Advanced Semi-Synthesis of Taxol and Docetaxel for Commercial Scale-Up
Advanced Semi-Synthesis of Taxol and Docetaxel for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methods for producing high-value anticancer agents, and patent CN101020676A presents a significant breakthrough in the semi-synthesis of Taxol and Docetaxel. This technology addresses the critical bottlenecks associated with natural extraction, such as low yield and ecological concerns, by introducing a streamlined chemical pathway that couples chiral side chains with protected 10-deacetyl baccatin III. The process is characterized by its mild reaction conditions, high overall yield, and exceptional suitability for industrial production, making it a vital asset for any reliable API intermediate supplier looking to optimize their portfolio. By leveraging this novel approach, manufacturers can secure a more stable supply of these essential oncology drugs while maintaining stringent quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of Taxol and Docetaxel has relied heavily on extraction from the bark or needles of yew trees, a method fraught with significant logistical and environmental challenges. The content of these active compounds in natural plant sources is extremely low, necessitating the processing of vast quantities of biomass to obtain clinically relevant amounts, which places immense pressure on natural resources and leads to inconsistent supply chains. Furthermore, total chemical synthesis of these complex molecules involves numerous steps, often exceeding forty stages, which results in prohibitively low overall yields and generates substantial chemical waste. These conventional methods struggle to meet the growing global demand for cancer therapeutics, creating vulnerabilities in the supply chain that can impact patient access and drive up costs for healthcare systems worldwide.
The Novel Approach
In contrast, the semi-synthetic route disclosed in the patent data offers a transformative solution by utilizing 10-deacetyl baccatin III as a abundant starting material derived from renewable plant sources. This method drastically shortens the synthetic sequence by focusing on the efficient coupling of a pre-formed chiral side chain to the taxane core, thereby bypassing the need to construct the complex ring system from scratch. The process employs mild reaction conditions and high-efficiency reagents that facilitate high conversion rates, ensuring that the final products meet the rigorous purity specifications demanded by the pharmaceutical industry. This approach not only enhances the economic viability of production but also aligns with green chemistry principles by reducing the overall environmental footprint associated with manufacturing these life-saving medications.
Mechanistic Insights into Chiral Side Chain Condensation and Inversion
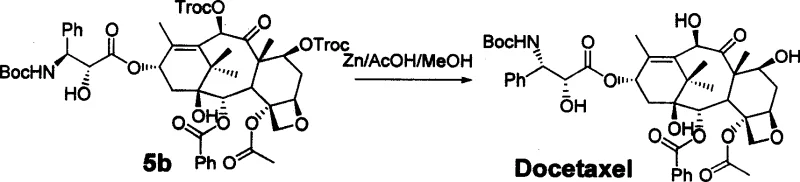
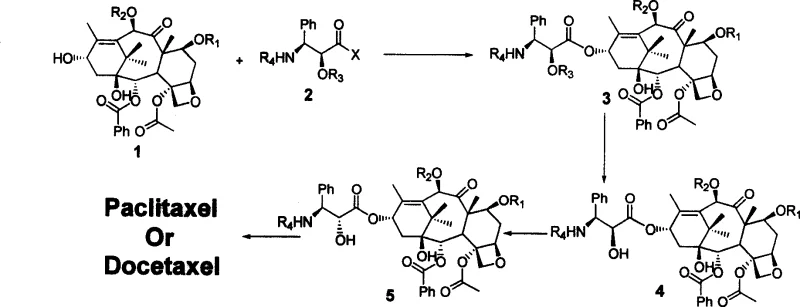
The core of this synthetic strategy lies in the precise condensation reaction between the protected 10-deacetyl baccatin III and a novel chiral side chain, which is facilitated by carbonyl diimine class condensing agents such as DCC in the presence of DMAP. This reaction is carefully controlled at temperatures between 30 to 40°C to ensure optimal stereochemical outcomes and minimize side reactions that could compromise the integrity of the sensitive taxane core. The use of specific protecting groups, such as TBS, TES, or Troc, allows for selective manipulation of hydroxyl functionalities, ensuring that the condensation occurs exclusively at the desired C13 position. This level of control is paramount for maintaining the biological activity of the final molecule, as even minor deviations in stereochemistry can render the compound inactive or toxic.
