Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Intermediates for Commercial Production
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Intermediates for Commercial Production
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. A significant breakthrough in this domain is detailed in patent CN116253692A, which discloses a highly efficient preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds. These scaffolds are critical building blocks in modern drug discovery, known for enhancing metabolic stability and lipophilicity in lead compounds. The disclosed methodology represents a paradigm shift from traditional transition-metal catalyzed processes to a benign, base-promoted cycloaddition strategy. By leveraging cheap and readily available starting materials like chlorohydrazones and trifluoroacetyl sulfur ylides, this innovation addresses key pain points in process chemistry, specifically regarding cost, safety, and environmental impact. For R&D teams and procurement specialists alike, understanding this technology is vital for securing a reliable pharma intermediate supplier capable of delivering high-purity materials without the baggage of heavy metal contamination.
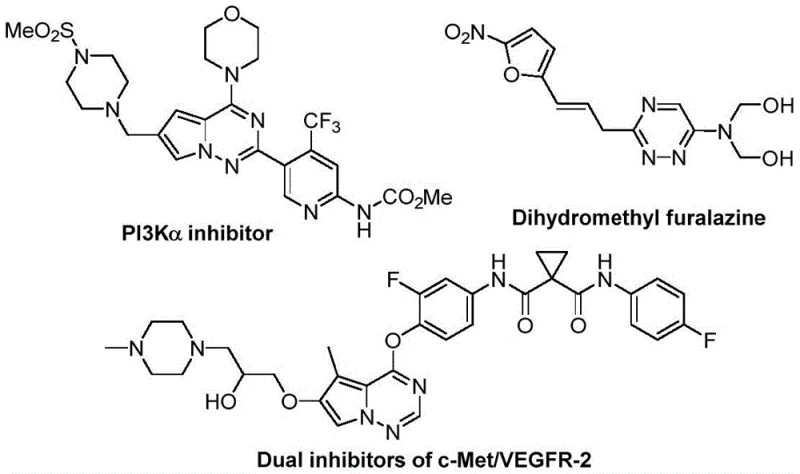
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied on condensation reactions between amidrazones and 1,2-diketones or alkynes, often necessitating harsh thermal conditions or stoichiometric amounts of dehydrating agents. Alternative multicomponent reactions involving hydrazides and dicarbonyl compounds frequently suffer from poor atom economy and limited substrate scope, restricting the structural diversity accessible to medicinal chemists. Furthermore, many contemporary cycloaddition strategies depend heavily on precious metal catalysts such as palladium, rhodium, or copper to drive the reaction forward. While effective on a milligram scale, these metal-dependent routes introduce severe complications during commercial scale-up, including the high cost of catalyst recovery and the stringent regulatory limits on residual metal levels in active pharmaceutical ingredients (APIs). The reliance on sensitive organometallic species also often mandates inert atmospheres and anhydrous conditions, significantly increasing operational complexity and energy consumption in a manufacturing setting.
The Novel Approach
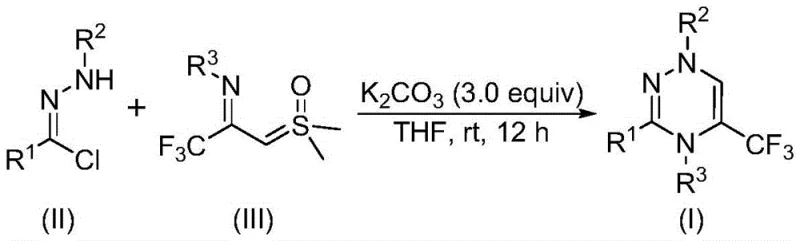
In stark contrast, the novel approach outlined in the patent utilizes a synergistic [3+3] cycloaddition between a nitrile imine intermediate, generated in situ from a chlorohydrazone, and a trifluoroacetyl sulfur ylide. This transformation is promoted solely by potassium carbonate, an inexpensive and non-toxic inorganic base that is ubiquitous in industrial chemistry. The reaction proceeds smoothly at room temperature (20-40°C) under an ambient air atmosphere, eliminating the need for specialized equipment like gloveboxes or high-pressure reactors. This metal-free protocol not only drastically simplifies the workup procedure—often requiring only filtration and standard chromatography—but also ensures that the final product is free from toxic metal residues. The broad tolerance for various functional groups on the aromatic rings of the starting materials allows for the rapid generation of diverse libraries of trifluoromethyl-triazines, making it an ideal platform for both lead optimization and process development.
