Advanced Synthesis of Oligomerized Thiophene Derivative P1 for High-Performance Organic Electronics
Advanced Synthesis of Oligomerized Thiophene Derivative P1 for High-Performance Organic Electronics
The rapid evolution of the organic electronics sector demands materials that offer precise control over energy levels and exceptional environmental stability. Patent CN108586483B introduces a significant breakthrough in this domain by disclosing an oligomerized thiophene derivative, designated as P1, which utilizes S,S-dioxo dibenzothiophene as a robust electron-accepting unit. This specific molecular architecture is engineered to address the critical need for materials with lower HOMO energy levels and appropriate optical band gaps, making it highly suitable for next-generation optoelectronic applications. The structural complexity of P1, featuring alternating donor and acceptor units, allows for fine-tuning of photoelectric conversion efficiency, a parameter vital for the performance of Organic Light Emitting Diodes (OLEDs) and Organic Solar Cells (OSCs). As a reliable OLED material supplier, understanding the nuanced synthesis of such advanced structures is paramount for ensuring supply chain continuity and product quality.
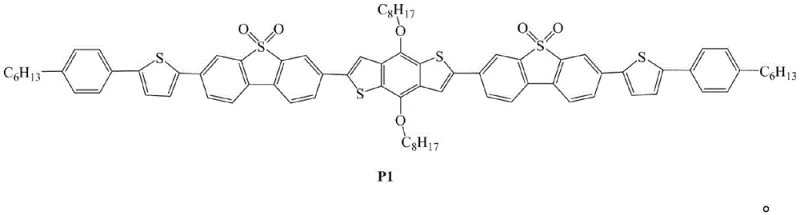
The molecular design presented in the patent leverages the unique properties of the dibenzothiophene-S,S-dioxide (DBTSO) moiety. Unlike traditional dibenzothiophene which often serves as an electron donor, the oxidation of the sulfur atom to its highest valence state transforms it into a potent electron acceptor. This modification not only enhances the affinity for electrons, facilitating better injection and transmission within the device, but also imparts superior oxidation resistance and thermal stability to the final polymer. The integration of this unit into an oligomeric backbone, flanked by thiophene and benzene derivatives with solubilizing alkyl chains, ensures that the material maintains processability while delivering high fluorescence quantum yields. For R&D directors focusing on purity and杂质谱 (impurity profiles), the defined structure offers a clear target for analytical validation and quality control protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance organic semiconductor materials has been plagued by challenges related to structural instability and difficult post-synthetic modifications. Conventional methods often rely on introducing electron-withdrawing groups through harsh oxidation steps after the polymer backbone has been assembled, which can lead to unpredictable side reactions and degradation of the conjugated system. Furthermore, achieving the precise Donor-Acceptor (D-A) alternating structure required for optimal carrier mobility is frequently complicated by poor regioselectivity in standard coupling reactions. These limitations result in materials with inconsistent batch-to-batch performance, broad polydispersity, and suboptimal energy levels that fail to match the work functions of common electrode materials. Such inefficiencies drive up the cost of goods sold and create bottlenecks in the commercial scale-up of complex electronic chemicals, forcing manufacturers to seek more robust synthetic pathways.
