bodies for pharmaceutical ingredients. These additional purification stages not only extend the reducing lead time for high-purity pharmaceutical intermediates but also result in substantial material loss, negatively impacting the overall atom economy and environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN113980028A introduces a streamlined organocatalytic pathway that circumvents these historical bottlenecks. By employing a quaternary ammonium iodide salt derived from cinchona alkaloids, the reaction proceeds via an in situ generated hypoiodite species that facilitates the oxidative coupling under remarkably mild conditions. The general reaction scheme illustrates the transformation of indolone derivatives bearing active hydrogen functional groups into the desired spirocyclic products with high efficiency.
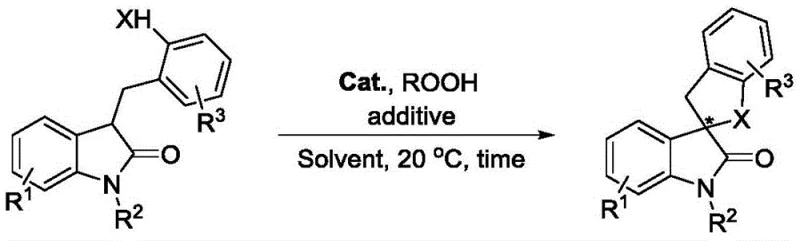
This novel approach eliminates the need for precious metal catalysts entirely, replacing them with inexpensive, readily available organic salts. The reaction operates effectively at 20°C in common organic solvents like n-propyl acetate, drastically simplifying the thermal management requirements for large-scale reactors. This simplicity translates directly into enhanced process safety and reduced energy consumption, making it an ideal candidate for green chemistry initiatives within the fine chemical sector. The ability to tolerate various functional groups on the indolone ring further underscores the versatility of this method for generating diverse libraries of bioactive compounds.
Mechanistic Insights into Organocatalytic Asymmetric Oxidative Coupling
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The catalytic cycle initiates with the oxidation of the iodide anion of the cinchona-derived catalyst by the peroxide oxidant, generating a reactive hypoiodite or hypoiodous acid species in situ. This electrophilic iodine species then interacts with the enolizable carbonyl of the indolone substrate to form a key N-I intermediate. The chirality of the cinchona backbone exerts precise stereocontrol during the subsequent intramolecular nucleophilic substitution (SN2) step, where the nitrogen or oxygen nucleophile attacks the activated carbon center to close the spiro ring. This tight ion-pairing mechanism between the rigid quaternary ammonium cation and the substrate anion is responsible for the high enantiomeric ratios observed.
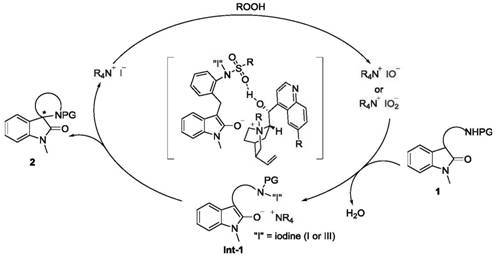
A critical aspect of this mechanism, often overlooked in standard oxidative couplings, is the management of reaction byproducts. The oxidative process inherently generates water molecules, which can inhibit the reaction rate or promote the hydrolysis of the active iodine species, leading to equilibrium stagnation. The patent explicitly highlights the strategic addition of molecular sieves (specifically 5Å MS) as an essential additive. These solid particles function as a desiccant within the liquid phase, continuously adsorbing the generated water. By Le Chatelier's principle, the removal of water drives the equilibrium towards the product side, significantly boosting both the reaction rate and the final isolated yield. This subtle yet powerful engineering of the reaction environment exemplifies how physical additives can be leveraged to enhance chemical performance without altering the core catalytic species.
How to Synthesize Chiral Spiro Indolone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the oxidant and the quality of the drying agents used. The standard protocol involves dissolving the indolone substrate in n-propyl acetate, adding the cinchona catalyst (typically 3 mol%), and introducing activated molecular sieves prior to the addition of the oxidant. Cumene hydroperoxide is the preferred oxidant due to its stability and reactivity profile in this specific system. The detailed standardized synthesis steps below outline the precise operational parameters required to achieve the reported high yields and stereoselectivity.
