Advanced Green Synthesis of Cortisone Acetate for Scalable Pharmaceutical Manufacturing
Advanced Green Synthesis of Cortisone Acetate for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, particularly for high-volume steroid intermediates like cortisone acetate. Patent CN108373492B, published in late 2021, introduces a groundbreaking preparation method that fundamentally alters the synthetic landscape for this critical adrenocortical hormone precursor. This technology addresses the long-standing environmental and economic challenges associated with traditional steroid synthesis by eliminating the reliance on toxic heavy metal oxidants. For R&D directors and procurement strategists, this patent represents a viable pathway to producing high-purity pharmaceutical intermediates with a significantly reduced environmental footprint. The method leverages a sophisticated sequence of electrophilic bromination, dehydrobromination, and hydrolysis to achieve mass yields approaching 100% in the final step, demonstrating exceptional process efficiency.
Furthermore, the robustness of this synthetic route offers substantial advantages for supply chain stability. By utilizing common, commercially available reagents such as dibromohydantoin and perchloric acid instead of specialized fermentation-derived oxidants, manufacturers can mitigate risks associated with raw material scarcity. The process operates under relatively mild conditions, with key reactions occurring between 2°C and 30°C, which reduces energy consumption and enhances operational safety. This technical insight report analyzes the mechanistic depth and commercial viability of this novel approach, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cortisone acetate and related steroid hormones has relied heavily on semi-synthetic routes starting from diosgenin or microbial fermentation products like 4-androstenedione (4AD). While the fermentation route offered a cost advantage over plant extraction, it introduced severe environmental liabilities. The conventional oxidation steps to convert fermentation intermediates into the requisite 11-keto functionality typically employ chromium-containing metal oxides as oxidants and manganese-containing compounds as catalysts. This reliance results in wastewater streams heavily contaminated with Cr3+ and Mn2+ ions, necessitating expensive and complex treatment protocols to meet environmental discharge standards. Moreover, the multi-step nature of the diosgenin route, involving ring opening, acetylation, and multiple oxidation-elimination sequences, inherently suffers from low overall yields and high cumulative waste generation.
The Novel Approach
The methodology disclosed in patent CN108373492B circumvents these issues through a clever redesign of the functionalization strategy. Instead of direct oxidation with heavy metals, the process initiates with the bromination of 17 alpha-hydroxypregna-4, 9-diene-3, 20-dione acetate. This transformation introduces a bromine atom at the C9 position and a hydroxyl group at C11 simultaneously, setting the stage for the subsequent formation of the 11-ketone without external oxidants. The reaction scheme below illustrates this elegant progression from the diene starting material through the brominated intermediate to the final cortisone acetate product.
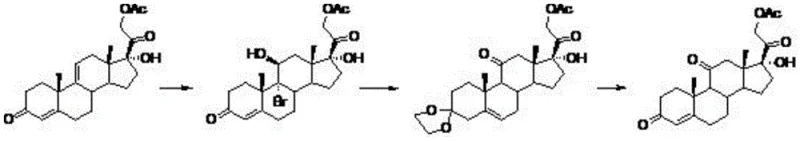
Following the initial bromination, the intermediate undergoes dehydrobromination coupled with ketal protection. This step is crucial as it stabilizes the sensitive carbonyl groups while facilitating the elimination of the bromine atom to establish the necessary double bond geometry. Finally, a mild acid hydrolysis removes the protecting groups and completes the oxidation state adjustments to yield cortisone acetate. This route not only avoids heavy metal pollution entirely but also allows for the recycling of solvents like acetone and ethyl acetate, drastically simplifying the downstream processing and waste management infrastructure required for production.
Mechanistic Insights into Electrophilic Bromination and Ketal Protection
The core of this synthetic innovation lies in the precise control of electrophilic addition and subsequent elimination reactions. In the first stage, the reaction is conducted in a mixed solvent system of acetone and water, acidified with perchloric acid. The use of dibromohydantoin or N-bromosuccinimide as the brominating agent ensures a controlled release of electrophilic bromine species. At the strictly maintained temperature of 2-4°C, the bromine adds across the 9(11) double bond of the pregna-diene substrate. The presence of water is critical here, as it acts as a nucleophile to trap the carbocation intermediate at the C11 position, resulting in the formation of the 9-bromo-11-beta-hydroxy motif found in Intermediate A. This regioselectivity is paramount, as it installs the oxygen functionality at C11 which will eventually become the ketone in the final cortisone structure.
Subsequent processing involves a thermal rearrangement and protection sequence. When Intermediate A is heated to 75°C in ethyl acetate with trimethyl orthoformate and ethylene glycol, a cascade of reactions occurs. The acidic conditions generated in situ promote the elimination of HBr, restoring unsaturation in the steroid nucleus. Simultaneously, the trimethyl orthoformate and ethylene glycol react with the C3 and C20 carbonyls to form cyclic ketals. This protection is strategic; it prevents unwanted side reactions at the carbonyl centers during the elimination phase and stabilizes the molecule for isolation. The final hydrolysis step using dilute sulfuric acid at 30°C is gentle enough to cleave the ketal protecting groups without degrading the sensitive steroid backbone, thereby delivering the target cortisone acetate with high structural integrity and minimal impurity formation.
