Advanced Catalytic Esterification for High-Purity Cyclopropane Intermediates and Commercial Scalability
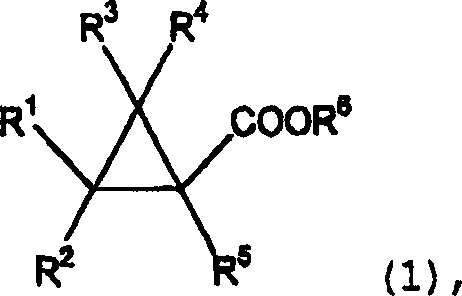
The landscape of fine chemical synthesis is constantly evolving, driven by the need for cleaner, more efficient pathways that align with modern industrial standards. A pivotal advancement in this domain is documented in patent CN1369476A, which discloses a robust method for preparing cyclopropane carboxylates of formula (1). This technology represents a significant departure from traditional esterification protocols by utilizing catalyst compounds containing elements from Group 4 of the Periodic Table, such as titanium, zirconium, and hafnium. Unlike conventional methods that rely on harsh protonic acids, this novel approach enables the dehydration reaction between cyclopropanecarboxylic acids and monohydric hydroxy compounds under remarkably mild yet effective conditions. For R&D directors and process chemists, this shift offers a compelling solution to long-standing challenges regarding impurity profiles and reaction selectivity. The ability to synthesize these critical intermediates with high fidelity is essential for the production of next-generation agrochemicals and pharmaceuticals, where structural integrity is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of carboxylic acid esters from carboxylic acids and alcohols has predominantly relied on the use of protic acid catalysts. Methods utilizing sulfuric acid or p-toluenesulfonic acid have been widely disclosed in prior art, such as Japanese Patent Laid-Open No. 9-188649 and No. 11-228491. However, these traditional methodologies suffer from inherent and severe drawbacks that limit their effectiveness in modern industrial production. The primary issue stems from the strong acidity of these inorganic or organic proton donors, which frequently induces unwanted side reactions. These side reactions often manifest as conspicuous coloration in the final product, indicating the formation of complex impurities and degradation byproducts. For a procurement manager focused on quality control, this discoloration necessitates additional, costly purification steps such as activated carbon treatment or extensive distillation, thereby inflating the overall cost of goods sold and extending production cycles.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes Lewis acidic compounds derived from Group 4 elements to catalyze the esterification process. This method fundamentally alters the reaction environment, moving away from proton donation to Lewis acid coordination. By employing catalysts such as zirconium tetrachloride, titanium tetraisopropoxide, or hafnium tetrachloride, the reaction proceeds with exceptional efficiency. Experimental data from the patent indicates that this method can achieve yields as high as 98% with selectivity reaching 99%, significantly outperforming the 61% to 81% yields observed with traditional sulfuric or p-toluenesulfonic acid catalysts. This dramatic improvement in performance metrics translates directly into operational excellence. For supply chain heads, this means a more reliable source of high-purity intermediates with consistent batch-to-batch quality, reducing the risk of downstream formulation failures and ensuring a steady flow of materials for final product assembly.
Mechanistic Insights into Group 4 Lewis Acid-Catalyzed Esterification
The mechanistic superiority of Group 4 catalysts lies in their specific mode of action as Lewis acids. Unlike Brønsted acids which donate protons and can potentially protonate sensitive functional groups within the cyclopropane ring or adjacent olefinic chains, Group 4 metals coordinate with the carbonyl oxygen of the carboxylic acid. This coordination activates the carbonyl carbon towards nucleophilic attack by the alcohol without generating a highly acidic medium that could trigger ring-opening or polymerization side reactions. This gentle activation is crucial for maintaining the stereochemical integrity of the molecule, particularly when dealing with chiral cyclopropane structures common in pyrethroid intermediates. The preservation of the E/Z ratio, as evidenced in the experimental examples where ratios remained stable or improved, underscores the mildness of this catalytic system. For R&D teams, understanding this mechanism provides confidence that complex, multifunctional substrates can be processed without compromising their delicate structural features.
Furthermore, the impurity control mechanism is intrinsically linked to the avoidance of strong acid-mediated degradation. In conventional acid-catalyzed reactions, the harsh conditions often lead to the formation of tars and colored oligomers, which are difficult to separate and can poison downstream catalysts in subsequent synthetic steps. The Group 4 catalyzed route minimizes these degradation pathways, resulting in a cleaner reaction mixture. The byproduct of the reaction is simply water, which can be continuously removed via azeotropic distillation using solvents like xylene or toluene, driving the equilibrium towards the ester product. This simplicity in byproduct management not only enhances the chemical yield but also simplifies the work-up procedure. The catalyst can typically be removed by a simple wash with water or acidic aqueous solution, eliminating the need for complex neutralization protocols required for strong mineral acids, thus streamlining the entire manufacturing workflow.
