Advanced Group 4 Catalyzed Esterification for High-Purity Cyclopropane Carboxylate Manufacturing and Commercial Scale-Up
The chemical industry is constantly seeking more efficient and cleaner pathways for synthesizing critical intermediates, and patent CN1210248C presents a groundbreaking advancement in the preparation of cyclopropane carboxylates. This intellectual property details a sophisticated esterification process that leverages catalysts containing Group 4 elements from the periodic table, specifically targeting the reaction between cyclopropanecarboxylic acids and monohydric hydroxy compounds. Unlike traditional methods that rely on harsh protonic acids, this novel approach utilizes Lewis acidic metal centers to drive the dehydration reaction with exceptional precision. The resulting esters, represented generally by Formula (1), are pivotal building blocks in the synthesis of high-value agrochemicals and pharmaceuticals, where structural integrity and purity are paramount for biological efficacy. By shifting the catalytic paradigm from Brønsted acids to transition metal complexes, this technology addresses long-standing issues regarding product discoloration and impurity profiles that have plagued industrial manufacturing for decades.
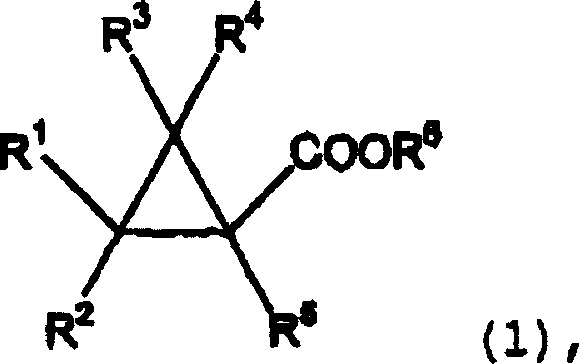
For procurement specialists and supply chain managers, the implications of adopting this Group 4 catalyzed methodology extend far beyond mere chemical curiosity; it represents a tangible opportunity for cost reduction in agrochemical intermediate manufacturing. The ability to produce high-purity cyclopropane carboxylates without the extensive downstream purification steps typically required to remove mineral acid residues translates directly into operational efficiency. Furthermore, the versatility of the substrate scope, allowing for various substituted alkyl and aryl groups on both the acid and alcohol components, ensures that this single platform technology can service a diverse portfolio of fine chemical products. As a reliable agrochemical intermediate supplier, understanding and implementing such patented innovations is crucial for maintaining a competitive edge in a market that increasingly demands sustainability and quality assurance alongside cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the esterification of carboxylic acids with alcohols has been predominantly achieved using strong protic acid catalysts such as concentrated sulfuric acid or p-toluenesulfonic acid, as documented in prior art like Japanese Patent Laid-Open No. 9-188649. While these methods are chemically feasible, they suffer from significant drawbacks that render them suboptimal for modern industrial applications, particularly when dealing with sensitive substrates like cyclopropane derivatives. The intense acidity of these traditional catalysts often promotes undesirable side reactions, including polymerization, rearrangement, or degradation of the delicate cyclopropane ring structure, leading to complex impurity profiles that are difficult to separate. Moreover, the use of strong mineral acids frequently results in conspicuous discoloration of the final product, necessitating additional decolorization steps such as activated carbon treatment or extensive recrystallization, which inevitably reduce overall yield and increase processing time. These inefficiencies accumulate to create substantial bottlenecks in production schedules and inflate the cost of goods sold, making the conventional protonic acid route increasingly unattractive for high-volume commercial scale-up of complex polymer additives or active ingredients.
The Novel Approach
In stark contrast to the brute force approach of mineral acids, the method disclosed in CN1210248C introduces a refined catalytic system based on compounds comprising elements from Group 4 of the periodic table, such as titanium, zirconium, and hafnium. This novel approach fundamentally alters the reaction mechanism by employing Lewis acidity rather than Brønsted acidity, thereby activating the carbonyl group of the carboxylic acid through coordination without generating the highly corrosive and reactive proton environment associated with sulfuric acid. The result is a remarkably clean reaction profile that minimizes side reactions and preserves the stereochemical integrity of the cyclopropane ring, which is often critical for the biological activity of the final agrochemical or pharmaceutical agent. By operating under these milder yet highly effective catalytic conditions, manufacturers can achieve superior selectivity and yield, often exceeding 90% in optimized examples, while simultaneously eliminating the need for rigorous neutralization and washing steps required to remove residual strong acids. This technological shift not only enhances product quality but also streamlines the workflow, offering a robust solution for the commercial scale-up of complex organic intermediates.
