Advanced Stereoselective Synthesis of Epipodophyllotoxin Intermediates for Commercial Scale-up
Introduction to Novel Epipodophyllotoxin Synthesis
The pharmaceutical industry continuously seeks robust pathways for the production of potent antineoplastic agents, particularly those derived from the podophyllotoxin class. Patent CN1020596C discloses a groundbreaking stereoselective total synthesis method for epipodophyllotoxin and its related intermediates, addressing critical bottlenecks in traditional manufacturing. This technology represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to secure the supply chain for cancer therapeutics like etoposide and teniposide. By circumventing the thermodynamic limitations of earlier epimerization strategies, this process offers a direct route to the desired cis-configured aryl tetralin scaffolds essential for biological activity.
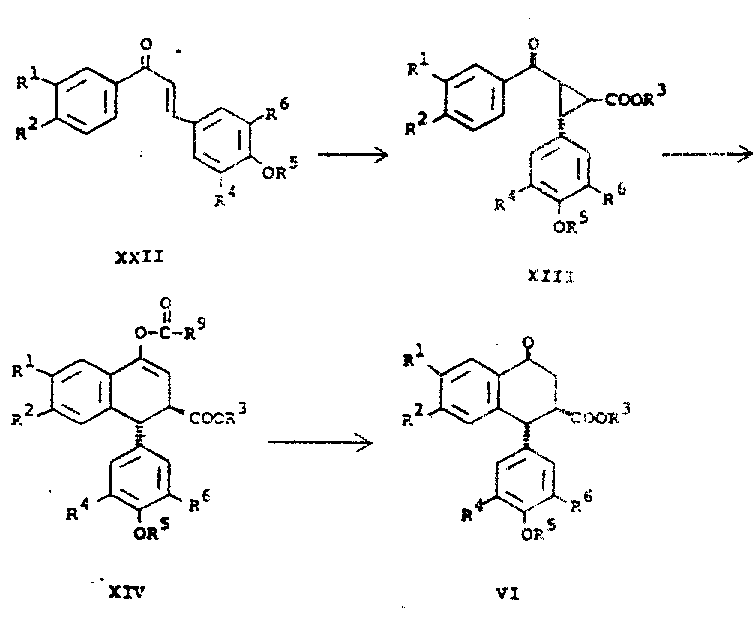
The core innovation lies in the efficient construction of the aryl tetralone system and the subsequent stereocontrolled functionalization via isoxazole intermediates. Unlike historical methods that struggled with low yields and complex mixtures, this approach leverages modern Lewis acid catalysis and dipolar cycloaddition chemistry. For R&D directors evaluating process feasibility, the ability to generate high-purity intermediates without extensive chromatographic purification is a compelling advantage. This patent provides the foundational intellectual property for producing high-purity pharmaceutical intermediates with improved economic viability and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of podophyllotoxin and its epimers has been plagued by inefficiencies that hinder commercial scalability. Seminal work by researchers such as Gensler and Kende described routes requiring upwards of 12 to 13 synthetic steps, resulting in disappointingly low overall yields, often below 5%. A major technical hurdle in these conventional pathways was the reliance on the epimerization of picropodophyllin to access podophyllotoxin, a process that frequently yielded near-equimolar mixtures of diastereomers. Separating these closely related isomers required laborious and costly chromatographic techniques, drastically inflating the cost of goods sold. Furthermore, the preparation of key precursors often involved unstable intermediates or harsh conditions that were difficult to control on a multi-kilogram scale, posing significant risks for supply chain continuity.
The Novel Approach
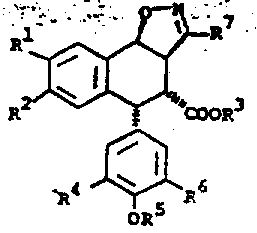
The methodology outlined in CN1020596C fundamentally reimagines the synthetic trajectory by introducing a highly efficient cyclization strategy. Instead of relying on thermodynamic equilibration, the process utilizes a Lewis acid-catalyzed rearrangement of cyclopropyl ketones to directly generate the trans-aryl tetralone scaffold with high fidelity. This is followed by a controlled epimerization to the cis-isomer, setting the stage for precise stereochemical outcomes. The introduction of the isoxazole ring via [3+2] cycloaddition serves as a versatile handle for introducing the necessary nitrogen functionality, which is subsequently transformed into the lactone ring. This strategic use of heterocyclic intermediates allows for better control over regiochemistry and stereochemistry, effectively bypassing the separation nightmares of older methods.
Mechanistic Insights into Lewis Acid Catalyzed Cyclization
The heart of this synthetic advancement is the transformation of cyclopropyl ketones into aryl tetralones, a reaction mediated by strong Lewis acids. The mechanism involves the activation of the carbonyl oxygen by species such as Boron Trifluoride Etherate (BF3·Et2O) or Zinc Chloride (ZnCl2), which increases the electrophilicity of the adjacent carbon centers. In the presence of acid anhydrides like acetic anhydride, the cyclopropane ring undergoes a concerted opening and reclosure, effectively expanding the three-membered ring into the six-membered tetralin system. This reaction is notably sensitive to solvent effects, with nitromethane proving superior to benzene or methylene chloride, likely due to its ability to stabilize polar transition states. The result is a rapid conversion, often completing within hours rather than the days required by previous methodologies, yielding the trans-aryl tetralone in excellent purity.
