Advanced Lewis Acid Catalysis for Scalable Diamine Derivative Production in Pharmaceutical Intermediates
Advanced Lewis Acid Catalysis for Scalable Diamine Derivative Production in Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing vicinal diamine scaffolds, which are ubiquitous motifs in bioactive molecules and chiral ligands. Patent CN108530381B introduces a groundbreaking approach for preparing diamine derivatives directly from amide esters, addressing long-standing challenges in ester alkoxy bond cleavage functionalization. Traditionally, activating stable ester bonds required aggressive conditions or precious transition metals, creating bottlenecks in cost and scalability. This invention leverages a Lewis acid-catalyzed substitution strategy using saccharin as a nitrogen nucleophile, enabling the transformation under remarkably mild and solvent-free conditions. By retaining the carboxylic acid amide from the starting material while substituting the ester group, the process delivers high-value ortho-diamine derivatives with exceptional efficiency. For R&D directors and procurement specialists, this technology represents a paradigm shift towards more sustainable and economically viable manufacturing of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of ester alkoxy bonds has been a formidable challenge in organic synthesis due to the inherent stability of the ester linkage. Existing methodologies often rely heavily on olefin-activated systems or necessitate the use of expensive transition metal catalysts such as palladium, nickel, or ruthenium to facilitate bond cleavage. For instance, prior art describes ruthenium-catalyzed reactions that require specific activation groups, limiting substrate scope and increasing raw material costs. Furthermore, non-activated alkyl esters typically demand severe reaction conditions, including high temperatures and pressures, which can lead to decomposition of sensitive functional groups and poor selectivity. Direct amination routes using ammonia water and copper oxide catalysts often result in complex mixtures that are difficult to separate, drastically reducing overall yield and purity. These limitations impose significant burdens on supply chain managers who must source costly metals and manage hazardous waste streams, while R&D teams struggle with reproducibility and scale-up issues associated with heterogeneous catalytic systems.
The Novel Approach
The methodology disclosed in CN108530381B circumvents these obstacles by employing a trifluoromethanesulfonate Lewis acid catalyst to activate the ester group for nucleophilic attack by saccharin. This innovative route eliminates the need for precious metals, utilizing abundant and inexpensive lanthanide or group 4 metal triflates instead. A key advantage is the ability to operate under solvent-free conditions, which not only simplifies the reaction setup but also dramatically reduces the environmental footprint and downstream solvent recovery costs. The reaction proceeds with high stereoselectivity, consistently yielding trans-diamine derivatives regardless of the starting material's configuration, a feature that is highly desirable for synthesizing chiral drug intermediates. By using commercially available amino alcohol precursors to generate the amide ester starting materials, the entire synthetic sequence becomes modular and cost-effective. This approach transforms a traditionally difficult transformation into a reliable, scalable process suitable for industrial manufacturing of high-purity pharmaceutical intermediates.
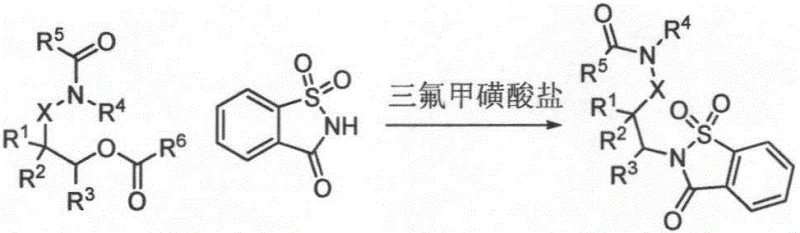
Mechanistic Insights into Lewis Acid-Catalyzed Ester Substitution
The core of this technological advancement lies in the activation mechanism facilitated by the metal triflate catalyst, such as hafnium(IV) triflate. In this system, the Lewis acidic metal center coordinates with the carbonyl oxygen of the ester group in the amide ester substrate, significantly increasing the electrophilicity of the carbonyl carbon. This activation lowers the energy barrier for the nucleophilic attack by the nitrogen atom of the saccharin molecule. Unlike traditional acyl substitution which might cleave the acyl-oxygen bond, this specific pathway targets the alkoxy-oxygen bond cleavage, effectively swapping the alkoxy group for the sulfonimide moiety. The choice of catalyst is critical; experimental data indicates that hafnium triflate outperforms other metal triflates like iron, aluminum, or scandium in terms of yield and reaction rate. The mechanism likely involves a tight ion pair or a coordinated transition state that stabilizes the leaving alkoxide group, preventing side reactions such as hydrolysis or elimination. This precise control over the reaction pathway ensures that the amide functionality present in the starting material remains intact, preserving the structural integrity required for downstream applications in drug synthesis.
Furthermore, the reaction exhibits remarkable stereochemical fidelity, which is a crucial parameter for R&D directors focusing on chiral pool synthesis. The process demonstrates a strong thermodynamic drive towards the formation of trans-diamine derivatives. Even when starting with cis-configured amino alcohol precursors or mixtures of stereoisomers, the reaction conditions promote equilibration or selective formation of the trans-product. This phenomenon suggests that the transition state favors a conformation where the bulky saccharin group and the existing amide substituent are anti-periplanar to minimize steric hindrance. Such high stereoselectivity eliminates the need for difficult chromatographic separations of diastereomers, thereby streamlining the purification process. For manufacturers of electronic chemicals or advanced pharmaceutical ingredients, this level of control over impurity profiles is invaluable, as it ensures consistent batch-to-bquality and reduces the risk of regulatory rejection due to unknown stereoisomeric impurities. The ability to predictably generate specific stereoisomers from diverse substrates underscores the robustness of this catalytic system.