Mechanistic Insights into Base-Promoted [3+3] Cycloaddition
The mechanistic pathway of this transformation is elegant in its simplicity yet powerful in its execution. Initially, the chlorohydrazone undergoes dehydrohalogenation in the presence of potassium carbonate to generate a highly reactive nitrile imine dipole. This transient species then engages in a concerted [3+3] cycloaddition with the electron-deficient trifluoroacetyl sulfur ylide. The sulfur ylide acts as a three-atom synthon, contributing the necessary carbon and nitrogen atoms to close the six-membered triazine ring while expelling dimethyl sulfoxide as a benign byproduct. This cascade effectively constructs the heterocyclic core and installs the crucial trifluoromethyl group in a single operation. The absence of radical intermediates or high-energy transition states explains the mild reaction conditions and the high chemoselectivity observed across a wide range of substrates. Understanding this mechanism is crucial for process engineers aiming to optimize reaction kinetics and minimize side-product formation during scale-up.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Since no transition metals are involved, there is no risk of metal-mediated side reactions such as homocoupling or C-H activation that often plague complex molecule synthesis. The primary byproducts are inorganic salts (potassium chloride) and dimethyl sulfoxide, both of which are easily removed during aqueous workup or crystallization. This clean reaction profile translates directly to higher crude purity, reducing the burden on downstream purification units. For quality control laboratories, this means simpler analytical methods and faster release times for batches. The robustness of the nitrile imine generation step ensures consistent conversion rates, provided that the stoichiometry of the base is maintained, thereby guaranteeing batch-to-batch reproducibility which is a cornerstone of Good Manufacturing Practice (GMP).
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and plant equipment. The process begins with the precise weighing of chlorohydrazone and trifluoroacetyl sulfur ylide, typically in a molar ratio favoring the ylide to ensure complete consumption of the hydrazone. These solids are suspended in an aprotic organic solvent, with tetrahydrofuran (THF) being the preferred medium due to its ability to solubilize both reactants and the inorganic base effectively. Potassium carbonate is then added, usually in a slight excess (e.g., 3.0 equivalents) to drive the dehydrohalogenation to completion. The mixture is stirred at ambient temperature for a period ranging from 10 to 14 hours, allowing the cycloaddition to proceed to full conversion without external heating. Detailed standardized synthesis steps follow below.
- Mix potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent such as THF.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of commodities like palladium or rhodium. Furthermore, the use of commodity chemicals like potassium carbonate and common solvents ensures that raw material sourcing is stable and geographically diverse, reducing the risk of supply chain disruptions. The mild reaction conditions also imply lower energy consumption, as there is no need for cryogenic cooling or high-temperature heating, contributing to a smaller carbon footprint and reduced utility costs. These factors collectively enhance the overall cost-efficiency of manufacturing high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the substitution of expensive catalytic systems with inexpensive inorganic bases. By removing the requirement for ligand synthesis and metal recovery processes, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the simplified workup procedure reduces the consumption of silica gel and eluents during purification, further driving down operational expenses. The high atom economy of the [3+3] cycloaddition minimizes waste generation, leading to substantial cost savings in waste disposal and treatment. This economic efficiency makes the process highly attractive for the commercial scale-up of complex pharmaceutical intermediates where margin pressure is often intense.
- Enhanced Supply Chain Reliability: Reliance on specialized catalysts often creates bottlenecks in the supply chain, particularly when global demand surges. This new method utilizes universally available reagents that can be sourced from multiple vendors worldwide, ensuring continuity of supply. The robustness of the reaction under air atmosphere means that production is less susceptible to delays caused by inert gas shortages or equipment failures related to nitrogen lines. Moreover, the stability of the starting materials allows for longer shelf-life and easier storage logistics, enabling manufacturers to maintain strategic stockpiles without degradation concerns. This resilience is critical for maintaining just-in-time delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety hazards, but this protocol operates safely at room temperature, minimizing the risk of thermal runaways. The absence of toxic heavy metals simplifies regulatory compliance, as there is no need for extensive testing and validation of metal clearance steps. The byproducts generated are environmentally benign, aligning with green chemistry principles and facilitating easier permitting for new production lines. This ease of scale-up from gram to kilogram and eventually to tonnage quantities ensures that the technology can meet growing market demands without requiring massive capital investment in specialized reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what potential partners and licensees can expect. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN116253692A utilizes inexpensive potassium carbonate as a promoter, completely eliminating the need for costly heavy metal catalysts and simplifying downstream purification.
Q: What are the optimal reaction conditions for this cycloaddition?
A: The reaction proceeds efficiently at room temperature (20-40°C) in an air atmosphere using tetrahydrofuran (THF) as the preferred solvent, requiring approximately 10 to 14 hours for completion.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the protocol is designed for scalability, utilizing cheap, non-toxic reagents and mild conditions that avoid the safety hazards associated with high-pressure or high-temperature reactions, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free heterocycle synthesis in accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to industrial reality. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of trifluoromethyl-substituted 1,2,4-triazine meets the highest international standards. We are committed to delivering high-purity OLED material and pharmaceutical intermediates that empower your innovation.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and reduce your time to market.
