Advanced Quinoline Synthesis Routes for Scalable Camptothecin Production
Advanced Quinoline Synthesis Routes for Scalable Camptothecin Production
The pharmaceutical industry continuously seeks robust and scalable pathways for producing high-value anticancer agents, particularly camptothecin derivatives which function as potent topoisomerase I inhibitors. Patent CN101851195B introduces a groundbreaking methodology for synthesizing substituted quinoline compounds that serve as critical intermediates in the total synthesis of camptothecin alkaloids. This technology addresses the longstanding bottlenecks associated with relying solely on natural extraction from Camptotheca acuminata, offering a chemically elegant solution that bridges the gap between laboratory discovery and industrial application. By leveraging a concise eleven-step sequence starting from commercially accessible aniline and aldehyde precursors, this process ensures a stable supply of high-purity intermediates essential for developing next-generation oncology therapeutics.
As a leading entity in fine chemical manufacturing, we recognize that the structural complexity of camptothecin, characterized by its unique pentacyclic ring system containing a chiral center at position 20, demands precise synthetic control. The patented approach detailed in CN101851195B provides a reliable framework for constructing this intricate architecture without the variability inherent in agricultural sourcing. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and commercial viability of this route is paramount for securing a competitive edge in the global API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of camptothecin and its analogues has been heavily dependent on semi-synthetic methods derived from natural plant extracts. However, the content of camptothecin in plants like Camptotheca acuminata is notoriously low, leading to significant supply chain vulnerabilities and fluctuating costs driven by harvest seasons and geopolitical factors. Furthermore, existing total synthesis routes reported in prior art often suffer from excessive step counts, cumbersome purification processes, and the utilization of hazardous reagents that are sensitive to moisture and oxygen. These factors collectively inflate the cost of goods sold (COGS) and introduce substantial risks during the technology transfer from pilot scale to commercial manufacturing, rendering many potential drug candidates economically unviable despite their biological efficacy.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN101851195B presents a streamlined, eleven-step synthetic strategy that dramatically improves efficiency and operational simplicity. The core innovation lies in the construction of the quinoline scaffold through a condensation reaction between specific aldehyde and aniline derivatives, catalyzed by benign organic amines and acids rather than harsh inorganic reagents. This novel approach eliminates the need for air-sensitive metal catalysts, thereby reducing the complexity of reactor requirements and safety protocols. The route is designed with scalability in mind, utilizing robust oxidation steps with manganese dioxide and efficient cyclization reactions that proceed under mild conditions. By shortening the synthetic timeline and maximizing yield at each stage, this process offers a compelling value proposition for cost reduction in API manufacturing, ensuring a consistent and high-quality supply of critical quinoline intermediates.
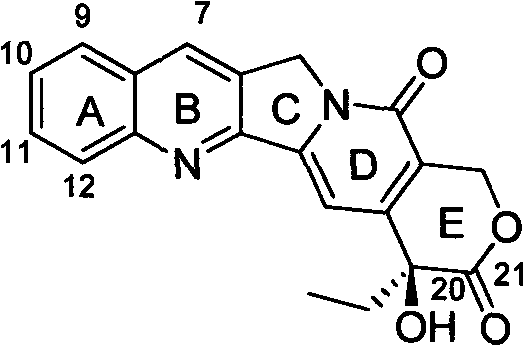
Mechanistic Insights into Quinoline Scaffold Construction and Functionalization
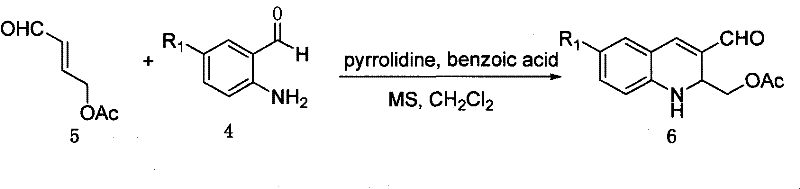
The chemical elegance of this patent is best understood through the detailed mechanistic progression from simple starting materials to the complex tetracyclic core. The synthesis initiates with a condensation reaction where compound 5, an acetoxy-substituted aldehyde, reacts with a substituted aniline (compound 4) in the presence of pyrrolidine and benzoic acid. This step effectively builds the quinoline ring system (compound 6) with high regioselectivity. Subsequent oxidation using activated manganese dioxide converts the intermediate into an aldehyde (compound 7), which is then transformed into an oxime (compound 8) using hydroxylamine hydrochloride. This sequence is critical for setting up the nitrogen functionality required for the later ring closures. The process continues with a reduction step using palladium on carbon to generate an amine, which is subsequently acylated to form an amide linkage, preparing the molecule for the pivotal intramolecular cyclization events that define the camptothecin skeleton.
