Advanced Valsartan Manufacturing: A Safer, High-Yield Synthetic Route for Global API Production
Introduction to Advanced Valsartan Synthesis Technology
The pharmaceutical landscape for Angiotensin II receptor blockers (ARBs) demands manufacturing processes that balance high purity with operational safety and cost efficiency. Patent CN100522953C introduces a transformative synthetic methodology for Valsartan, a blockbuster antihypertensive agent, addressing critical bottlenecks found in legacy production routes. This innovation centers on a streamlined sequence involving the alkylation of a stable tetrazole-containing biphenyl derivative with valine methyl ester, followed by controlled acylation and hydrolysis. Unlike earlier generations of synthesis which relied on hazardous organotin reagents or complex hydrogenation steps, this approach leverages robust nucleophilic substitution chemistry to construct the core molecular architecture. For R&D directors and procurement strategists, this patent represents a pivotal shift towards greener, more reliable pharmaceutical intermediates sourcing. The method not only secures the stereochemical integrity of the L-valine moiety but also simplifies the downstream purification processes, offering a compelling value proposition for generic API manufacturers seeking to optimize their supply chains.
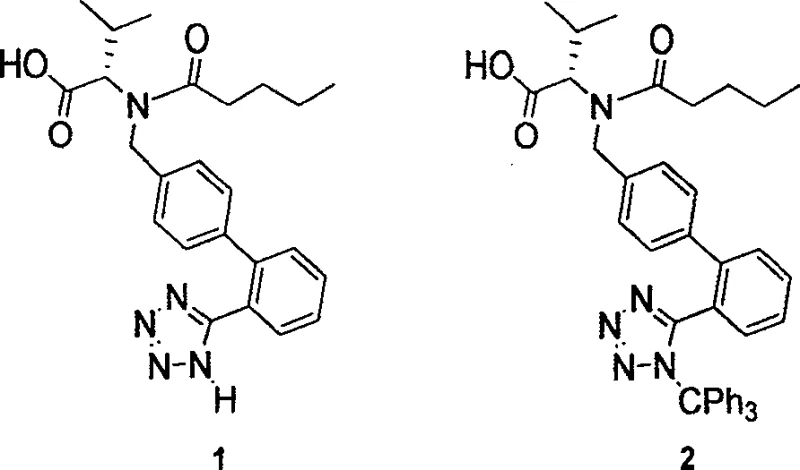
The core of this technology lies in the preparation of the key intermediate, N-(1-oxopentyl)-N-[[2'-(N'-trityl-tetrazole-5-yl)-(1,1'-phenylbenzene)-4-yl]-methyl]-L-valine. By establishing a convergent route that couples the biphenyl-tetrazole fragment early in the sequence, the process minimizes the number of unit operations required to reach the final active pharmaceutical ingredient. This structural strategy ensures that the sensitive tetrazole ring is protected throughout the vigorous acylation and hydrolysis steps, preventing degradation and ensuring consistent batch-to-batch quality. As we delve deeper into the technical specifics, it becomes evident that this methodology is not merely an academic exercise but a commercially viable solution designed to meet the rigorous demands of modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Valsartan has been plagued by significant safety and efficiency challenges that impact both cost and supply continuity. One prominent legacy route, described in U.S. Patent 5,399,578, relies on the reaction of a nitrile precursor with tributyltin azide to generate the tetrazole ring. This transformation requires extreme thermal conditions, typically between 120°C and 140°C, posing severe safety risks due to the potential for exothermic runaway and the inherent toxicity of organotin compounds. Furthermore, the removal of these tin residues requires extensive and costly purification steps to meet strict heavy metal limits in pharmaceutical products. Another historical approach involves the use of sodium azide with phase transfer catalysts, which, while avoiding tin, introduces the handling hazards associated with explosive inorganic azides. Additionally, older strategies often employed benzyl protecting groups that necessitated catalytic hydrogenation for removal. This requirement mandates the use of specialized high-pressure hydrogenation reactors, increasing capital expenditure and limiting the flexibility of multi-purpose manufacturing facilities. These conventional pathways often suffer from suboptimal yields, with some reports indicating total recoveries as low as 50%, thereby inflating the cost of goods sold and creating supply vulnerabilities.