How to Synthesize Cyclopropane Carboxylate Efficiently
The synthesis of these valuable esters follows a straightforward yet highly optimized protocol designed for reproducibility and scalability. The process begins with the combination of the cyclopropanecarboxylic acid substrate and the chosen monohydric alcohol in an inert organic solvent. The choice of solvent is critical; aromatic hydrocarbons like xylene or toluene are preferred due to their ability to form azeotropes with water, facilitating its removal. Once the reactants are mixed, a catalytic amount of the Group 4 compound is introduced. The reaction mixture is then heated to reflux, typically around 145°C, under an inert atmosphere to prevent oxidation. The detailed standardized synthesis steps see the guide below.
- Combine cyclopropanecarboxylic acid and a monohydric hydroxy compound in an inert solvent such as xylene or toluene.
- Add a catalytic amount of a Group 4 element compound, such as zirconium tetrachloride or titanium tetraisopropoxide.
- Heat the mixture to reflux (approximately 145°C) while continuously removing water via a Dean-Stark trap to drive the dehydration reaction to completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this Group 4 catalytic technology offers transformative benefits that extend far beyond simple chemical yield improvements. The shift from corrosive protonic acids to stable metal-based catalysts fundamentally reshapes the cost structure and risk profile of the manufacturing process. By eliminating the use of concentrated sulfuric acid or p-toluenesulfonic acid, facilities can significantly reduce the corrosion burden on reactor vessels and piping systems. This reduction in equipment wear and tear translates into lower maintenance costs and extended asset life, providing a tangible financial advantage over the long term. Additionally, the cleaner reaction profile means less waste generation, specifically reducing the volume of acidic wastewater that requires neutralization and treatment before disposal, aligning with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the simplification of the downstream processing train. Traditional methods often require multiple purification stages to remove color bodies and acid residues, consuming significant amounts of energy and consumables. In contrast, the novel Group 4 catalyzed process produces a much cleaner crude product, often requiring only a simple wash and distillation. This drastic simplification of the purification workflow leads to substantial cost savings in terms of labor, energy consumption, and solvent usage. Furthermore, the higher selectivity ensures that less raw material is wasted on byproduct formation, maximizing the atom economy and reducing the effective cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the variability of raw material quality and the complexity of synthesis. The robustness of the Group 4 catalyst system mitigates these risks by providing a wider operating window and greater tolerance to variations in feedstock quality. The catalysts themselves, such as zirconium tetrachloride and titanium alkoxides, are commercially available commodities with stable supply lines, unlike some specialized organocatalysts that may face sourcing bottlenecks. This accessibility ensures that production schedules can be maintained without interruption. Moreover, the consistency of the reaction outcome reduces the likelihood of batch failures, ensuring a steady and predictable output of high-purity intermediates for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often introduces new challenges, particularly regarding heat management and safety. The Group 4 catalyzed esterification is inherently safer as it avoids the exothermic risks associated with mixing strong acids and alcohols. The reaction conditions are moderate and easily controlled using standard industrial equipment. From an environmental perspective, the reduction in acidic waste and the elimination of heavy decolorization steps contribute to a smaller environmental footprint. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the brand value of the final product in markets that prioritize sustainability and responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced esterification technology. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for making informed decisions about process adoption and supplier selection.
Q: Why are Group 4 catalysts preferred over traditional proton acids for cyclopropane esterification?
A: Traditional proton acids like sulfuric acid often cause undesirable side reactions and significant discoloration due to their strong acidity. Group 4 catalysts act as milder Lewis acids, preserving the integrity of sensitive functional groups and resulting in significantly higher selectivity and product clarity.
Q: What represents the primary commercial advantage of this catalytic system for supply chains?
A: The primary advantage lies in the simplification of downstream processing. By eliminating the need for extensive neutralization and decolorization steps associated with strong mineral acids, manufacturers can achieve substantial cost savings and reduce lead time for high-purity agrochemical intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent data demonstrates high yields (up to 98%) and selectivity under standard reflux conditions using commercially available solvents and catalysts. The absence of corrosive strong acids also reduces equipment maintenance requirements, facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropane Carboxylate Supplier
The technical potential of the Group 4 catalyzed synthesis route for cyclopropane carboxylates is immense, offering a pathway to superior product quality and operational efficiency. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such sophisticated chemistry to the market. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of cyclopropane intermediate meets the exacting standards demanded by the global agrochemical and pharmaceutical industries. We understand that transitioning to a new synthetic route requires a partner who can navigate the complexities of process optimization and regulatory compliance with expertise.
We invite you to engage with our technical procurement team to discuss how this innovative technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this catalytic method can optimize your manufacturing economics. We are prepared to provide specific COA data and route feasibility assessments tailored to your specific project needs. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
