Advanced Cu-SBA-15 Catalyzed Oxidation for High-Purity Vitamin E Precursors
The global demand for high-purity Vitamin E intermediates continues to surge, driven by expanding applications in pharmaceuticals, nutraceuticals, and animal husbandry sectors. Patent CN102219665B introduces a transformative approach to synthesizing 2,3,5-trimethylbenzoquinone (TMBQ) and 2,3,5-trimethylhydroquinone (TMHQ), which are critical precursors in the value chain of tocopherol production. This intellectual property outlines a novel one-step oxidation methodology that leverages a Cu-SBA-15 mesoporous molecular sieve as a heterogeneous catalyst, fundamentally shifting the paradigm from multi-step, waste-intensive processes to a streamlined, green chemical synthesis. For R&D directors and technical decision-makers, this patent represents a significant opportunity to enhance process efficiency while adhering to increasingly stringent environmental regulations. The core innovation lies in the specific architecture of the catalyst, which facilitates superior mass transfer and product diffusion, thereby addressing common bottlenecks in traditional oxidation reactions. By adopting this technology, manufacturers can achieve a more sustainable production model that aligns with modern green chemistry principles without compromising on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
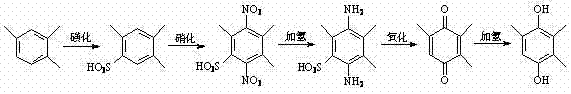
Historically, the industrial production of 2,3,5-trimethylhydroquinone has relied heavily on the trimethylbenzene method, a convoluted sequence involving sulfonation, nitration, hydrogenation, oxidation, and a final reduction step. This legacy pathway, while established, suffers from severe inherent inefficiencies, including an excessively long synthetic route that accumulates impurities at every stage and results in substantially low overall yields. The process generates massive quantities of hazardous three-waste streams, particularly acidic wastewater containing sulfur and nitrogen compounds, which imposes a heavy burden on environmental treatment facilities and escalates operational costs significantly. Furthermore, the requirement for multiple unit operations increases capital expenditure on equipment and extends production lead times, creating vulnerabilities in the supply chain for high-purity pharmaceutical intermediates. As regulatory pressures mount globally, the sustainability profile of this traditional route has become untenable for forward-thinking chemical enterprises seeking long-term viability.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach detailed in the patent utilizes a direct one-step oxidation of 2,3,6-trimethylphenol to generate both TMBQ and TMHQ simultaneously. This streamlined strategy eliminates the need for preliminary sulfonation and nitration steps, drastically reducing the number of unit operations and the associated energy consumption. The use of hydrogen peroxide as the terminal oxidant ensures that the only stoichiometric byproduct is water, effectively neutralizing the environmental impact associated with toxic effluent discharge. Moreover, the reaction proceeds under relatively mild conditions, typically between 50°C and 80°C, which lowers the thermal stress on equipment and enhances operational safety. This methodological shift not only simplifies the process flow but also opens avenues for significant cost reduction in vitamin E precursor manufacturing by minimizing raw material usage and waste disposal fees.
Mechanistic Insights into Cu-SBA-15 Catalyzed Oxidation
The efficacy of this synthesis hinges on the unique properties of the Cu-SBA-15 mesoporous molecular sieve, which serves as a robust heterogeneous catalyst with a highly ordered pore structure. The mesoporous channels of the SBA-15 framework provide an expansive surface area that promotes the uniform dispersion of active copper sites, facilitating efficient contact between the oxidant and the organic substrate. This structural advantage is critical for overcoming diffusion limitations often encountered in bulky molecule transformations, ensuring that the reaction kinetics are optimized for maximum conversion. Unlike homogeneous catalysts that require complex and costly separation procedures, the solid nature of the Cu-SBA-15 allows for straightforward filtration and recovery, enabling potential catalyst recycling and further driving down production costs. The copper species embedded within the silica matrix act as the active centers for activating hydrogen peroxide, generating reactive oxygen species that selectively oxidize the phenolic ring to the desired quinone and hydroquinone derivatives.
From an impurity control perspective, the specificity of the Cu-SBA-15 catalyst plays a pivotal role in defining the final product quality and purity profile. The shape-selective nature of the mesopores helps to suppress the formation of unwanted side products and over-oxidized species, which are common issues in non-selective oxidation processes. By maintaining a neutral reaction environment in acetonitrile solvent, the system avoids the acid-catalyzed degradation pathways that often plague traditional methods, resulting in a cleaner crude product that requires less intensive purification. This high level of selectivity is essential for meeting the rigorous specifications demanded by the pharmaceutical industry, where trace impurities can compromise the safety and efficacy of the final Vitamin E product. Consequently, this mechanistic advantage translates directly into a more reliable supply of high-purity OLED material or pharmaceutical grade intermediates, reducing the risk of batch failures and ensuring consistent quality.
