Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups due to their profound impact on metabolic stability and lipophilicity. A significant breakthrough in this domain is documented in Chinese patent CN111423381A, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. This technology represents a paradigm shift from traditional high-energy processes to a mild, transition metal-catalyzed carbonylation cascade that operates effectively at just 30°C. For R&D directors and procurement specialists evaluating new supply chains, this patent offers a compelling route that combines high reaction efficiency with exceptional substrate compatibility. The ability to synthesize diversely substituted imidazole scaffolds using cheap and readily available starting materials addresses critical pain points in the manufacturing of complex API intermediates. By leveraging this specific catalytic system, manufacturers can achieve substantial improvements in process safety and operational simplicity, paving the way for more reliable production of high-value chemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant technical challenges and economic inefficiencies. Conventional literature methods often rely on the direct reaction of trifluoromethyl synthons, such as trifluorodiazoethane, with suitable substrates, which can pose severe safety hazards due to the explosive nature of diazo compounds. Furthermore, many existing protocols require harsh reaction conditions, including elevated temperatures and the use of expensive or toxic reagents that complicate downstream processing and waste management. The limited availability of specialized trifluoroethylimide acid halides in the past has also restricted the widespread application of certain synthetic routes, forcing chemists to rely on multi-step sequences that erode overall yield and increase production costs. These traditional approaches often suffer from poor functional group tolerance, limiting the structural diversity accessible to medicinal chemists during the lead optimization phase. Consequently, the industry has faced persistent bottlenecks in scaling these reactions for commercial purposes, necessitating a search for more benign and versatile synthetic alternatives.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a transition metal palladium-catalyzed carbonylation series reaction that fundamentally simplifies the synthetic landscape. By employing trifluoroethylimide chloride, propargylamine, and diaryliodonium salts as starting materials, this method bypasses the need for hazardous diazo reagents while maintaining high reaction efficiency. The process is remarkably mild, proceeding at a constant temperature of 30°C over a period of 16 to 24 hours, which drastically reduces energy consumption compared to thermal-intensive alternatives. The use of a formic acid and acetic anhydride mixture serves as a safe carbon monoxide substitute, generating the necessary carbonyl species in situ without the risks associated with handling high-pressure CO gas. This strategy not only enhances operational safety but also improves the atom economy of the transformation. The broad substrate compatibility allows for the introduction of various substituents on the aryl rings, enabling the rapid generation of diverse chemical libraries for biological screening without the need for extensive process re-optimization.
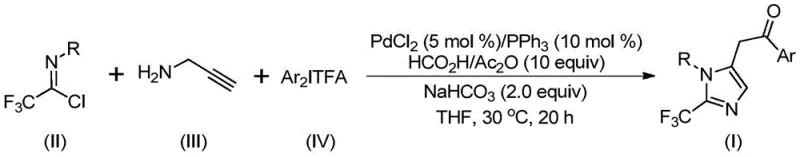
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
A deep understanding of the catalytic cycle is essential for R&D teams aiming to optimize this process for commercial scale-up. The reaction mechanism likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine intermediate. This species then undergoes isomerization, setting the stage for the palladium-catalyzed aminopalladation of the alkyne moiety to generate a key alkenyl palladium intermediate. Subsequent isomerization converts this into an alkyl palladium species, which is poised for the crucial carbonylation step. Under the influence of carbon monoxide released from the formic acid and acetic anhydride mixture, the alkyl palladium intermediate undergoes carbonylation to form an acyl palladium complex. This acyl species is then subjected to oxidative addition with the diaryliodonium salt, forming a high-valent tetravalent palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole product and regenerates the active palladium catalyst for the next turnover. This intricate dance of organometallic steps ensures high selectivity and minimizes the formation of unwanted byproducts.
Controlling the impurity profile is paramount for pharmaceutical intermediates, and this mechanism offers inherent advantages in purity management. The mild reaction temperature of 30°C significantly suppresses thermal decomposition pathways that often lead to complex impurity profiles in high-temperature reactions. Furthermore, the specific choice of ligands and additives, such as triphenylphosphine and sodium bicarbonate, helps stabilize the palladium center and prevents the aggregation of metal particles that could catalyze non-selective side reactions. The use of diaryliodonium salts as electrophiles is particularly advantageous as they are generally more reactive and selective than traditional aryl halides, reducing the likelihood of unreacted starting materials carrying through to the final product. Post-processing involves straightforward filtration and silica gel mixing, followed by column chromatography, which is a standard and scalable purification technique in the fine chemical industry. The robustness of this catalytic system means that even with variations in raw material quality, the process maintains a consistent impurity spectrum, facilitating easier validation and regulatory compliance for GMP manufacturing environments.