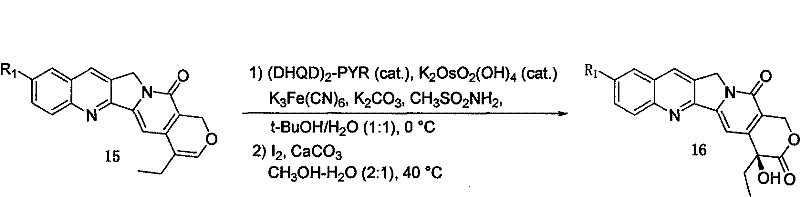
A particularly sophisticated aspect of this synthesis is the final stereoselective functionalization, which ensures the correct configuration at the chiral center. The pathway employs a Sharpless asymmetric dihydroxylation using ligands such as (DHQD)2-PYR and osmium tetroxide catalysts to introduce hydroxyl groups with high enantiomeric excess. This is followed by an iodine-mediated oxidative lactonization, which closes the E-ring to form the final lactone moiety characteristic of camptothecin. The ability to control stereochemistry at this late stage is vital for biological activity, as the S-configuration at C-20 is essential for topoisomerase I inhibition. The patent demonstrates that this complex transformation can be achieved with excellent yields and purity, validating the robustness of the chemical design for industrial replication.
How to Synthesize Quinoline Intermediates Efficiently
Executing this synthesis requires careful attention to reaction conditions and stoichiometry to maximize the yield of the quinoline intermediates. The process begins with the condensation of the aldehyde and aniline components in dichloromethane, utilizing molecular sieves to drive the equilibrium forward by removing water. Following the formation of the quinoline core, a series of oxidation and protection/deprotection steps are performed to install the necessary side chains. The detailed standardized synthesis steps见下方的指南 ensure that operators can replicate the high yields reported in the patent examples, such as the 82% yield in the initial condensation and the 89% yield in the manganese dioxide oxidation. Adhering to these protocols allows for the production of high-purity quinoline derivatives suitable for downstream processing into active pharmaceutical ingredients.
- Condensation of acetoxy-aldehyde derivatives with substituted anilines using pyrrolidine and benzoic acid catalysts to form the initial quinoline scaffold.
- Oxidation and functional group manipulation involving manganese dioxide and hydroxylamine to prepare the core for cyclization.
- Final asymmetric dihydroxylation using Sharpless catalysts followed by iodine-mediated lactonization to achieve the target camptothecin structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the decoupling of production from agricultural constraints. By shifting from plant extraction to total synthesis using petrochemical-derived starting materials, manufacturers can guarantee year-round production stability, immune to crop failures or seasonal price spikes. This reliability is crucial for maintaining continuous supply lines for life-saving oncology medications. Furthermore, the simplified operational profile of this route, which avoids cryogenic temperatures and pyrophoric reagents, lowers the barrier for entry for contract manufacturing organizations (CMOs), increasing the pool of qualified suppliers and fostering a more competitive pricing environment.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the significant shortening of the synthetic route and the elimination of expensive, sensitive catalysts. By utilizing common reagents like pyrrolidine, benzoic acid, and manganese dioxide, the direct material costs are drastically reduced compared to traditional methods requiring noble metals or specialized organometallics. Additionally, the high yields observed in key steps minimize waste generation and solvent consumption, leading to substantial cost savings in waste disposal and raw material procurement. The overall efficiency gains allow for a more favorable cost structure, enabling competitive pricing for the final API while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials such as substituted anilines and simple aldehydes ensures a robust upstream supply chain. These commodities are produced globally in vast quantities, mitigating the risk of shortages that often plague specialty chemical markets. The synthetic route's resilience to minor variations in reaction conditions further enhances supply security, as it reduces the likelihood of batch failures due to slight deviations in temperature or reagent quality. This stability allows supply chain planners to forecast demand with greater accuracy and maintain optimal inventory levels without the fear of sudden supply disruptions.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with green chemistry principles. The avoidance of heavy metal contaminants simplifies the purification process and reduces the burden on wastewater treatment facilities, facilitating easier compliance with stringent environmental regulations. The operational simplicity, characterized by reactions that can be performed at room temperature or with moderate heating, reduces energy consumption significantly. These factors combined make the commercial scale-up of complex pharmaceutical intermediates not only technically feasible but also environmentally sustainable, appealing to stakeholders focused on corporate social responsibility and long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial implications of the quinoline synthesis method described in patent CN101851195B. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on how this technology compares to existing standards in the industry. Understanding these details is essential for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: Unlike natural extraction which is limited by plant resource availability and seasonal variations, this patented synthetic method utilizes readily available raw materials. It offers a consistent supply chain, higher overall yields, and avoids the ecological impact of harvesting rare plants like Camptotheca acuminata.
Q: Does this process require air-sensitive or expensive metal reagents?
A: No, a key innovation of this patent is the avoidance of metal reagents that are sensitive to air and water. The process utilizes robust catalysts like pyrrolidine, benzoic acid, and manganese dioxide, significantly simplifying operational requirements and reducing safety hazards associated with handling pyrophoric materials.
Q: Is this methodology suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights the suitability for industrial amplification. The route consists of only eleven steps with high individual step yields (often exceeding 80%), simple operation conditions such as room temperature reactions, and standard workup procedures like filtration and column chromatography, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camptothecin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring this advanced quinoline synthesis technology to life on a global scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity quinoline derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex multi-step syntheses involving sensitive intermediates positions us as an ideal partner for pharmaceutical companies seeking to secure their supply of critical anticancer drug precursors.
We invite you to collaborate with us to explore the full potential of this synthetic route for your specific product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this methodology can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your mission to deliver life-saving therapies to patients worldwide.
