Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic methodologies to access fluorinated heterocycles, which are critical motifs in modern drug design due to their enhanced metabolic stability and lipophilicity. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing significant limitations in existing synthetic routes. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing readily available trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts as key building blocks. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this protocol offers a compelling alternative to hazardous diazo-based chemistries. The process operates under remarkably mild conditions, typically at 30°C, ensuring high reaction efficiency and broad substrate compatibility. By eliminating the need for high-pressure carbon monoxide gas and unstable diazo reagents, this innovation significantly de-risks the manufacturing process while maintaining high purity standards essential for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl groups has relied heavily on synthons such as trifluorodiazoethane. While effective in certain contexts, these traditional approaches present severe safety and operational challenges for large-scale manufacturing. Trifluorodiazoethane is inherently unstable and potentially explosive, requiring specialized handling equipment and stringent safety protocols that drastically increase capital expenditure and operational complexity. Furthermore, conventional methods often suffer from poor functional group tolerance, limiting the structural diversity accessible to medicinal chemists during lead optimization phases. The reliance on harsh reaction conditions or toxic reagents also complicates waste management and environmental compliance, creating bottlenecks for cost reduction in API manufacturing. These factors collectively hinder the rapid scale-up of promising drug candidates, forcing supply chain heads to manage volatile raw material sources and extended lead times for high-purity intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN111423381A utilizes a safe and efficient palladium-catalyzed carbonylation strategy that circumvents the pitfalls of diazo chemistry. By employing trifluoroethylimidoyl chloride and diaryliodonium salts, the reaction proceeds through a controlled cascade mechanism that constructs the imidazole core with precision. As illustrated in the general reaction scheme below, the process integrates C-N bond formation, isomerization, and carbonylation into a single pot operation. This streamlining not only reduces the number of unit operations but also enhances the overall atom economy of the synthesis. The use of a formic acid and acetic anhydride mixture serves as a convenient carbon monoxide surrogate, eliminating the need for dangerous high-pressure gas cylinders. This approach enables the synthesis of diversified 2-trifluoromethyl imidazoles with excellent yields, as demonstrated by the successful preparation of various derivatives shown in the subsequent examples.
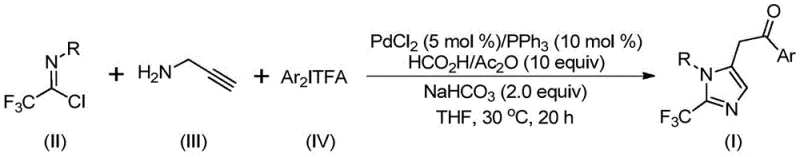
The versatility of this novel approach is further evidenced by its ability to accommodate a wide range of substituents on both the imidoyl chloride and the diaryliodonium salt components. Whether incorporating electron-donating groups like methoxy or electron-withdrawing groups like nitro and halogens, the system maintains high conversion rates. This flexibility is paramount for generating diverse libraries of high-purity pharmaceutical intermediates required for structure-activity relationship (SAR) studies. Moreover, the reaction conditions are sufficiently mild to preserve sensitive functional groups that might otherwise degrade under traditional harsh cyclization conditions. For procurement teams, this translates to a more resilient supply chain where raw material variability does not compromise the final product quality. The ability to synthesize complex scaffolds like those found in antihistamines or kinase inhibitors using this robust platform underscores its potential as a cornerstone technology for modern fine chemical production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align the alkyne moiety for palladium coordination. The palladium catalyst, generated in situ from palladium chloride and triphenylphosphine, facilitates the aminopalladation of the alkyne, forming a key alkenyl-palladium intermediate. Following another isomerization step to an alkyl-palladium species, the system engages in carbonylation. Crucially, the carbon monoxide required for this step is released in situ from the decomposition of the formic acid and acetic anhydride mixture, ensuring a steady, low-concentration supply of CO that drives the reaction forward without necessitating high-pressure equipment. This generates an acyl-palladium intermediate, which is then subjected to oxidative addition by the diaryliodonium salt.