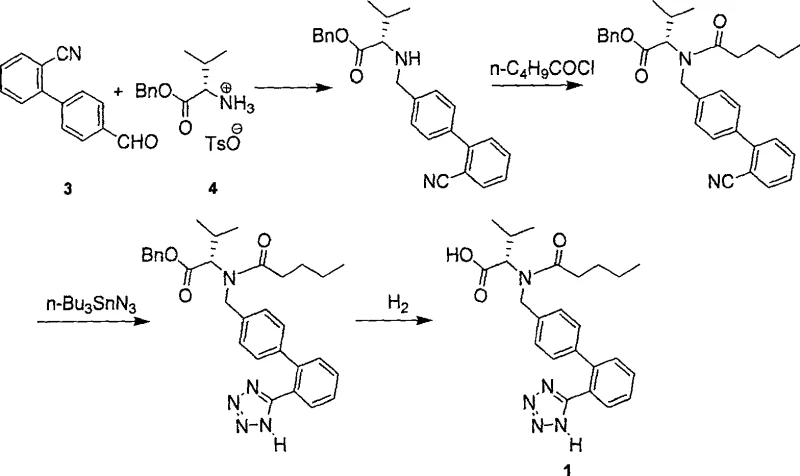
The Novel Approach
In stark contrast to these hazardous and inefficient legacy methods, the novel synthesis outlined in Patent CN100522953C offers a paradigm shift by utilizing a pre-functionalized building block, 2'-(N-trityl)tetrazole-4-bromomethylbiphenyl (BBTT). This strategic choice eliminates the need for on-site tetrazole ring formation, thereby bypassing the use of toxic tin azides and high-temperature reactions entirely. The new route initiates with a mild nucleophilic substitution between BBTT and valine methyl ester, conducted at temperatures ranging from -20°C to 80°C, which is far more energy-efficient and safer to operate. Following alkylation, the introduction of the valeryl side chain is achieved through standard acylation chemistry, avoiding the water-sensitive conditions that plagued previous Schiff-base reduction methods. Crucially, the deprotection strategy employs acidic hydrolysis rather than catalytic hydrogenation, removing the need for expensive high-pressure equipment and precious metal catalysts. This streamlined workflow not only enhances the overall yield to over 67% but also significantly improves the optical purity of the final product to greater than 99%, ensuring superior therapeutic efficacy and regulatory compliance for high-purity pharmaceutical intermediates.
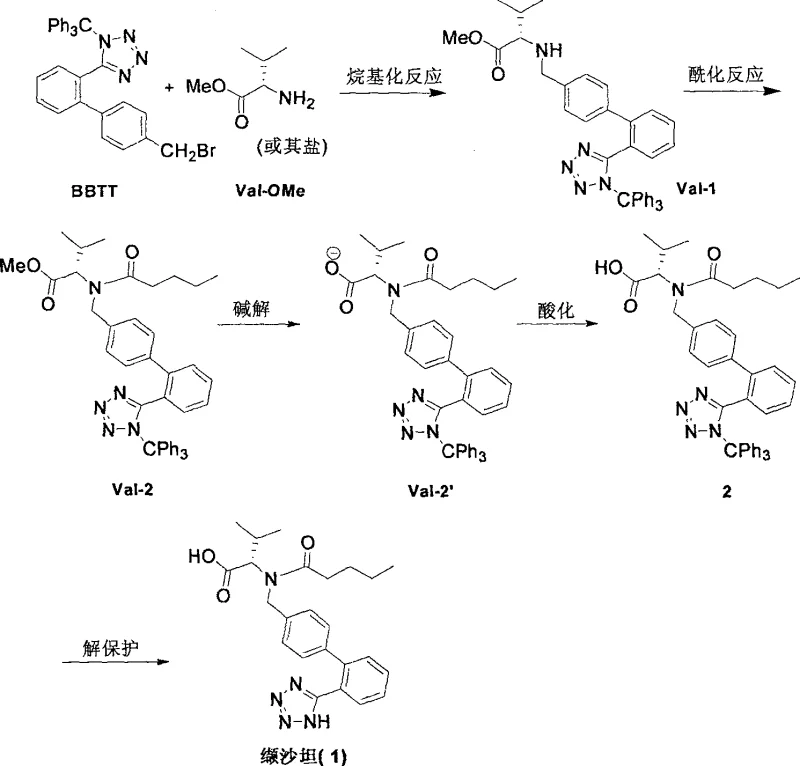
Mechanistic Insights into Nucleophilic Substitution and Acylation
The chemical elegance of this synthesis lies in its precise control over reaction kinetics and stereochemistry during the bond-forming events. The initial alkylation step involves the nucleophilic attack of the amino group of valine methyl ester on the benzylic carbon of the BBTT molecule. This reaction is facilitated by the use of organic bases such as diisopropylethylamine (DIPEA) or triethylamine, which serve to scavenge the hydrobromic acid byproduct and drive the equilibrium forward. The choice of solvent, typically dichloromethane or acetonitrile, is critical for solubilizing both the lipophilic biphenyl substrate and the amino acid ester, ensuring a homogeneous reaction environment that maximizes collision frequency. By maintaining the reaction temperature between -10°C and 10°C during the addition phase, the process effectively suppresses potential side reactions such as elimination or over-alkylation, preserving the integrity of the chiral center at the alpha-carbon of the valine residue. This meticulous temperature control is a hallmark of robust process chemistry, ensuring that the resulting secondary amine intermediate retains the desired L-configuration essential for biological activity.
