Advanced Photocatalytic Synthesis of C-3 Difluoromethyl Quinoxalinone Derivatives for Commercial Scale Production
Advanced Photocatalytic Synthesis of C-3 Difluoromethyl Quinoxalinone Derivatives for Commercial Scale Production
The pharmaceutical industry continuously seeks efficient and sustainable pathways to access complex heterocyclic scaffolds, particularly those containing fluorine motifs which are crucial for enhancing metabolic stability and bioavailability. Patent CN110105293B discloses a groundbreaking synthetic methodology for preparing C-3-position difluoromethyl substituted quinoxalinone derivatives, a class of compounds renowned for their potent pharmacological activities including anti-tumor and anti-fungal properties. This technology leverages visible light as a clean energy source to drive the difluoroalkylation process, marking a significant departure from traditional thermal or stoichiometric oxidant-dependent protocols. By utilizing a photocatalytic system involving iridium complexes or organic dyes, the process achieves high selectivity and yield under remarkably mild conditions, typically ranging from 20°C to 60°C. For R&D directors and process chemists, this represents a viable strategy for diversifying quinoxalinone libraries with minimal environmental impact. The ability to introduce difluoromethyl ester or amide groups directly at the C-3 position expands the chemical space available for drug discovery programs targeting serious diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the C-3 position on quinoxalinone cores has relied heavily on transition metal catalysis or harsh oxidative conditions that pose significant challenges for large-scale manufacturing. Prior art methods often necessitate the use of expensive and toxic metals such as copper, silver, or iron, which require rigorous removal steps to meet stringent pharmaceutical purity standards regarding heavy metal residues. Furthermore, many existing protocols depend on stoichiometric amounts of strong oxidants to facilitate the coupling reaction, leading to the generation of substantial quantities of chemical waste and raising safety concerns related to exothermic events. The requirement for elevated temperatures or specialized ligands further complicates the operational simplicity, increasing both the energy consumption and the overall cost of goods sold. Additionally, the substrate scope in conventional methods is frequently limited, struggling to accommodate sensitive functional groups that might decompose under aggressive reaction conditions. These limitations collectively hinder the rapid scale-up and cost-effective production of diverse fluorinated quinoxalinone intermediates needed for modern drug development pipelines.
The Novel Approach
In stark contrast, the novel photocatalytic approach detailed in the patent utilizes visible light irradiation to activate the bromodifluoromethyl reagent, generating reactive radical species under ambient or near-ambient temperatures. This method eliminates the need for stoichiometric oxidants and significantly reduces the reliance on precious transition metals, often requiring only catalytic loading of the photocatalyst such as Ir(ppy)3. The reaction proceeds smoothly in common organic solvents like acetonitrile or dichloromethane, compatible with a wide array of substituents including electron-withdrawing nitro groups and electron-donating methoxy groups. By operating at temperatures between 20°C and 60°C, the process minimizes thermal degradation of sensitive substrates and reduces the energy footprint of the manufacturing operation. The use of simple inorganic bases like potassium carbonate and readily available organic amines further streamlines the reagent profile, making the process highly attractive for cost reduction in pharmaceutical intermediate manufacturing. This green chemistry approach not only enhances safety but also simplifies the downstream purification process, resulting in higher overall efficiency and product quality.
Mechanistic Insights into Visible Light Photocatalytic Difluoromethylation
The core of this transformative synthesis lies in the photoredox catalytic cycle initiated by the absorption of visible light by the photocatalyst, typically an iridium complex like Ir(ppy)3 or an organic dye such as Rose Bengal. Upon irradiation, the photocatalyst transitions to an excited state, possessing sufficient redox potential to engage in single-electron transfer (SET) processes with the bromodifluoromethyl compound. This interaction facilitates the homolytic cleavage of the carbon-bromine bond, generating a nucleophilic difluoromethyl radical species along with a bromide anion. The generated radical then adds selectively to the C-3 position of the quinoxalinone ring, forming a transient radical intermediate that is subsequently oxidized to restore aromaticity and yield the final difluoromethylated product. The presence of an inorganic base and an organic amine plays a critical role in neutralizing acidic byproducts and facilitating the turnover of the catalytic cycle. This mechanism avoids the high-energy barriers associated with thermal activation, allowing the reaction to proceed with exceptional chemoselectivity even in the presence of other reactive functional groups.
From an impurity control perspective, the mild nature of the photocatalytic conditions inherently suppresses the formation of thermal decomposition byproducts and oligomerization side reactions that often plague high-temperature processes. The specificity of the radical addition ensures that the difluoromethyl group is installed precisely at the C-3 position without affecting the N-substituents or the carbonyl functionality of the quinoxalinone core. Furthermore, the absence of strong oxidants prevents the over-oxidation of sensitive moieties such as sulfides or amines that might be present on the substrate. The reaction system is designed to be robust, with the photocatalyst loading optimized to balance reaction rate and cost, typically ranging from 0.5% to 2% molar ratio. Post-reaction workup involves standard aqueous extraction and column chromatography, which effectively removes the photocatalyst and inorganic salts, ensuring the final API intermediate meets rigorous purity specifications. This level of control is essential for maintaining batch-to-batch consistency in commercial production environments.
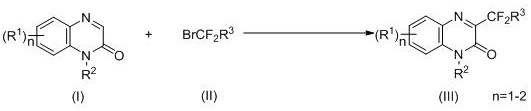
How to Synthesize C-3 Difluoromethyl Quinoxalinone Efficiently
To implement this synthesis effectively, one must carefully control the stoichiometry of the bromodifluoromethyl reagent, which is typically used in a slight excess relative to the quinoxalinone substrate to drive the reaction to completion. The choice of solvent is pivotal, with acetonitrile often providing superior solubility and reaction kinetics compared to dichloromethane, although both are viable options depending on the specific substrate properties. Maintaining an inert atmosphere, such as nitrogen, is recommended to prevent quenching of the excited photocatalyst by oxygen, which could otherwise lead to reduced yields. The reaction time can vary from 6 to 36 hours depending on the light intensity and the electronic nature of the substituents on the quinoxalinone ring. Detailed standardized synthesis steps see the guide below.
- Dissolve the quinoxalinone derivative, bromodifluoromethyl compound, photocatalyst (e.g., Ir(ppy)3), inorganic base, and organic amine in an organic solvent like acetonitrile.
- Maintain the reaction mixture under nitrogen atmosphere and irradiate with visible light (blue or white) at a temperature between 20-60°C for 6 to 36 hours.
- Upon completion, perform aqueous workup, extract with organic solvent, dry over sodium sulfate, and purify the residue via column chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers tangible benefits in terms of cost structure and operational reliability. The shift away from expensive transition metal catalysts and stoichiometric oxidants directly translates to a reduction in raw material costs and waste disposal fees. The mild reaction conditions reduce the demand for high-energy heating or cooling systems, contributing to lower utility costs during manufacturing. Furthermore, the use of commercially available and stable reagents ensures a secure supply chain, minimizing the risk of production delays caused by the scarcity of specialized chemicals. The scalability of the process is enhanced by the simplicity of the equipment required, as standard photoreactors can be employed without the need for high-pressure vessels or complex containment systems. These factors collectively support a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like palladium or rhodium, and the replacement of stoichiometric oxidants with catalytic visible light energy, significantly lowers the direct material costs associated with production. By avoiding the need for extensive heavy metal scavenging steps, the downstream processing becomes less resource-intensive, reducing the consumption of specialized resins and solvents. The mild temperature range of 20-60°C minimizes energy consumption for heating and cooling, leading to substantial operational expenditure savings over the lifecycle of the product. Additionally, the high atom economy of the radical addition reaction ensures that a greater proportion of the starting materials are incorporated into the final product, reducing waste generation and associated disposal costs.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including bromodifluoromethyl esters and common inorganic bases, are widely available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the photocatalytic system means that minor variations in raw material quality are less likely to cause batch failures, ensuring consistent output and reliable delivery schedules. The simplified reaction setup reduces the complexity of the manufacturing infrastructure, allowing for faster turnaround times and greater flexibility in responding to market demand fluctuations. This reliability is crucial for maintaining continuous supply lines for critical pharmaceutical intermediates, preventing costly stockouts and production bottlenecks.
- Scalability and Environmental Compliance: The process is inherently scalable, as the use of visible light allows for efficient photon flux management in larger reactors without the safety risks associated with thermal runaways. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, simplifying the permitting process and reducing the liability associated with chemical disposal. The use of green solvents and the absence of toxic heavy metals make the process more sustainable, enhancing the corporate social responsibility profile of the manufacturing operation. This environmental compliance not only mitigates regulatory risk but also appeals to eco-conscious partners and customers in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industrial application. Understanding these details is critical for process engineers and quality assurance teams evaluating the feasibility of adopting this technology for commercial production.
Q: What are the advantages of using visible light photocatalysis for quinoxalinone functionalization?
A: Visible light photocatalysis offers a greener and safer alternative to traditional methods that often require harsh oxidants or expensive transition metal catalysts. It operates under mild temperatures (20-60°C) and reduces the generation of hazardous waste.
Q: Which photocatalysts are suitable for this difluoromethylation reaction?
A: The patent specifies several effective photocatalysts including Ir(ppy)3 (tris(2-phenylpyridine)iridium), Eosin Y, and Rose Bengal. Ir(ppy)3 is particularly preferred for achieving high yields and selectivity.
Q: Can this method tolerate various substituents on the quinoxalinone ring?
A: Yes, the method demonstrates broad substrate scope. It tolerates various substituents such as methyl, methoxy, nitro, fluoro, and chloro groups on the benzene ring, as well as different N-substituents like alkyl, benzyl, and ester groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxalinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the visible-light driven difluoromethylation described in CN110105293B for developing next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinoxalinone derivatives meets the highest international standards for pharmaceutical intermediates. Our commitment to quality and technical excellence makes us a trusted partner for companies seeking to leverage cutting-edge chemistry for their drug development pipelines.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements with customized solutions. Request a Customized Cost-Saving Analysis to understand how implementing this photocatalytic route can optimize your budget without compromising quality. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions about your supply chain strategy. Partner with us to secure a reliable source of high-quality C-3 difluoromethyl quinoxalinone derivatives for your future success.