How to Synthesize Diamine Derivatives Efficiently
To implement this synthesis effectively, operators should begin by preparing the amide ester substrate through conventional acylation of readily available amino alcohols, ensuring high purity before the substitution step. The detailed standardized synthesis steps for the substitution reaction are outlined below, focusing on the optimal conditions identified in the patent examples. It is essential to maintain strict anhydrous conditions to prevent catalyst deactivation and ensure maximum conversion rates. The following guide summarizes the critical parameters for achieving the reported high yields and stereoselectivity.
- Mix amide ester substrate and saccharin in a reaction vessel without solvent, maintaining a molar ratio of approximately 1.2: 1.
- Add hafnium trifluoromethanesulfonate catalyst at a loading of 1 mol% relative to the saccharin nucleophile.
- Heat the mixture to 150°C under stirring for 24 hours to complete the substitution, followed by column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond simple yield improvements. The shift away from precious metal catalysts like palladium and ruthenium to base metal triflates represents a significant mitigation of supply risk and price volatility. Since the raw materials, specifically amino alcohols and saccharin, are commodity chemicals produced on a massive global scale, sourcing is straightforward and不受 geopolitical constraints that often affect rare earth metals or specialized ligands. The solvent-free nature of the reaction further enhances supply chain resilience by removing the dependency on large volumes of organic solvents, which are subject to fluctuating prices and stringent transportation regulations. This simplification of the bill of materials allows for more accurate forecasting and inventory management, reducing the capital tied up in raw material stockpiles. Additionally, the high stereoselectivity reduces the burden on quality control laboratories, as fewer resources are needed to monitor and separate unwanted isomers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts directly translates to substantial savings in raw material costs, as hafnium triflate is used in very low catalytic loadings compared to stoichiometric reagents. Moreover, the solvent-free protocol removes the significant operational expenses associated with solvent purchase, storage, recycling, and disposal, which often constitute a major portion of manufacturing overhead. The simplified workup procedure, which primarily involves column chromatography or crystallization without extensive aqueous washes to remove metal residues, reduces labor hours and utility consumption. By avoiding the need for specialized equipment to handle high-pressure hydrogenation or inert atmospheres required by other methods, capital expenditure for new production lines is also minimized. These cumulative efficiencies result in a lower cost of goods sold (COGS), providing a competitive pricing advantage in the market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals like saccharin and amino alcohols ensures a stable and continuous supply chain, unaffected by the bottlenecks common to specialized catalytic reagents. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by sensitivity to minor variations in raw material quality or environmental factors. The high conversion rates and selectivity reduce the generation of off-spec material, maximizing the throughput of the manufacturing facility and ensuring timely delivery to customers. This reliability is critical for maintaining just-in-time inventory systems and meeting the rigorous delivery deadlines demanded by downstream pharmaceutical clients. Furthermore, the simplicity of the process facilitates technology transfer between different manufacturing sites, enhancing overall supply network flexibility.
- Scalability and Environmental Compliance: The absence of volatile organic solvents significantly lowers the fire hazard and toxicity risks associated with large-scale chemical production, simplifying compliance with increasingly strict environmental, health, and safety (EHS) regulations. The reduction in waste generation, particularly heavy metal waste from catalysts and solvent waste from reaction media, aligns with green chemistry principles and corporate sustainability goals. Scaling this process from laboratory to commercial production is straightforward because the reaction does not rely on complex mass transfer limitations often seen in heterogeneous catalysis or gas-liquid reactions. The thermal stability of the reagents allows for safe operation at the optimized temperature of 150°C without requiring exotic pressure vessels. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand without compromising product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diamine synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this diamine synthesis method over traditional transition metal catalysis?
A: Unlike conventional methods requiring expensive palladium or ruthenium catalysts and harsh conditions, this process utilizes inexpensive Lewis acid catalysts like hafnium triflate under solvent-free conditions, significantly lowering production costs and simplifying waste treatment.
Q: How does this method handle stereochemical control in diamine production?
A: The reaction exhibits exceptional stereoselectivity, converting both cis and trans amino alcohol precursors into completely trans-diamine derivatives, which is critical for maintaining the biological activity of downstream pharmaceutical intermediates.
Q: Is the saccharin protecting group difficult to remove after synthesis?
A: No, the saccharin group introduced during the substitution can be readily converted to a free amino group using standard deprotection reagents such as NaI/TMSCl or HBr/AcOH, ensuring seamless integration into existing synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diamine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Lewis acid-catalyzed synthesis for producing high-value diamine derivatives used in modern drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle solvent-free reactions and sensitive Lewis acid chemistries with precision, adhering to stringent purity specifications required by global regulatory bodies. With our rigorous QC labs and dedicated process development teams, we guarantee the consistent quality and supply continuity necessary for your critical pharmaceutical programs.
We invite you to collaborate with us to leverage this efficient technology for your specific intermediate needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact us to request specific COA data for similar diamine scaffolds and comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-purity pharmaceutical intermediates while driving down your overall manufacturing costs through innovation.