Subsequent acylation with n-valeryl chloride proceeds via a classic nucleophilic acyl substitution mechanism. The secondary amine nitrogen attacks the carbonyl carbon of the acid chloride, forming a tetrahedral intermediate that collapses to release chloride ion and form the stable amide bond. The presence of a base is again essential here to neutralize the generated HCl, preventing the protonation of the amine which would render it non-nucleophilic. The final hydrolysis step converts the methyl ester to the free carboxylic acid using lithium hydroxide in a mixed solvent system of THF and water. This saponification is highly selective for the ester moiety, leaving the amide bond and the trityl-protected tetrazole ring intact. The use of lithium hydroxide is particularly advantageous as it often provides cleaner reaction profiles and easier workup compared to sodium or potassium hydroxide. Finally, the removal of the trityl group under acidic conditions reveals the acidic tetrazole proton, completing the synthesis. This sequence demonstrates a deep understanding of orthogonal protecting group chemistry, allowing for the construction of a complex molecule with minimal interference between functional groups.
How to Synthesize Valsartan Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and purity. The process is divided into four distinct stages: alkylation, acylation, hydrolysis, and deprotection. Each stage utilizes common industrial solvents and reagents, making the transition from laboratory to pilot plant straightforward. The following guide outlines the critical operational windows and reagent stoichiometry derived from the experimental examples provided in the intellectual property documentation. For process engineers and chemists, understanding these specific conditions is vital for replicating the high success rates reported in the patent.
- Perform nucleophilic substitution between 2'-(N-trityl)tetrazole-4-bromomethylbiphenyl (BBTT) and valine methyl ester in the presence of an organic base like DIPEA at -10 to 10°C to form the secondary amine intermediate.
- Conduct acylation of the secondary amine with n-valeryl chloride using triethylamine in ethyl acetate at temperatures ranging from -10°C to 35°C to introduce the side chain.
- Execute alkaline hydrolysis of the methyl ester using lithium hydroxide in THF/water mixture, followed by acidification and crystallization to obtain the protected acid intermediate with high optical purity.
