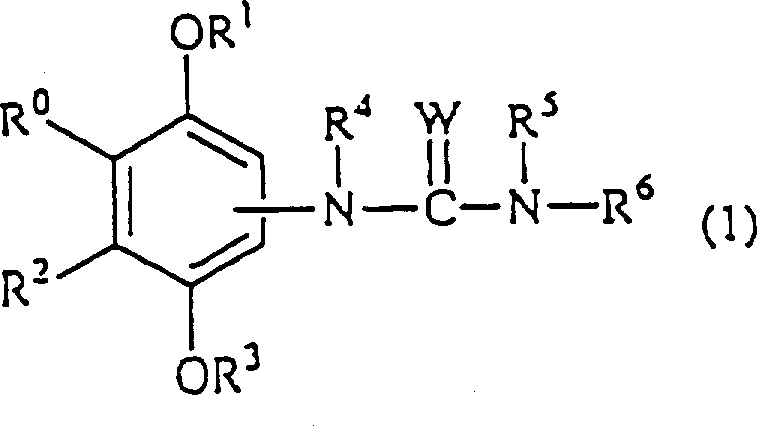
Advanced Phenol Derivative Synthesis For Commercial Scale-Up Of Complex Ureido Compounds
The pharmaceutical landscape for cardiovascular therapeutics is undergoing a significant paradigm shift, driven by the urgent need to address the root causes of atherosclerosis rather than merely managing symptoms. Patent CN1165815A introduces a groundbreaking class of phenol compounds that offer a dual-mechanism approach, simultaneously targeting Acyl-CoA Cholesterol Acyltransferase (ACAT) inhibition and oxidative stress reduction. This technological breakthrough represents a critical advancement for research teams focused on developing next-generation anti-atherosclerotic agents that can prevent foam cell formation more effectively than traditional statins. The core innovation lies in the specific structural arrangement of the ureido-substituted phenol scaffold, which allows for potent biological activity while maintaining a favorable safety profile. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this chemistry is essential for scaling these molecules from benchtop discovery to commercial viability. The following analysis dissects the synthetic strategy and commercial implications of this patent, providing actionable insights for R&D directors and procurement specialists aiming to secure a competitive edge in the cardiovascular drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of anti-atherosclerotic therapies has been plagued by a singular focus on lowering serum cholesterol levels, often neglecting the critical role of oxidative modification in low-density lipoprotein (LDL). Conventional methods typically rely on HMG-CoA reductase inhibitors, which, while effective at reducing cholesterol biosynthesis, do not adequately prevent the accumulation of cholesterol esters within macrophage foam cells. Furthermore, many existing candidates lack the inherent antioxidant capacity required to neutralize reactive oxygen species (ROS) at the site of arterial injury. This limitation results in a therapeutic gap where plaque progression continues despite lipid-lowering therapy. From a manufacturing perspective, older synthetic routes for similar phenolic compounds often suffered from poor regioselectivity, leading to complex impurity profiles that were difficult and costly to purge. The reliance on harsh reaction conditions in traditional phenol functionalization also posed significant safety hazards and environmental burdens, complicating the path to regulatory approval and commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN1165815A overcomes these historical barriers by integrating a robust urea linkage directly onto a sterically hindered phenol core. This novel approach ensures that the phenolic hydroxyl group remains available to scavenge free radicals, thereby inhibiting the oxidative modification of LDL, which is a prerequisite for foam cell formation. Simultaneously, the ureido side chain is engineered to fit the active site of the ACAT enzyme, blocking the re-esterification of cholesterol. This dual functionality is achieved through a modular synthetic strategy that allows for extensive structure-activity relationship (SAR) exploration without compromising the core stability. By utilizing protected intermediates, such as methoxymethyl ethers, the process avoids premature oxidation during the coupling steps, significantly enhancing the overall yield and purity of the final active pharmaceutical ingredient. This strategic design not only improves therapeutic efficacy but also streamlines the manufacturing workflow, offering substantial cost savings in API manufacturing by reducing the number of purification cycles required.