The Novel Approach
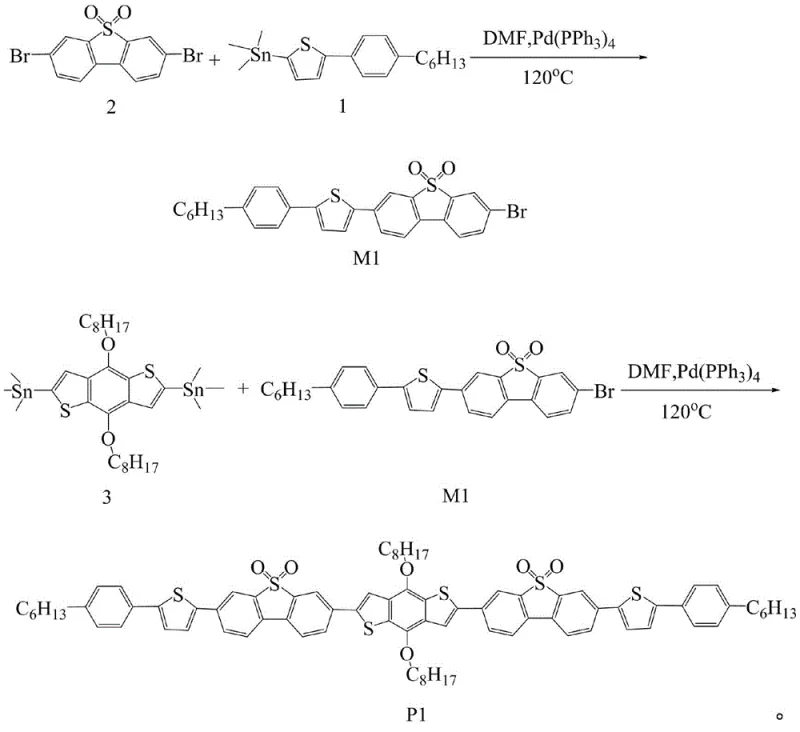
The methodology outlined in patent CN108586483B circumvents these issues by employing a modular Stille cross-coupling strategy that builds the D-A architecture from pre-functionalized, stable precursors. By utilizing 3,7-dibromo-S,S-dioxo-dibenzothiophene as a core building block, the electron-accepting capability is intrinsic to the monomer, eliminating the need for risky post-polymerization oxidation. This approach allows for the precise assembly of the oligomer P1 through sequential coupling reactions, ensuring high structural fidelity and reproducibility. The use of organotin reagents in conjunction with palladium catalysis provides excellent tolerance for various functional groups, including the alkoxy chains necessary for solubility. This novel route significantly simplifies the purification process and enhances the overall yield of the target semiconductor material, representing a substantial advancement in cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Stille Cross-Coupling
The core of this synthesis lies in the palladium-catalyzed Stille cross-coupling reaction, a powerful tool for forming carbon-carbon bonds between organotin compounds and organic halides. In the first stage, the reaction between 2-(trimethyltin)-5-(4-n-hexylphenyl)thiophene and 3,7-dibromo-S,S-dioxo-dibenzothiophene proceeds via a catalytic cycle involving oxidative addition, transmetallation, and reductive elimination. The tetrakis(triphenylphosphine)palladium catalyst facilitates the activation of the carbon-bromine bond on the dibenzothiophene core, allowing for the selective attachment of the thiophene unit. Maintaining an inert nitrogen atmosphere is critical during this phase to prevent the oxidation of the phosphine ligands and the decomposition of the sensitive organotin species, which could otherwise lead to the formation of homocoupling byproducts. The reaction temperature is carefully controlled between 100°C and 120°C to balance reaction kinetics with the thermal stability of the intermediates.
Impurity control is meticulously managed through the stoichiometry of the reactants and the subsequent purification workflow. The patent specifies a molar ratio of 3:1 for the dibromo-species to the tin-reagent in the first step to favor the formation of the mono-substituted intermediate M1, minimizing the formation of the di-substituted side product. Following the reaction, the crude mixture undergoes a rigorous workup involving extraction with dichloromethane and drying over anhydrous magnesium sulfate to remove inorganic salts and polar impurities. The final purification via column chromatography, utilizing a specific eluent system of petroleum ether and dichloromethane (1:2 volume ratio), ensures the removal of residual catalyst, unreacted starting materials, and oligomeric byproducts. This level of detail in the purification protocol is essential for achieving the high-purity OLED material standards required for commercial device fabrication.
How to Synthesize Oligomerized Thiophene Derivative P1 Efficiently
The synthesis of P1 is a sophisticated two-step process that requires strict adherence to anhydrous conditions and precise temperature control to maximize yield and purity. The initial formation of the brominated intermediate M1 sets the foundation for the final coupling, and any deviation in the stoichiometry or reaction time can propagate errors into the final product. Operators must ensure that all solvents, particularly DMF, are thoroughly dried to prevent hydrolysis of the organotin reagents, which would quench the catalytic cycle. The detailed standardized synthetic steps provided below outline the exact parameters for reagent addition, heating profiles, and workup procedures necessary to replicate the results described in the patent documentation successfully.
- Synthesize the intermediate M1 by reacting 2-(trimethyltin)-5-(4-n-hexylphenyl)thiophene with 3,7-dibromo-S,S-dioxo-dibenzothiophene using a palladium catalyst in DMF at 100-120°C.