Following the formation of the tetralone core, the process employs a sophisticated stereoselective reduction and dehydration sequence to establish the cis-alkene geometry required for the subsequent cycloaddition. The reduction of the ketone moiety is performed under selective conditions using hydride sources like sodium borohydride or lithium borohydride, ensuring that ester groups remain intact. Subsequent dehydration under acidic conditions generates the trans-alkene, which is then subjected to base-catalyzed epimerization at low temperatures, typically around -78°C. This low-temperature enolate quenching is critical for locking in the desired cis-configuration, preventing the reversion to the more stable trans-isomer. This precise control over stereochemistry ensures that the downstream [3+2] cycloaddition with nitrile oxides proceeds with high regiospecificity, minimizing the formation of unwanted byproducts.
How to Synthesize Epipodophyllotoxin Intermediates Efficiently
The synthesis of these complex intermediates requires strict adherence to the optimized reaction conditions detailed in the patent to ensure maximum yield and stereochemical integrity. The process begins with the preparation of the cyclopropyl ketone precursor, followed by the critical Lewis acid-mediated cyclization step. Operators must maintain anhydrous conditions and precise temperature controls, particularly during the epimerization and cycloaddition phases, to avoid side reactions. The detailed standardized synthesis steps见下方的指南 provide a comprehensive roadmap for executing this chemistry in a pilot or production environment.
- Prepare cyclopropyl ketone intermediates via cyclopropanation of phenyl styryl ketones using sulfonium ylides.
- Convert cyclopropyl ketones to trans-aryl tetralones using Lewis acids like BF3·Et2O and acid anhydrides in nitromethane.
- Perform stereoselective [3+2] cycloaddition with nitrile oxides to form isoxazole adducts, followed by ring opening and lactonization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the purification workflow. By avoiding the formation of difficult-to-separate diastereomeric mixtures common in older epimerization processes, manufacturers can significantly reduce the consumption of silica gel and solvents associated with chromatography. This reduction in downstream processing directly translates to lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the reagents employed, such as Lewis acids and simple acid anhydrides, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that often plague specialized catalytic processes.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates several redundant steps found in traditional routes, such as the isolation of picropodophyllin and its subsequent conversion. By reducing the total step count and improving the yield of the key cyclization step to over 90% in optimized examples, the overall cost of goods is substantially lowered. The avoidance of expensive transition metal catalysts or biocatalysts further enhances the economic profile, making the production of these high-value intermediates more accessible for generic drug manufacturers seeking cost-effective solutions.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations described ensures consistent batch-to-batch quality, a critical factor for regulatory compliance in API manufacturing. The use of stable intermediates like the isoxazole adducts allows for potential storage and inventory management flexibility, decoupling upstream and downstream production schedules. This resilience against process variability means that suppliers can commit to tighter delivery windows and larger volume contracts without the fear of yield fluctuations that typically accompany complex natural product syntheses.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, with exotherms and gas evolution managed through standard chemical engineering controls. The solvents used, primarily nitromethane and alcohols, are well-understood in industrial settings, facilitating easier permitting and waste treatment compared to exotic halogenated solvents. The high atom economy of the cycloaddition step and the efficient recycling of acid catalysts contribute to a cleaner process profile, helping companies meet increasingly stringent environmental regulations while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating the process for adoption. Understanding these nuances is essential for successful technology transfer and process validation.
Q: How does this patent improve upon traditional Gensler synthesis methods?
A: Traditional methods often require 12-13 steps with low overall yields and difficult separations of picropodophyllin epimers. This patent introduces a streamlined route utilizing Lewis acid-catalyzed cyclization and isoxazole intermediates to achieve higher stereoselectivity and fewer purification steps.
Q: What are the key reagents for the cyclization step?
A: The process utilizes Lewis acids such as Boron Trifluoride Etherate (BF3·Et2O), Zinc Chloride (ZnCl2), or Tin Tetrachloride (SnCl4) in conjunction with acid anhydrides like acetic anhydride, typically in nitromethane solvent.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the avoidance of complex enzymatic resolutions and the use of robust chemical reagents like Lewis acids and nitrile oxides makes the pathway highly amenable to commercial scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epipodophyllotoxin Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for the oncology sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1020596C can be translated into reliable industrial reality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of epipodophyllotoxin intermediate meets the exacting standards required for final API synthesis. Our commitment to technical excellence ensures that your development timelines are met without compromise on quality.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your volume requirements. We encourage you to contact us for specific COA data and route feasibility assessments to determine the best path forward for your project. Let us partner with you to bring life-saving medications to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