How to Synthesize 2,3,5-Trimethylbenzoquinone Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of catalyst preparation and reaction execution to ensure reproducibility and safety. The process begins with the hydrothermal synthesis of the Cu-SBA-15 catalyst, followed by the dissolution of the 2,3,6-trimethylphenol substrate in acetonitrile within a standard three-necked flask reactor. Operators must strictly adhere to the specified temperature ranges and molar ratios of hydrogen peroxide to maintain optimal selectivity between the quinone and hydroquinone products. While the general procedure is straightforward, precise control over reaction parameters is essential to maximize yield and minimize byproduct formation. The detailed standardized synthesis steps, including specific reagent quantities and workup procedures, are outlined in the guide below for technical reference.
- Prepare the Cu-SBA-15 catalyst by dissolving triblock copolymer P123 in acidic solution, mixing with aluminum isopropoxide, copper acetate, and TEOS, followed by hydrothermal crystallization and calcination.
- Dissolve 2,3,6-trimethylphenol in acetonitrile solvent within a reaction vessel and add the prepared Cu-SBA-15 catalyst.
- Heat the mixture to 50-80°C, add hydrogen peroxide oxidant, maintain reaction for approximately 3 hours, and analyze products via gas chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling strategic benefits that extend beyond mere technical feasibility. The transition to a one-step oxidation process fundamentally alters the cost structure of production by eliminating several expensive and time-consuming intermediate stages. This simplification of the manufacturing workflow leads to substantial cost savings by reducing labor requirements, energy consumption, and the capital intensity of the production facility. Furthermore, the reliance on readily available raw materials like 2,3,6-trimethylphenol and hydrogen peroxide mitigates supply chain risks associated with specialized or scarce reagents, ensuring a more stable and continuous supply of critical intermediates. The environmental friendliness of the process also reduces the regulatory burden and potential liabilities associated with hazardous waste management, making it a future-proof investment for sustainable operations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts that require expensive removal steps, combined with the removal of sulfonation and nitration units, drastically simplifies the production line and lowers operational expenditures. By avoiding the use of corrosive acids and the subsequent neutralization steps, the process significantly reduces the consumption of auxiliary chemicals and the wear and tear on reactor vessels. This streamlined approach allows for a more efficient allocation of resources, directing capital towards capacity expansion rather than waste treatment infrastructure. Ultimately, the reduced complexity of the process translates into a lower cost of goods sold, enhancing the competitiveness of the final product in the global market.
- Enhanced Supply Chain Reliability: The use of robust heterogeneous catalysts and stable reaction conditions ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted supply to downstream pharmaceutical customers. The simplified process flow reduces the number of potential failure points, thereby minimizing the risk of production delays caused by equipment malfunction or process deviations. Additionally, the ease of catalyst separation and potential for reuse contributes to a more resilient supply chain by reducing dependency on fresh catalyst shipments. This reliability is paramount for securing long-term contracts with major multinational corporations that prioritize supply continuity and quality assurance in their vendor selection criteria.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by water as the sole byproduct, aligns perfectly with global trends towards sustainable chemical manufacturing and circular economy principles. Scaling up this process is inherently safer and more manageable due to the absence of highly exothermic nitration steps and the use of mild oxidation conditions. This facilitates rapid commercial scale-up of complex pharmaceutical intermediates without the need for extensive environmental impact assessments or costly pollution control upgrades. Companies adopting this technology position themselves as leaders in eco-friendly manufacturing, appealing to environmentally conscious stakeholders and complying with increasingly strict international environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational advantages and technical nuances for potential partners and licensees evaluating this technology. Understanding these details is crucial for assessing the feasibility of integrating this process into existing manufacturing frameworks. The answers provided reflect the specific findings and claims made within the intellectual property documentation.
Q: What are the primary advantages of the Cu-SBA-15 catalyst over traditional methods?
A: The Cu-SBA-15 mesoporous molecular sieve offers superior product diffusion due to its pore structure and is significantly easier to separate from the reaction mixture compared to homogeneous catalysts, reducing downstream processing complexity.
Q: How does this method improve environmental compliance?
A: By utilizing hydrogen peroxide as the oxidant, the only byproduct generated is water, eliminating the massive amounts of acidic wastewater and heavy metal waste associated with traditional sulfonation and nitration routes.
Q: What are the optimal reaction conditions described in the patent?
A: The patent specifies a reaction temperature range of 50°C to 80°C using acetonitrile as the solvent, with a molar ratio of hydrogen peroxide to substrate between 1:1 and 3:1 for optimal conversion and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Trimethylhydroquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Cu-SBA-15 catalyzed oxidation route for producing high-value Vitamin E precursors with exceptional efficiency and purity. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2,3,5-trimethylhydroquinone or 2,3,5-trimethylbenzoquinone meets the highest international standards. We are committed to leveraging advanced catalytic technologies to deliver superior products that empower our clients to succeed in the competitive pharmaceutical and fine chemical markets.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible benefits of partnering with a leader in green chemical synthesis. Let us help you secure a reliable source of high-purity intermediates that drives your business forward.