How to Synthesize Cortisone Acetate Efficiently
The synthesis of cortisone acetate via this green pathway requires strict adherence to temperature controls and stoichiometric ratios to maximize yield and purity. The process is divided into three distinct operational stages: the low-temperature bromination to form the 9-bromo intermediate, the reflux-based dehydrobromination and protection to generate the ketal, and the final mild acid hydrolysis. Each step builds upon the previous one to ensure the correct stereochemistry and oxidation state are achieved without the need for hazardous oxidants. For detailed laboratory protocols and specific equipment configurations required to implement this route, please refer to the standardized synthesis guide below.
- Perform electrophilic bromination of 17 alpha-hydroxypregna-4, 9-diene-3, 20-dione acetate using dibromohydantoin at 2-4°C in an acetone-water-perchloric acid system to yield the 9-bromo intermediate.
- Execute dehydrobromination and carbonyl protection by refluxing the intermediate with trimethyl orthoformate and ethylene glycol in ethyl acetate at 75°C to form the ketal-protected steroid.
- Conduct final hydrolysis using dilute sulfuric acid in acetone at 30°C, followed by neutralization and crystallization to isolate cortisone acetate with purity exceeding 92%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this heavy-metal-free synthesis route offers transformative benefits beyond mere regulatory compliance. The elimination of chromium and manganese reagents removes the need for specialized wastewater treatment facilities designed to precipitate and remove toxic heavy metals. This simplification of the effluent treatment process translates directly into significant operational cost savings and reduced liability. Furthermore, the solvents employed in this process, primarily acetone and ethyl acetate, are commodity chemicals with stable global supply chains. Their volatility allows for efficient recovery and reuse through standard distillation units, creating a closed-loop solvent system that minimizes raw material expenditure and reduces the volume of hazardous waste requiring disposal.
- Cost Reduction in Manufacturing: The removal of expensive and regulated heavy metal oxidants fundamentally lowers the bill of materials for cortisone acetate production. Additionally, the high mass yield reported in the patent examples suggests that less starting material is wasted, improving the overall atom economy of the process. By avoiding the complex purification steps often needed to strip trace metals from pharmaceutical intermediates, manufacturers can reduce both processing time and the consumption of auxiliary adsorbents or chelating agents, leading to substantial cost savings in the final product cost structure.
- Enhanced Supply Chain Reliability: Relying on common organic reagents like dibromohydantoin and perchloric acid mitigates the supply risk associated with specialized fermentation substrates or rare metal catalysts. These chemicals are produced at a massive scale for various industries, ensuring consistent availability and price stability. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream API manufacturers, thereby strengthening the resilience of the entire pharmaceutical supply network against raw material shortages.
- Scalability and Environmental Compliance: The mild reaction conditions, particularly the low-temperature bromination and ambient-pressure hydrolysis, make this process highly amenable to large-scale reactor operations without requiring exotic high-pressure or cryogenic equipment. The absence of heavy metal discharge simplifies environmental permitting and compliance reporting, allowing facilities to operate with greater flexibility. This environmental friendliness aligns perfectly with the increasing global demand for green chemistry practices, enhancing the marketability of the final product to eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel cortisone acetate synthesis route. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on purity profiles, reaction mechanisms, and scalability factors. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this new process eliminate heavy metal contamination?
A: Unlike traditional fermentation routes that require chromium-containing oxidants and manganese catalysts, this method utilizes dibromohydantoin for bromination and organic acids for hydrolysis, ensuring wastewater is free of toxic Cr3+ and Mn2+ ions.
Q: What is the expected purity profile of the final cortisone acetate?
A: Experimental data from the patent indicates that the final product achieves a high-performance liquid chromatography (HPLC) purity ranging from 92% to 98%, with major impurities controlled below 2.5% through precise pH adjustment and crystallization.
Q: Is the solvent system suitable for industrial recycling?
A: Yes, the process primarily employs acetone and ethyl acetate, which are volatile organic solvents that can be efficiently recovered via reduced pressure concentration and distillation, significantly lowering raw material consumption and waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cortisone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient synthetic routes for high-value steroid intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the heavy-metal-free cortisone acetate process can be seamlessly transitioned from the laboratory to the manufacturing floor. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite forward-thinking pharmaceutical companies to collaborate with us on optimizing this green synthesis pathway for your specific supply chain needs. By leveraging our technical expertise, you can achieve a Customized Cost-Saving Analysis tailored to your production volumes. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a secure and sustainable supply of high-purity cortisone acetate for your drug development pipelines.