- Purify the crude intermediate M1 via extraction with dichloromethane and column chromatography using petroleum ether and dichloromethane.
- Perform the final Stille coupling between intermediate M1 and 2,6-bis(trimethyltin)-4,8-bis(2-ethylhexyloxy)benzo[1,2-b: 4,5-b']dithiophene to obtain the final product P1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers distinct logistical and economic benefits over traditional methods. The reliance on stable, commercially available precursors such as dibromo-dibenzothiophene derivatives and stannylated thiophenes reduces the risk of raw material shortages and price volatility. Furthermore, the elimination of hazardous post-synthetic oxidation steps simplifies the manufacturing workflow, reducing the need for specialized corrosion-resistant equipment and lowering the overall operational expenditure. The robustness of the Stille coupling under the specified conditions suggests a high degree of scalability, allowing for seamless transition from laboratory gram-scale synthesis to multi-kilogram production runs without significant re-optimization of the process parameters.
- Cost Reduction in Manufacturing: The strategic use of pre-oxidized DBTSO building blocks eliminates the need for expensive and dangerous oxidizing agents typically required to convert sulfide units to sulfones post-polymerization. This modification not only removes a costly processing step but also reduces the generation of hazardous waste streams associated with strong oxidants, leading to significant savings in waste disposal and environmental compliance costs. Additionally, the high selectivity of the palladium catalyst minimizes the formation of difficult-to-separate isomers, thereby improving the overall mass balance and reducing the loss of valuable intermediates during purification.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on fundamental organic transformations that are well-understood and widely supported by the global chemical supply base. The precursors involved, such as trimethyltin derivatives and brominated aromatics, are commodity chemicals with established supply chains, mitigating the risk of single-source dependency. By standardizing the reaction conditions to common solvents like DMF and standard catalysts like Pd(PPh3)4, manufacturers can easily source alternatives in case of supply disruptions, ensuring continuous production schedules and reliable delivery timelines for downstream electronics clients.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (100-120°C) and atmospheric pressure, which are ideal conditions for scaling up in standard glass-lined or stainless steel reactors without requiring high-pressure autoclaves. The use of nitrogen protection is a standard industrial practice that poses minimal engineering challenges. Moreover, the defined purification protocol using standard chromatography techniques can be adapted to preparative HPLC or recrystallization methods for larger batches, ensuring that the final product meets stringent purity specifications while maintaining a manageable environmental footprint through efficient solvent recovery systems.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of this oligomeric thiophene derivative. These answers are derived directly from the experimental data and technical disclosures within the patent, providing clarity on reaction mechanisms, material properties, and potential industrial applications. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this material into their existing product lines or device architectures.
Q: What is the primary function of the S,S-dioxo dibenzothiophene unit in this material?
A: The S,S-dioxo dibenzothiophene (DBTSO) unit acts as a strong electron-accepting unit within the donor-acceptor (D-A) alternating structure. This configuration effectively improves carrier mobility and provides a lower HOMO energy level (-5.35 eV), which is crucial for efficient electron injection and transmission in organic semiconductor devices.
Q: What are the optimal reaction conditions for the Stille coupling described in the patent?
A: The patent specifies that the Stille cross-coupling reactions should be conducted under an inert nitrogen atmosphere using anhydrous N,N-Dimethylformamide (DMF) as the solvent. The catalyst of choice is tetrakis(triphenylphosphine)palladium, with reaction temperatures maintained between 100°C and 120°C for durations ranging from 12 to 72 hours depending on the specific step.
Q: What are the potential applications for this oligomeric thiophene derivative?
A: Due to its suitable optical band gap of approximately 2.24 eV and excellent thermal stability, this material is specifically designed for application in new materials prepared for Organic Light-Emitting Diodes (OLEDs) and Organic Solar Cell (OSC) devices, where stable charge transport properties are required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oligomerized Thiophene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-performance materials in driving the next generation of organic electronics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like the oligomerized thiophene derivative P1 can be manufactured with consistent quality and reliability. We are committed to meeting stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and electronic properties of every batch. Our expertise in palladium-catalyzed cross-coupling reactions allows us to optimize yields and minimize impurities, delivering a product that is ready for immediate integration into OLED and OSC device fabrication.
We invite you to collaborate with us to explore how this advanced material can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and processing capabilities. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can support your supply chain goals and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