- Prepare the reaction mixture by combining the indolone-derived substrate containing active hydrogen functional groups with a cinchona alkaloid-derived quaternary ammonium iodide salt catalyst in an organic solvent such as n-propyl acetate.
- Add molecular sieves as an additive to the reaction vessel to act as a water scavenger, ensuring the equilibrium shifts towards product formation by removing the water byproduct generated during oxidation.
- Introduce the oxidant, preferably cumene hydroperoxide, and stir the mixture at a mild temperature of 20°C for approximately 12 hours to complete the intramolecular carbonyl alpha-asymmetric oxidative coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this organocatalytic method offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the decoupling of the supply chain from the volatile market of precious metals. By utilizing iodide salts derived from abundant cinchona alkaloids, manufacturers can secure a stable and predictable cost structure for their catalytic systems. This stability is crucial for long-term contract manufacturing organizations (CMOs) that need to guarantee pricing to their pharmaceutical clients over multi-year agreements. Furthermore, the elimination of heavy metals simplifies the regulatory filing process, as there is no need to validate complex metal clearance protocols, thereby accelerating the time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior to traditional methods due to the drastic simplification of the downstream processing train. Without the need for scavengers to remove palladium, rhodium, or other transition metals, the number of unit operations—such as filtration through silica pads or treatment with specialized resins—is significantly reduced. This reduction in processing steps leads to lower solvent consumption, reduced labor hours, and decreased waste disposal costs. Additionally, the high atom economy and excellent yields reported in the patent minimize the amount of starting material required per kilogram of final product, directly lowering the raw material cost basis and improving the overall margin profile for the manufactured intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain resilience. Operating at ambient temperature (20°C) removes the dependency on complex heating or cryogenic cooling infrastructure, which are common points of failure in chemical plants. The reagents involved, such as cumene hydroperoxide and n-propyl acetate, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source bottlenecks. This redundancy ensures that production schedules can be maintained even during periods of regional supply disruption, providing a reliable flow of critical intermediates to downstream formulation facilities and preventing costly production stoppages.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology aligns perfectly with modern green chemistry principles. The absence of toxic heavy metals reduces the hazard profile of the waste stream, simplifying effluent treatment and lowering the cost of environmental compliance. The mild reaction conditions also reduce the energy intensity of the process, contributing to a lower carbon footprint for the manufacturing site. As regulatory pressures regarding industrial emissions and waste increase globally, adopting such inherently cleaner technologies future-proofs the manufacturing asset, ensuring continued operational licensure and enhancing the corporate sustainability profile which is increasingly important to investors and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric oxidative coupling technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What represents the primary advantage of this oxidative coupling method over traditional Lewis acid catalysis?
A: Unlike traditional methods that often require harsh Lewis acids or complex 1,3-dipolar cycloadditions, this novel approach utilizes a mild organocatalytic system at ambient temperature (20°C), significantly reducing energy consumption and simplifying the purification process by avoiding heavy metal residues.
Q: How does the use of molecular sieves impact the reaction efficiency?
A: Water is a byproduct of the oxidative coupling reaction which can slow down the process or shift the equilibrium backwards. The inclusion of 5Å molecular sieves effectively absorbs this water in situ, driving the reaction forward to completion and enhancing both the chemical yield and the stereochemical purity of the final spiro indolone product.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up due to its operation at room temperature without the need for cryogenic conditions or high-pressure equipment. Furthermore, the catalysts are derived from abundant cinchona alkaloids, and the oxidants are commodity chemicals, ensuring a robust and cost-effective supply chain for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic methods like the one described in CN113980028A for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR capabilities to guarantee that every batch of chiral spiro indolone meets the exacting standards required for clinical and commercial applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge synthesis technology for their pipeline projects. By partnering with our technical procurement team, you can receive a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this organocatalytic route for your specific molecule. We encourage you to contact us today to request specific COA data from our recent batches and to discuss route feasibility assessments tailored to your unique synthetic challenges, ensuring a secure and cost-effective supply of your critical chiral building blocks.