Mechanistic Insights into Group 4 Lewis Acid-Catalyzed Esterification
The mechanistic superiority of this process lies in the specific interaction between the Group 4 metal center and the oxygen atoms of the reactants, creating a highly organized transition state that favors ester formation. When a catalyst such as zirconium tetrachloride or titanium isopropoxide is introduced into the reaction mixture, the metal atom acts as a potent Lewis acid, coordinating with the lone pair electrons on the carbonyl oxygen of the cyclopropanecarboxylic acid substrate. This coordination significantly increases the electrophilicity of the carbonyl carbon, making it much more susceptible to nucleophilic attack by the hydroxyl group of the alcohol reactant. Unlike protonation, which can lead to indiscriminate activation of various functional groups and subsequent decomposition, this metal-mediated activation is more selective and controlled, reducing the likelihood of acid-catalyzed degradation pathways that compromise product purity. The formation of the tetrahedral intermediate is thus facilitated under conditions that are thermodynamically favorable yet kinetically controlled to prevent unwanted side reactions, ensuring that the reaction proceeds efficiently towards the desired ester product.
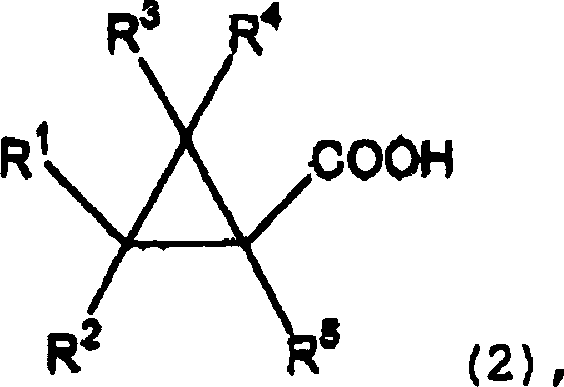
Furthermore, the choice of Group 4 catalysts offers distinct advantages in terms of impurity control and downstream processing, which are critical concerns for R&D directors focused on quality assurance. Since the catalysts are typically metal halides or alkoxides, they can be effectively deactivated and removed from the reaction mixture simply by washing with water or mild acidic aqueous solutions, converting the metal species into water-soluble hydroxides or oxides that partition into the aqueous phase. This ease of removal stands in sharp contrast to organic sulfonic acids which may require multiple extraction cycles or chromatographic purification to eliminate trace residues that could affect the stability or toxicity profile of the final API. Additionally, the absence of strong protons in the reaction medium prevents the acid-catalyzed isomerization of double bonds or the opening of the strained cyclopropane ring, phenomena that are common pitfalls in traditional esterification protocols. Consequently, the impurity spectrum of the product obtained via this Group 4 catalyzed route is significantly cleaner, simplifying the analytical burden and ensuring consistent batch-to-batch reproducibility essential for regulatory compliance in regulated industries.
How to Synthesize Cyclopropane Carboxylate Efficiently
The practical implementation of this patented synthesis route involves a straightforward yet precise set of operational parameters designed to maximize conversion while maintaining safety and efficiency. The process begins with the charging of a suitable reactor, typically equipped with a Dean-Stark trap or similar water-removal apparatus, with the cyclopropanecarboxylic acid substrate, the chosen monohydric alcohol, and a catalytic amount of the Group 4 element compound. The reaction is then heated, preferably under an inert atmosphere such as nitrogen or argon to prevent oxidation, to temperatures ranging from approximately 20°C to 200°C, with reflux conditions often being optimal to drive the equilibrium forward by continuously removing the water byproduct. Detailed standardized synthetic steps see the guide below.
- Charge a reactor with cyclopropanecarboxylic acid, a monohydric alcohol, and a catalytic amount of a Group 4 element compound such as zirconium tetrachloride.
- Heat the reaction mixture to reflux temperatures between 20°C and 200°C while continuously removing water byproduct using a Dean-Stark trap or azeotropic distillation.