Following the initial coupling, the process involves a critical configuration inversion step to establish the correct stereochemistry at the 2'-position of the side chain, which is essential for the drug's mechanism of action. This inversion can be achieved through either an oxazoline-mediated pathway for Taxol or a Mitsunobu reaction for Docetaxel, both of which are highly efficient methods for stereoselective transformation. The subsequent removal of protecting groups is executed under mild acidic or reductive conditions, such as using zinc powder in acetic acid, which preserves the delicate ester linkages within the molecule. This meticulous attention to mechanistic detail ensures that the final product possesses the exact structural attributes required for potent microtubule stabilization and anticancer activity.

How to Synthesize Taxol and Docetaxel Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these complex molecules, starting with the condensation of the core structure with the chiral side chain followed by sequential deprotection and inversion steps. This standardized approach allows for reproducible results and facilitates the transfer of technology from laboratory scale to commercial manufacturing environments. For detailed operational parameters, reagent ratios, and specific workup procedures, manufacturers should refer to the standardized synthesis steps provided in the technical documentation below, which ensures consistency and compliance with Good Manufacturing Practices (GMP).
- Condensation of protected 10-deacetyl baccatin III with a chiral side chain using DCC and DMAP at 30-40°C.
- Removal of the 2'-hydroxyl protecting group to expose the reactive site for configuration inversion.
- Inversion of the side chain configuration via Oxazoline or Mitsunobu reaction to achieve the bioactive stereochemistry.
- Final global deprotection using Zinc powder in acetic acid and methanol to yield the final Taxol or Docetaxel product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this semi-synthetic route offers substantial advantages for procurement managers and supply chain heads by significantly simplifying the production landscape and reducing dependency on volatile natural resources. The ability to synthesize the trans chiral side chain with greater ease compared to cis variants translates directly into lower raw material costs and reduced processing time, which enhances the overall cost-effectiveness of the manufacturing process. Furthermore, the high yields reported in the patent examples indicate a robust process that minimizes waste and maximizes output per batch, providing a competitive edge in pricing strategies for generic and branded pharmaceutical products alike.
- Cost Reduction in Manufacturing: The elimination of complex total synthesis steps and the use of readily available starting materials like 10-deacetyl baccatin III lead to a drastic simplification of the production workflow. By avoiding the need for expensive transition metal catalysts and harsh reaction conditions, the process inherently lowers the operational expenditure associated with energy consumption and waste disposal. This streamlined approach allows for significant cost savings that can be passed down the supply chain, making high-quality anticancer treatments more accessible to global markets without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: Relying on semi-synthesis rather than direct extraction mitigates the risks associated with seasonal variations and geopolitical instability affecting raw plant material availability. The chemical synthesis of the side chain can be scaled independently and stored, ensuring a continuous supply of key intermediates even when natural harvests are insufficient. This decoupling of supply from agricultural cycles provides procurement teams with greater predictability and control over inventory levels, reducing the likelihood of stockouts and ensuring uninterrupted production schedules for critical medication.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this process make it exceptionally well-suited for commercial scale-up from pilot plants to multi-ton production facilities. The reduction in hazardous waste generation and the use of less toxic reagents align with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing sites. This sustainability profile not only protects the company from regulatory risks but also enhances its corporate reputation as a responsible manufacturer committed to eco-friendly production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic technology, based on the specific advantages and mechanisms detailed in the patent literature. These insights are designed to clarify the feasibility of adoption and the potential impact on production efficiency for stakeholders evaluating this technology for their supply chains. Understanding these nuances is crucial for making informed decisions about integrating this process into existing manufacturing frameworks to maximize value and operational excellence.
Q: What is the key advantage of using trans chiral side chains in this synthesis?
A: The use of trans chiral side chains significantly reduces synthetic difficulty compared to cis side chains, leading to simpler reaction conditions and higher overall yields suitable for industrial production.
Q: How does this process address the supply limitations of natural extraction?
A: By utilizing semi-synthesis from 10-deacetyl baccatin III, this method bypasses the low content and ecological constraints associated with extracting taxanes directly from yew plants, ensuring a more reliable and scalable supply chain.
Q: What are the typical reaction conditions for the condensation step?
A: The condensation reaction is conducted under mild conditions, typically between 30 to 40°C, using dicyclohexyl carbonyl diimine (DCC) and DMAP as catalysts in toluene, which facilitates high conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taxol and Docetaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for life-saving oncology medications and possess the technical expertise to bring complex semi-synthetic routes like this to commercial fruition. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Taxol or Docetaxel intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our advanced process development capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, where we can provide specific COA data and route feasibility assessments tailored to your production needs. By partnering with us, you gain access to a wealth of chemical expertise and a commitment to quality that will strengthen your position in the competitive pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →