Advanced Manufacturing of Imatinib Intermediates via Catalyst-Free Nucleophilic Substitution
Introduction to Next-Generation Imatinib Synthesis
The pharmaceutical landscape for kinase inhibitors continues to evolve, driven by the urgent need for more efficient and scalable manufacturing processes. Patent CN101016293A, published in August 2007, introduces a transformative approach to the preparation of Imatinib, a critical tyrosine kinase inhibitor used in the treatment of chronic myeloid leukemia. This technology represents a paradigm shift from transition-metal catalyzed cross-couplings to a robust, base-mediated nucleophilic aromatic substitution strategy. By leveraging simple alkali metal compounds and readily available organic solvents, this method addresses the longstanding bottlenecks of cost, safety, and purification complexity that have plagued earlier synthetic routes. For global supply chain leaders and R&D directors, understanding this methodology is essential for securing a reliable API intermediate supplier capable of delivering high-volume, cost-effective solutions.
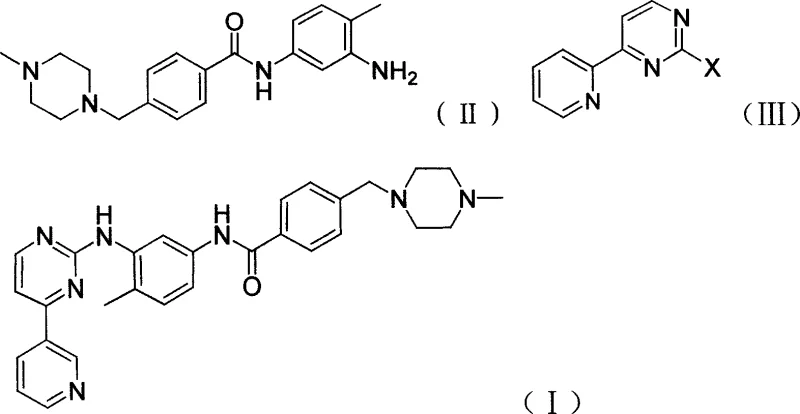
The core innovation lies in the direct coupling of N-(4-methyl-3-amino-phenyl)-4-(4-methyl-piperazine-1-ylmethyl)-benzamide with 2-halo-4-(3-pyridyl)-pyrimidine. Unlike traditional methods that rely on precious metals, this process utilizes fundamental organic chemistry principles to achieve high conversion rates. The elimination of exotic catalysts not only streamlines the reaction workflow but also aligns with modern green chemistry initiatives by reducing heavy metal waste. As we delve deeper into the technical specifics, it becomes evident that this patent offers a viable pathway for the commercial scale-up of complex kinase inhibitors, ensuring consistent quality and supply continuity for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the industrial synthesis of Imatinib was fraught with significant technical and economic challenges. As illustrated in the background art of the patent, earlier methods such as those disclosed in European patent EP 564409 suffered from abysmal yields, often hovering around 20% in the final steps, necessitating extensive recycling or disposal of materials. Furthermore, these legacy routes frequently required column chromatography for purification, a technique that is notoriously difficult to scale and incredibly solvent-intensive, driving up both operational costs and environmental impact. Another prominent method, described in Chinese patent CN1630648A, relied on the use of trimethylaluminum as a condensing agent.