Mechanistic Insights into ACAT Inhibition and Antioxidant Activity
The biological efficacy of these phenol derivatives is deeply rooted in their precise molecular architecture, which facilitates a synergistic interaction with cellular targets. The phenolic hydroxyl group, particularly when positioned ortho to bulky substituents like the tert-butyl group, acts as a potent hydrogen donor to neutralize lipid peroxyl radicals. This antioxidant mechanism is crucial for stabilizing the arterial wall environment and preventing the cascade of inflammatory responses triggered by oxidized LDL. In parallel, the urea moiety extends into the hydrophobic channel of the ACAT enzyme, forming key hydrogen bonds that competitively inhibit the transfer of fatty acyl groups to cholesterol. This dual action prevents the transformation of macrophages into lipid-laden foam cells, effectively halting the progression of atherosclerotic plaques at a cellular level. The structural flexibility provided by the various R-group substitutions allows medicinal chemists to fine-tune the lipophilicity and metabolic stability of the compound, ensuring optimal bioavailability. Understanding this mechanistic interplay is vital for R&D teams aiming to optimize lead candidates for clinical trials.
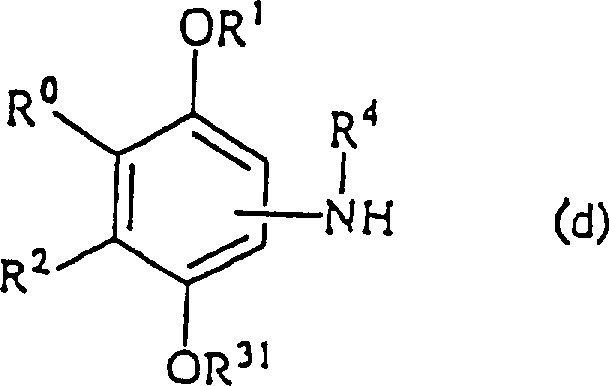
Controlling the impurity profile during the synthesis of these complex molecules is paramount to ensuring patient safety and regulatory compliance. The patent outlines specific protection strategies, such as the use of methoxymethyl groups, which shield the reactive phenolic oxygen from unwanted side reactions during the formation of the urea bond. This protective measure is critical because free phenols are prone to oxidation and polymerization under the basic conditions often required for amine coupling. By masking the hydroxyl group, the reaction proceeds with high chemoselectivity, minimizing the formation of colored impurities and oligomeric byproducts that are notoriously difficult to remove. Furthermore, the final deprotection step is designed to be mild yet efficient, typically employing acidic hydrolysis that cleaves the ether without degrading the sensitive urea linkage. This rigorous control over the reaction pathway ensures that the final high-purity phenol derivatives meet the stringent specifications required for parenteral or oral administration, thereby reducing the risk of adverse immune reactions caused by trace contaminants.
How to Synthesize 2-Ureido-4-Methoxy-6-Tert-Butylphenol Efficiently
The synthesis of these high-value intermediates follows a logical sequence designed to maximize yield while maintaining operational safety and scalability. The process begins with the preparation of a protected aniline precursor, where the phenolic hydroxyl is masked to prevent interference during subsequent coupling reactions. This intermediate is then reacted with an activated carbonyl source, such as an isocyanate or a chloroformate derivative, to establish the critical urea backbone. The reaction conditions are carefully controlled to manage exotherms and ensure complete conversion, often utilizing common organic solvents that are easily recovered and recycled. Following the coupling, the protecting group is removed under acidic conditions to reveal the active phenolic moiety, followed by crystallization to achieve the desired purity. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this process.
- Protect the phenolic hydroxyl group of the starting aniline derivative using a methoxymethyl ether group to prevent side reactions during coupling.
- React the protected aniline intermediate with an isocyanate or thiophosgene-derived active species to form the core ureido or thioureido linkage.