The final stages of the catalytic cycle involve the formation of a tetravalent palladium intermediate followed by reductive elimination to release the desired 2-trifluoromethyl substituted imidazole compound and regenerate the active palladium catalyst. This intricate dance of organometallic steps ensures high regioselectivity and minimizes the formation of side products, which is vital for maintaining a clean impurity profile. The choice of sodium bicarbonate as an additive plays a dual role in neutralizing acidic byproducts and facilitating the initial nucleophilic attack. For quality control laboratories, understanding this pathway allows for better prediction of potential impurities, such as unreacted starting materials or incomplete cyclization products. The robustness of the catalytic cycle against various electronic environments on the aryl rings explains the broad substrate scope observed in the experimental data. This mechanistic clarity provides a solid foundation for scaling the reaction from milligram discovery batches to multi-kilogram commercial production runs with confidence.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible even for facilities without specialized high-pressure reactors. The protocol involves charging a reaction vessel with the palladium catalyst system, the CO surrogate mixture, and the specific substrates in a suitable organic solvent such as tetrahydrofuran (THF). The reaction is then stirred at a mild temperature of 30°C for a duration of 16 to 24 hours, allowing sufficient time for the cascade to reach completion. Post-reaction workup is straightforward, involving simple filtration to remove inorganic salts followed by silica gel column chromatography to isolate the pure product. This simplicity significantly reduces the technical barrier for adoption, enabling rapid technology transfer from R&D to pilot plants. For detailed standard operating procedures and specific stoichiometric ratios tailored to your specific substrate requirements, please refer to the comprehensive guide below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial strategic advantages for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The primary driver of cost efficiency lies in the substitution of expensive and hazardous reagents with commodity chemicals. Trifluoroethylimidoyl chlorides and diaryliodonium salts are generally more stable and easier to source in bulk quantities compared to specialized diazo compounds. Furthermore, the use of a palladium chloride and triphenylphosphine catalyst system utilizes widely available transition metals, avoiding the need for exotic ligands that can drive up raw material costs. The mild reaction temperature of 30°C implies significantly lower energy consumption for heating and cooling compared to processes requiring reflux or cryogenic conditions. This energy efficiency directly correlates to reduced utility costs per kilogram of product, enhancing the overall margin profile for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure represents a major capital expenditure saving. Traditional carbonylation reactions often require autoclaves rated for high pressures, which are expensive to purchase, maintain, and certify. By generating CO in situ from liquid reagents, this process can be run in standard glass-lined or stainless steel reactors, drastically lowering the barrier to entry for production. Additionally, the high reaction efficiency and yields reported in the patent minimize the loss of valuable starting materials, ensuring that the cost of goods sold (COGS) remains competitive. The simplified purification process, relying on standard column chromatography or crystallization rather than complex distillation setups, further reduces processing time and solvent consumption.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents. This method relies on starting materials that are either commercially available or easily synthesized from abundant aromatic amines and boronic acids. The broad substrate tolerance means that if a specific aryl group becomes unavailable, alternative substituents can often be used without re-optimizing the entire process, providing flexibility in raw material sourcing. The robustness of the reaction conditions also reduces the risk of batch failures due to minor fluctuations in temperature or mixing, ensuring consistent delivery schedules. For supply chain heads, this reliability translates to reduced safety stock requirements and improved cash flow management.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scalability. The absence of explosive diazo intermediates removes a significant safety hazard associated with scale-up, simplifying the regulatory approval process for new manufacturing sites. The use of THF as a solvent, while requiring recovery systems, is well-established in the industry with mature recycling technologies. Moreover, the high atom economy of the cascade reaction means less waste generation per unit of product, aligning with green chemistry principles and reducing waste disposal costs. This environmental friendliness is increasingly important for meeting the sustainability goals of major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio. Whether you are concerned about catalyst loading, solvent selection, or product purity, the answers below provide a foundational understanding of the process capabilities.
Q: What are the key advantages of this palladium-catalyzed method over traditional diazo-based routes?
A: This method avoids the use of unstable and potentially explosive trifluorodiazoethane, utilizing safer trifluoroethylimidoyl chlorides instead. It operates under mild conditions (30°C) with excellent functional group tolerance.
Q: Can this synthesis be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is scalable to gram levels and uses commercially available, cheap starting materials, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of the formic acid and acetic anhydride mixture in this reaction?
A: The mixture acts as a carbon monoxide surrogate, releasing CO in situ to facilitate the crucial carbonylation step within the palladium catalytic cycle without requiring high-pressure gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned to industrial reality. We possess the technical capability to implement the palladium-catalyzed carbonylation route described in CN111423381A, adhering to stringent purity specifications required for GMP manufacturing. Our rigorous QC labs are equipped to analyze complex fluorinated heterocycles, guaranteeing that every batch of 2-trifluoromethyl imidazole meets the highest quality standards. By leveraging our infrastructure, partners can bypass the lengthy process validation phases typically associated with novel chemistries.
We invite you to collaborate with us to explore how this efficient synthesis can optimize your supply chain and reduce overall project costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us to request specific COA data for similar fluorinated intermediates and discuss route feasibility assessments for your upcoming projects. Together, we can secure a reliable supply of high-value pharmaceutical intermediates that drive innovation in the global healthcare market.