- Remove the trityl protecting group under acidic conditions (e.g., HCl in DCM) and isolate the final Valsartan product through pH adjustment and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain directors tasked with securing reliable sources of cardiovascular API intermediates. The most immediate benefit is the drastic reduction in raw material hazards. By eliminating tributyltin azide and sodium azide, manufacturers can significantly lower their environmental, health, and safety (EHS) compliance costs. There is no longer a need for specialized waste streams to handle toxic tin residues or explosive azide byproducts, which simplifies waste management and reduces disposal fees. Furthermore, the avoidance of catalytic hydrogenation removes a major bottleneck in production scheduling; hydrogenation units are often shared resources in multi-product facilities and can be prone to downtime. By switching to a chemical deprotection method, production throughput becomes more predictable and less dependent on specialized equipment availability, directly enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The economic implications of this route are substantial, driven primarily by the simplification of the process workflow. Traditional methods often require multiple purification steps to remove heavy metal catalysts and tin residues, which consume large volumes of silica gel, solvents, and chelating agents. This novel method produces a cleaner crude profile, reducing the load on downstream purification units and lowering the consumption of chromatography media. Additionally, the higher overall yield (>67% compared to ~50% in older methods) means that less starting material is required to produce the same amount of final API, directly reducing the cost of goods sold. The use of commodity reagents like valine methyl ester and valeryl chloride, which are available in bulk quantities at competitive prices, further stabilizes the cost structure against market volatility.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for generic drug manufacturers, and this synthesis enhances stability by relying on robust, commercially available starting materials. The key building block, BBTT, is a stable solid that can be stockpiled without significant degradation, unlike sensitive azide reagents that may have limited shelf lives or require cold chain logistics. The reaction conditions are mild and tolerant of minor variations in temperature and mixing, making the process robust against operational deviations that might otherwise lead to batch failures. This robustness translates to fewer rejected batches and a more consistent supply of high-purity pharmaceutical intermediates to the formulation teams, ensuring that drug product manufacturing schedules are met without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this route is designed with scalability in mind. The exotherms associated with the alkylation and acylation steps are manageable with standard jacketed reactor cooling systems, eliminating the need for cryogenic setups that are difficult to scale. Moreover, the environmental footprint of the process is significantly reduced. The absence of heavy metals aligns with increasingly stringent global regulations regarding elemental impurities in drugs (ICH Q3D). By generating less hazardous waste and avoiding persistent organic pollutants like organotins, manufacturers can more easily obtain environmental permits and maintain a sustainable operating license, which is a critical factor for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Valsartan synthesis technology. These answers are derived directly from the experimental data and claims presented in Patent CN100522953C, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific manufacturing contexts.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: Traditional methods often utilize toxic tributyltin azide at high temperatures (120-140°C) to form the tetrazole ring. This novel route starts with a pre-formed tetrazole building block (BBTT), completely eliminating the need for hazardous tin reagents and high-temperature azide chemistry, significantly reducing workplace toxicity risks.
Q: What represents the key advantage in terms of optical purity for this process?
A: The process maintains the chiral integrity of the L-valine starting material throughout the alkylation and acylation steps. By avoiding harsh racemization conditions and utilizing mild hydrolysis protocols, the final Valsartan product achieves an optical purity exceeding 99%, which is critical for regulatory compliance in cardiovascular therapeutics.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route is designed for industrial scalability. It utilizes common organic solvents like dichloromethane, THF, and ethyl acetate, and operates within a moderate temperature range (-20°C to 80°C). Furthermore, it avoids the need for specialized hydrogenation equipment required in older benzyl-deprotection methods, simplifying the engineering requirements for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a new synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in complex organic synthesis, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactor trains capable of handling the specific solvent systems and temperature profiles required for this Valsartan process, including low-temperature alkylation and precise pH-controlled hydrolysis. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of intermediate meets the highest standards of quality, ready for subsequent conversion to the final API. Our commitment to excellence ensures that your supply chain remains resilient and compliant with global regulatory requirements.
We invite you to collaborate with us to leverage this advanced synthesis technology for your Valsartan projects. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us help you secure a competitive advantage in the cardiovascular market with a supply partner dedicated to innovation and reliability.