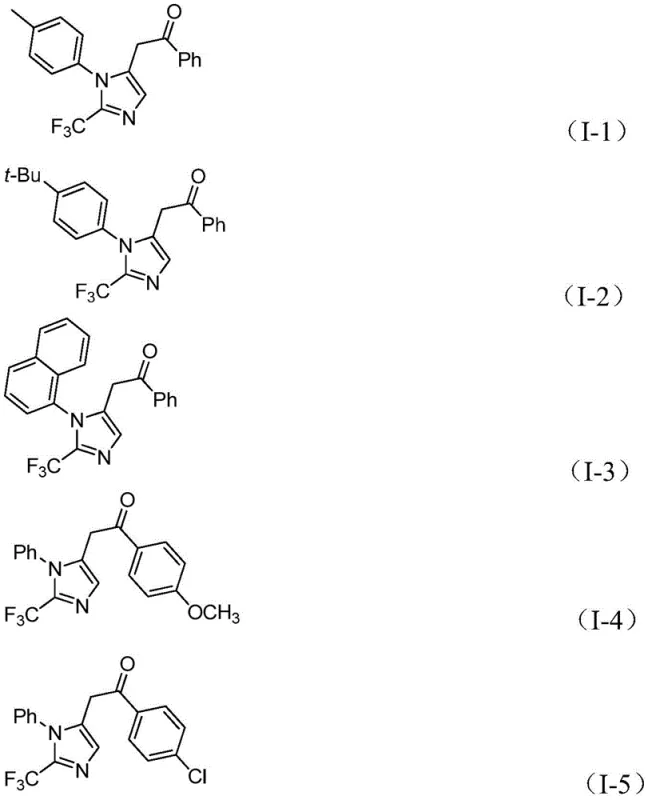
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and solvent choices as outlined in the patent data. The process begins with the preparation of a catalytic solution containing palladium chloride, triphenylphosphine, and sodium bicarbonate in an aprotic organic solvent, with tetrahydrofuran (THF) being the preferred medium due to its superior solubilizing power and reaction promotion capabilities. The molar ratio of the catalyst system is critical, typically utilizing 0.05 equivalents of PdCl2 and 0.1 equivalents of PPh3 relative to the substrate to ensure complete conversion without excessive metal loading. Key reactants, including trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt, are added in optimized molar ratios, often with a slight excess of the imidoyl chloride to drive the reaction to completion. The reaction mixture is then stirred at 30°C for a duration of 16 to 24 hours, allowing sufficient time for the cascade transformation to reach full conversion. Detailed standardized synthesis steps follow below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by combining palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add the key substrates: trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the catalytic system under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, followed by filtration and purification via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this patented technology are substantial and multifaceted. The primary driver for cost reduction lies in the utilization of cheap and readily available starting materials, such as aromatic amines and commercially sourced palladium catalysts, which eliminates the dependency on exotic or custom-synthesized reagents. The mild reaction conditions translate directly into lower energy costs, as maintaining a temperature of 30°C requires significantly less heating or cooling infrastructure compared to cryogenic or high-temperature processes. Additionally, the simplified post-processing workflow, which avoids complex extraction or distillation steps in favor of standard chromatography, reduces labor hours and solvent consumption. These factors combine to create a manufacturing process that is not only economically viable but also resilient to fluctuations in raw material pricing. The ability to source precursors from multiple vendors further mitigates supply risk, ensuring continuity of supply for critical pharmaceutical projects.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts beyond standard palladium sources and the use of inexpensive carbon monoxide substitutes drastically lower the bill of materials. By avoiding the need for specialized high-pressure equipment required for gaseous CO, capital expenditure for reactor setup is significantly minimized. The high reaction efficiency and yield reported in the patent examples mean that less raw material is wasted, directly improving the cost-per-kilogram metric for the final intermediate. Furthermore, the compatibility with common solvents like THF allows for easier solvent recovery and recycling, contributing to long-term operational savings. These cumulative effects result in a leaner manufacturing cost structure that enhances competitiveness in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as propargylamine and diaryliodonium salts ensures that the supply chain is not vulnerable to single-source bottlenecks. Since the reaction tolerates a wide range of functional groups, manufacturers can flexibly switch between different substituted aryl amines based on availability without redesigning the entire process. The scalability of the method from gram scale to potential tonnage production provides confidence for long-term supply agreements with pharmaceutical clients. Reduced lead times are achieved through the simplicity of the operation, which requires fewer unit operations and less time for reactor turnaround. This agility allows suppliers to respond quickly to changes in demand, securing their position as a reliable partner in the pharmaceutical value chain.
- Scalability and Environmental Compliance: The process demonstrates excellent potential for commercial scale-up, with the patent explicitly noting its applicability for industrial large-scale production. The use of a closed system for in situ CO generation minimizes the release of toxic gases, aligning with stringent environmental health and safety regulations. Waste generation is minimized due to the high atom economy of the carbonylation cascade and the ability to recycle solvents effectively. The avoidance of hazardous diazo compounds removes a major safety liability, simplifying the permitting process for new manufacturing lines. Overall, the green chemistry attributes of this method, including mild conditions and reduced waste, support sustainability goals while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for potential partners. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing portfolios. The responses highlight the balance between technical performance and operational practicality that defines this innovation.
Q: What are the primary advantages of this Pd-catalyzed method over traditional synthesis?
A: This method operates at a mild temperature of 30°C using cheap and readily available starting materials, avoiding the harsh conditions often required for introducing trifluoromethyl groups into heterocycles.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and uses common solvents like THF, indicating strong potential for commercial scale-up and industrial application.
Q: What is the source of the carbonyl group in this reaction mechanism?
A: The carbonyl group is generated in situ from a mixture of formic acid and acetic anhydride, which acts as a safe and effective carbon monoxide substitute under the reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN111423381A for the development of next-generation therapeutics. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates. We understand that consistency and quality are non-negotiable in the life sciences sector, and our team is dedicated to delivering materials that exceed industry standards. By partnering with us, you gain access to a wealth of technical expertise that can optimize this specific palladium-catalyzed route for your unique production needs.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific requirements. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this efficient manufacturing process. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making. Let us help you secure a stable, cost-effective supply of high-purity 2-trifluoromethyl imidazoles for your critical drug development programs. Contact us today to initiate a dialogue about your supply chain needs.