- Upon completion, wash the reaction mixture with water or acidic aqueous solution to remove the catalyst, followed by standard isolation techniques like distillation or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Group 4 catalyzed esterification technology offers compelling strategic advantages that directly impact the bottom line and operational resilience. The primary benefit stems from the elimination of corrosive mineral acids, which drastically simplifies the equipment requirements by reducing the need for specialized glass-lined or Hastelloy reactors capable of withstanding extreme pH conditions, thereby lowering capital expenditure and maintenance costs. Moreover, the simplified workup procedure, which relies on basic aqueous washing rather than complex neutralization and extraction sequences, significantly reduces the volume of chemical waste generated, aligning with increasingly stringent environmental regulations and reducing disposal costs. This streamlined process flow also shortens the overall cycle time per batch, enhancing throughput capacity without the need for additional hardware, which is a critical factor in meeting tight delivery schedules for high-purity intermediates.
- Cost Reduction in Manufacturing: The transition from traditional strong acid catalysts to Group 4 Lewis acids eliminates the necessity for expensive corrosion-resistant infrastructure and reduces the consumption of neutralizing agents, leading to substantial cost savings in raw materials and utilities. By avoiding the formation of colored byproducts and tars, the process minimizes the loss of yield during purification steps such as filtration or charcoal treatment, ensuring that a higher percentage of input materials are converted into saleable product. Furthermore, the catalytic nature of the metal complexes means that only small quantities are required to achieve high turnover, reducing the direct material cost associated with the catalyst itself compared to stoichiometric reagents often used in older methods.
- Enhanced Supply Chain Reliability: The catalysts employed in this process, such as zirconium tetrachloride and titanium alkoxides, are commercially available commodity chemicals with stable and robust global supply chains, mitigating the risk of raw material shortages that can plague proprietary or exotic catalyst systems. The tolerance of the reaction to a wide range of substrates allows for flexibility in sourcing different grades of starting alcohols and acids, providing procurement teams with the leverage to negotiate better pricing or switch suppliers without revalidating the entire process. Additionally, the improved stability of the reaction mixture reduces the risk of batch failures due to runaway exotherms or decomposition, ensuring a more predictable and reliable production schedule that supports just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently scalable as it utilizes standard unit operations like reflux and azeotropic distillation which are well-understood and easily implemented from pilot plant to multi-ton production scales. The reduction in hazardous waste generation, specifically the avoidance of large volumes of acidic wastewater requiring neutralization before discharge, significantly lowers the environmental footprint of the manufacturing site and simplifies permitting processes. This alignment with green chemistry principles not only reduces regulatory burden but also enhances the corporate sustainability profile, which is increasingly becoming a key criterion for selection by major multinational customers in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Group 4 catalyzed esterification technology, derived directly from the experimental data and background analysis of the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this process for potential integration into their manufacturing portfolios. Understanding these nuances is essential for making informed decisions about technology transfer and process optimization.
Q: Why is the Group 4 catalyst preferred over traditional sulfuric acid for this esterification?
A: Traditional strong protic acids like sulfuric acid often cause significant discoloration and side reactions due to their harsh acidity, whereas Group 4 Lewis acid catalysts provide milder conditions that preserve product integrity and color.
Q: What specific Group 4 compounds are effective for this synthesis?
A: The patent highlights zirconium compounds like zirconium tetrachloride and zirconium tert-butoxide, as well as titanium and hafnium analogues, which demonstrate excellent catalytic activity and selectivity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes standard industrial equipment such as reactors equipped with Dean-Stark traps for water removal and allows for easy catalyst removal via aqueous washing, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropane Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies like the one described in CN1210248C to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Group 4 catalyzed process are fully realized in a practical, industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of cyclopropane carboxylate meets the exacting standards required for agrochemical and pharmaceutical applications. Our commitment to quality is matched only by our dedication to continuous improvement, constantly evaluating new catalytic systems to enhance efficiency and sustainability.
We invite you to engage with our technical procurement team to discuss how this innovative esterification technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this cleaner, more efficient process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the performance of our high-purity intermediates against your internal benchmarks. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing, delivering both technical excellence and commercial reliability.