- Remove the protecting group under acidic conditions to reveal the free phenolic hydroxyl, crucial for antioxidant activity, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers distinct logistical and economic benefits that extend beyond simple unit cost calculations. The reliance on readily available starting materials, such as substituted phenols and common amines, mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Additionally, the robustness of the protection-deprotection strategy allows for the isolation of stable intermediates, which can be stockpiled without significant degradation, providing a buffer against demand fluctuations. This flexibility enhances supply chain reliability and ensures consistent delivery schedules for downstream API manufacturers. By eliminating the need for exotic catalysts or extreme reaction conditions, the process also reduces the burden on facility infrastructure, allowing for production in standard multipurpose reactors. These factors collectively contribute to a more resilient and cost-effective supply network for cardiovascular therapeutics.
- Cost Reduction in Manufacturing: The synthetic pathway described in the patent eliminates the need for expensive transition metal catalysts often required in traditional cross-coupling reactions, leading to significant cost optimization. By utilizing straightforward condensation reactions and acid hydrolysis, the process reduces the consumption of high-cost reagents and simplifies the waste treatment protocol. The ability to isolate stable intermediates also minimizes material loss due to degradation during storage, further improving the overall mass balance. Consequently, manufacturers can achieve substantial cost savings without compromising the quality or potency of the final pharmaceutical intermediate. This economic efficiency makes the technology highly attractive for generic drug producers looking to optimize their margins.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are commodity chemicals with established global supply networks, reducing the dependency on single-source vendors. The stability of the protected intermediates allows for strategic inventory management, enabling manufacturers to build safety stock without the fear of rapid decomposition. This capability is crucial for maintaining continuity of supply in the face of geopolitical disruptions or transportation delays. Furthermore, the synthetic steps are compatible with standard stainless steel equipment, removing the need for specialized glass-lined or Hastelloy reactors. These attributes collectively strengthen the supply chain, ensuring that high-purity antioxidants and ACAT inhibitors are available when needed for critical drug production.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram-level laboratory batches to multi-ton commercial production without significant re-engineering. The use of common solvents like dichloromethane and ethyl acetate facilitates efficient solvent recovery systems, aligning with modern green chemistry principles and environmental regulations. The absence of heavy metal residues simplifies the purification process and reduces the environmental footprint associated with waste disposal. This compliance with strict environmental standards not only avoids regulatory fines but also enhances the corporate sustainability profile of the manufacturing entity. Such scalability ensures that the commercial scale-up of complex ureido compounds can meet global demand efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these phenol derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They cover aspects ranging from reaction kinetics to impurity control, offering clarity for stakeholders evaluating this technology for integration into their product pipelines. Understanding these details is essential for making informed decisions about licensing, procurement, or internal development.
Q: What is the primary therapeutic mechanism of these phenol derivatives?
A: These compounds function through a dual mechanism: inhibiting Acyl-CoA Cholesterol Acyltransferase (ACAT) to prevent cholesterol esterification and scavenging reactive oxygen species (ROS) to inhibit LDL oxidation.
Q: Are the synthesis intermediates stable for large-scale storage?
A: Yes, the patent describes protected intermediates such as methoxymethyl ethers which exhibit enhanced stability compared to the free phenol, facilitating safer transport and storage before final deprotection.
Q: Can this process be adapted for GMP manufacturing?
A: The synthetic route utilizes standard organic transformations like urea condensation and acid hydrolysis, which are well-established in GMP environments and allow for rigorous impurity control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ureido-4-Methoxy-6-Tert-Butylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent chemistry into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from pilot plant to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenol derivative meets the highest international standards. Our commitment to quality assurance means that you can trust our materials for your most sensitive clinical and commercial applications. We understand the complexities of cardiovascular drug synthesis and are equipped to handle the specific challenges associated with urea linkages and phenolic stability.
We invite you to collaborate with us to leverage this advanced technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline. Let us be your partner in delivering high-quality pharmaceutical intermediates that drive therapeutic success.
