Advanced Ruthenium-Catalyzed Asymmetric Hydrogenation for High-Purity Pharmaceutical Intermediates
The landscape of chiral synthesis in the fine chemical and pharmaceutical sectors is continually evolving, driven by the demand for higher enantiomeric purity and more efficient manufacturing processes. Patent CN101400632A represents a significant technological leap in this domain, disclosing a sophisticated method for the stereoselective hydrogenation of racemic aldehydes and ketones. This innovation centers on the utilization of specialized ruthenium complexes containing bidentate ligands with coordinating phosphorus and nitrogen atoms, combined with monophosphine ligands. Unlike traditional hydrogenation methods that often struggle with substrates possessing stereogenic carbon atoms at the alpha position relative to the carbonyl group, this patented approach facilitates a simultaneous dynamic-kinetic resolution. This means that even when starting with a racemic mixture, the process can selectively convert the material into a predominantly single enantiomeric primary alcohol or diastereomeric secondary alcohol with exceptional optical yields. For R&D directors and process chemists, this technology offers a robust pathway to access high-purity chiral building blocks essential for active pharmaceutical ingredients (APIs) and agrochemical intermediates without the substantial yield losses associated with classical resolution techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of prochiral ketones has been a cornerstone of chiral synthesis, yet it faces distinct challenges when the substrate contains a pre-existing stereocenter at the alpha position. Conventional catalytic systems, such as those described in prior art like WO 2004/050585, while effective for simple prochiral ketones, often fail to control the stereochemistry at the alpha-carbon during the reduction of the carbonyl group. This limitation frequently results in the formation of mixtures of diastereomers, necessitating cumbersome and costly downstream separation processes like chromatography or recrystallization to isolate the desired isomer. Furthermore, standard catalysts may lack the necessary activity to drive the reaction to completion under mild conditions, often requiring elevated temperatures that can lead to unwanted racemization of the sensitive alpha-stereocenter. These inefficiencies translate directly into increased production costs, longer lead times, and a larger environmental footprint due to solvent waste and lower overall atom economy, posing significant hurdles for procurement managers aiming to optimize supply chain efficiency.
The Novel Approach
The methodology outlined in CN101400632A overcomes these historical bottlenecks through the deployment of a uniquely designed ruthenium catalyst system. By integrating a bidentate ligand of Formula (II) featuring a specific P-Z-N backbone with a monophosphine ligand, the catalyst achieves a level of stereocontrol previously difficult to attain. 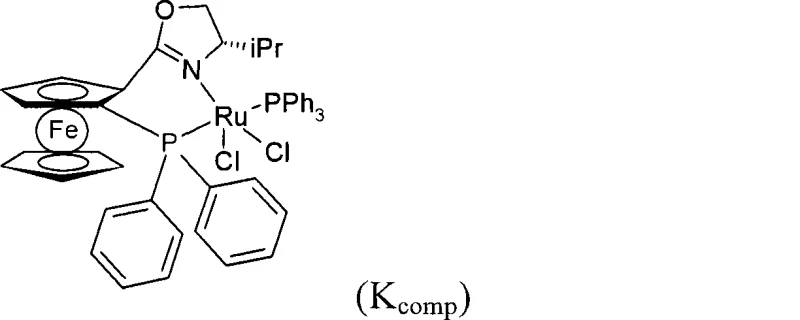 As illustrated by the comparison with known complexes like Kcomp, the novel ligand architecture creates a chiral environment that not only discriminates between the enantiotopic faces of the ketone but also interacts favorably with the alpha-substituent to enforce high diastereoselectivity. Crucially, the presence of a base in the reaction medium facilitates the continuous racemization of the unreacted enantiomer of the starting material. This dynamic kinetic resolution mechanism ensures that theoretically 100% of the racemic starting material can be converted into the desired chiral product, drastically improving yield compared to the maximum 50% theoretical yield of a standard kinetic resolution. This breakthrough provides a reliable pharmaceutical intermediates supplier with the capability to deliver complex chiral alcohols with superior cost-efficiency and purity profiles.
As illustrated by the comparison with known complexes like Kcomp, the novel ligand architecture creates a chiral environment that not only discriminates between the enantiotopic faces of the ketone but also interacts favorably with the alpha-substituent to enforce high diastereoselectivity. Crucially, the presence of a base in the reaction medium facilitates the continuous racemization of the unreacted enantiomer of the starting material. This dynamic kinetic resolution mechanism ensures that theoretically 100% of the racemic starting material can be converted into the desired chiral product, drastically improving yield compared to the maximum 50% theoretical yield of a standard kinetic resolution. This breakthrough provides a reliable pharmaceutical intermediates supplier with the capability to deliver complex chiral alcohols with superior cost-efficiency and purity profiles.
Mechanistic Insights into Ru-P-N Catalyzed Dynamic Kinetic Resolution
The core of this technology lies in the intricate interplay between the ruthenium metal center and the multifunctional ligand system. The active catalyst species, generally represented by the formula [XYRu(PR1R2R3)(P-Z-N)], operates through a mechanism that harmonizes hydrogen activation with substrate coordination. The bidentate P-Z-N ligand, particularly those derived from ferrocenyl backbones with oxazoline moieties, imposes a rigid chiral pocket around the ruthenium atom. 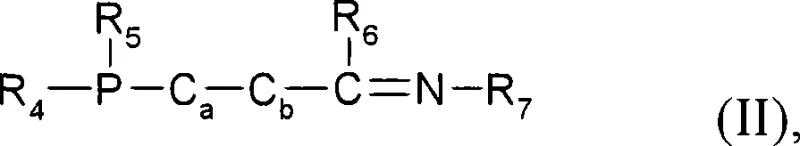 This structural rigidity is paramount for transmitting chiral information to the substrate during the hydride transfer step. When the racemic ketone substrate, characterized by the structural unit -(O)C-C*-CH-, coordinates to the metal, the steric bulk of the ligand substituents (such as ortho-substituted aryl groups on the phosphorus) directs the approach of the hydride to a specific face of the carbonyl group. Simultaneously, the basic conditions promote the rapid equilibration of the substrate enantiomers via enolization. This ensures that the 'slow-reacting' enantiomer is constantly converted into the 'fast-reacting' one, funneling the entire substrate pool into the desired product channel. For technical teams, understanding this mechanism is vital for troubleshooting; it highlights why maintaining the correct base-to-substrate ratio and ensuring the integrity of the ligand's chiral centers are critical for maintaining high enantiomeric excess (ee) and diastereomeric ratio (dr).
This structural rigidity is paramount for transmitting chiral information to the substrate during the hydride transfer step. When the racemic ketone substrate, characterized by the structural unit -(O)C-C*-CH-, coordinates to the metal, the steric bulk of the ligand substituents (such as ortho-substituted aryl groups on the phosphorus) directs the approach of the hydride to a specific face of the carbonyl group. Simultaneously, the basic conditions promote the rapid equilibration of the substrate enantiomers via enolization. This ensures that the 'slow-reacting' enantiomer is constantly converted into the 'fast-reacting' one, funneling the entire substrate pool into the desired product channel. For technical teams, understanding this mechanism is vital for troubleshooting; it highlights why maintaining the correct base-to-substrate ratio and ensuring the integrity of the ligand's chiral centers are critical for maintaining high enantiomeric excess (ee) and diastereomeric ratio (dr).
Furthermore, the impurity profile of the resulting alcohol is significantly managed by this high selectivity. In conventional non-dynamic processes, the presence of the mismatched enantiomer often leads to the formation of the undesired diastereomer, which can be structurally very similar to the product and difficult to remove. The novel catalyst system minimizes the formation of these diastereomeric impurities by enforcing a strict stereochemical pathway. The patent data indicates that for substrates like 2-trifluoromethylacetophenone, the new catalysts can achieve up to 96% ee, significantly outperforming comparative catalysts which might only reach 90% ee under similar conditions. This reduction in impurity load simplifies the purification train, potentially allowing for direct crystallization rather than extensive chromatographic purification. For quality control departments, this translates to more consistent batch-to-batch quality and easier compliance with stringent regulatory specifications for chiral drugs, reinforcing the value of this technology for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Chiral Alcohols Efficiently
Implementing this hydrogenation technology requires careful attention to the preparation of the catalyst and the control of reaction parameters to maximize the benefits of the dynamic kinetic resolution. The patent describes a versatile protocol where the ruthenium complex can be generated in situ or pre-formed, offering flexibility for different manufacturing scales. The process typically involves dissolving the catalyst precursor and the specific chiral ligand in a solvent like toluene or isopropanol, followed by the addition of the substrate and a stoichiometric amount of base. The reaction is then conducted under hydrogen pressure, where the interplay of temperature and pressure dictates the reaction rate and selectivity. Detailed standard operating procedures for ligand synthesis, complex formation, and the hydrogenation run are critical for reproducibility.
- Prepare the catalyst solution by dissolving the Ruthenium complex (e.g., K1, K2, or K3) in an inert solvent such as toluene or isopropanol under an argon atmosphere.
- Dissolve the racemic ketone substrate in the same solvent system in a separate vessel and transfer both solutions into a pre-flushed autoclave.
- Add the base solution (e.g., aqueous NaOH or potassium tert-butoxide in isopropanol), purge the system with hydrogen, pressurize to 40-80 bar, and stir at 20-35°C until hydrogen uptake ceases.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101400632A offers compelling strategic advantages beyond mere technical performance. The primary value driver is the potential for substantial cost reduction in pharmaceutical intermediates manufacturing. By enabling dynamic kinetic resolution, the process eliminates the inherent 50% yield loss associated with traditional resolution of racemates. This doubling of theoretical yield from the same amount of starting raw material directly lowers the cost of goods sold (COGS). Additionally, the high selectivity reduces the burden on downstream purification, meaning less solvent consumption, reduced waste disposal costs, and shorter cycle times. The ability to use readily available racemic starting materials, rather than expensive enantiopure precursors, further enhances the economic viability of the route, making it an attractive option for cost-sensitive large-volume production.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the high atom economy of the dynamic kinetic process lead to significant operational savings. By converting the entire racemic feedstock into the desired chiral product, manufacturers avoid the costs associated with discarding or recycling the unwanted enantiomer. Furthermore, the mild reaction conditions (often near room temperature) reduce energy consumption compared to high-temperature processes, contributing to a leaner manufacturing budget and improved margin profiles for the final API.
- Enhanced Supply Chain Reliability: The robustness of the ruthenium catalyst system contributes to a more stable supply chain. The catalysts described exhibit high turnover numbers, meaning less catalyst is required per kilogram of product, reducing dependency on precious metal supply fluctuations. Moreover, the tolerance of the system to various functional groups allows for a broader range of commercially available starting materials to be used, mitigating the risk of raw material shortages. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing companies to respond more agilely to market demands.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard hydrogenation equipment found in most fine chemical plants. The use of common solvents like toluene and isopropanol simplifies solvent recovery and recycling protocols. From an environmental perspective, the high efficiency and reduced waste generation align with green chemistry principles, facilitating easier regulatory compliance and supporting corporate sustainability goals. The ability to scale from gram to multi-ton quantities without losing selectivity ensures a seamless transition from pilot plant to commercial production.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding its application and performance. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for process development discussions. Understanding the scope and limitations of the catalyst system is essential for successful technology transfer and risk mitigation during the scale-up phase.
Q: What is the primary advantage of the novel Ru-P-N catalysts over conventional methods?
A: The novel catalysts enable dynamic kinetic resolution, allowing racemic substrates with stereogenic alpha-carbons to be converted into a single predominant enantiomer with high optical yield, eliminating the need for downstream resolution steps.
Q: What are the typical reaction conditions for this hydrogenation process?
A: The process operates under mild conditions, typically at temperatures between 20°C and 35°C and hydrogen pressures ranging from 40 to 80 bar, using solvents like toluene or isopropanol with a suitable base.
Q: Can this method handle substrates with sensitive functional groups?
A: Yes, the catalyst system demonstrates high chemoselectivity, effectively hydrogenating the ketone moiety while leaving other unsaturated groups intact, which is crucial for complex pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced asymmetric hydrogenation technologies like those disclosed in CN101400632A for the production of high-value chiral intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a manufacturing setting. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of chiral alcohol meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and technical excellence makes us a trusted partner for companies seeking to optimize their chiral synthesis routes.
We invite you to engage with our technical procurement team to discuss how this catalytic hydrogenation technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this dynamic kinetic resolution process. We encourage you to contact us to obtain specific COA data for relevant intermediates and to receive detailed route feasibility assessments. Let us help you accelerate your development timeline and secure a competitive advantage in the market with our reliable supply of high-purity chiral building blocks.