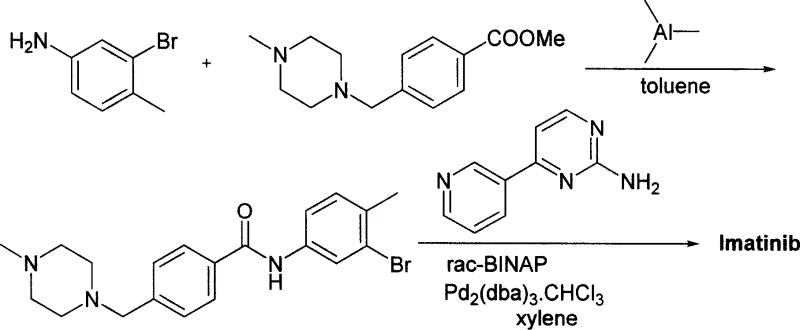
The use of trimethylaluminum presents severe safety hazards due to its pyrophoric nature and violent reaction with water, posing unacceptable risks for large-scale chemical manufacturing. Additionally, the reliance on palladium catalysts combined with expensive ligands like BINAP introduced a substantial cost burden. These transition metal systems often generated isomeric impurities, such as the 10% isomer noted in the prior art, which required complex reversed-phase chromatography to remove. The cumulative effect of these limitations was a fragile supply chain vulnerable to raw material price volatility and regulatory scrutiny regarding heavy metal residues in the final Active Pharmaceutical Ingredient (API).
The Novel Approach
In stark contrast, the method disclosed in CN101016293A simplifies the molecular construction of Imatinib by bypassing the need for transition metal catalysis entirely. The novel approach employs a direct nucleophilic substitution where the amino group of the benzamide derivative attacks the halogenated pyrimidine ring. This reaction is facilitated by strong alkali metal bases, such as sodium hydride, potassium tert-butoxide, or sodium hydroxide, which activate the nucleophile effectively. By shifting to this mechanism, the process eliminates the formation of metal-associated isomers and removes the necessity for chromatographic purification. The result is a cleaner reaction profile that yields high-purity Imatinib through straightforward crystallization or extraction techniques.
This strategic pivot from catalytic coupling to base-mediated substitution fundamentally alters the economic model of production. It replaces rare, expensive metals with commodity chemicals that are globally sourced and price-stable. Moreover, the reaction conditions are flexible, accommodating a wide range of solvents including DMF, toluene, THF, and dioxane, allowing manufacturers to optimize for solubility and recovery rates. This flexibility ensures that the process can be adapted to existing infrastructure without requiring specialized equipment for handling pyrophoric reagents or recovering precious metals, thereby enhancing the overall cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Base-Mediated Nucleophilic Substitution
The chemical elegance of this synthesis lies in its reliance on the inherent reactivity of the heteroaromatic system. The reaction proceeds via a classic Nucleophilic Aromatic Substitution (SnAr) mechanism. In the first stage, the alkali metal base deprotonates the aniline nitrogen of the N-(4-methyl-3-amino-phenyl)-4-(4-methyl-piperazine-1-ylmethyl)-benzamide intermediate. This deprotonation generates a highly nucleophilic anionic species, significantly increasing its electron density and readiness to attack an electrophilic center. The choice of base is critical; stronger bases like sodium hydride or potassium tert-butoxide ensure complete deprotonation, driving the equilibrium towards the formation of the reactive anion.
Subsequently, this activated amine attacks the C-2 position of the 2-halo-4-(3-pyridyl)-pyrimidine. The pyrimidine ring, being electron-deficient due to the nitrogen atoms, is susceptible to nucleophilic attack, particularly at the position ortho to a ring nitrogen and bearing a good leaving group (chlorine or bromine). The addition of the nucleophile forms a transient Meisenheimer-like complex, which then collapses to expel the halide ion, restoring aromaticity and forming the stable C-N bond characteristic of Imatinib. This mechanism is highly selective, minimizing the formation of regioisomers that often plague palladium-catalyzed aminations.
From an impurity control perspective, this mechanism offers distinct advantages. Transition metal catalysis often leads to homocoupling side reactions or incomplete dehalogenation, creating difficult-to-remove impurities. In contrast, the SnAr pathway described here produces primarily inorganic salts (e.g., NaCl, KBr) as byproducts, which are easily removed during the aqueous workup. The absence of metal-ligand complexes simplifies the downstream processing significantly. Furthermore, the reaction temperature can be tuned between 30°C and 150°C depending on the solvent and base selected, providing a thermal window to optimize kinetics without degrading the sensitive piperazine or pyridine moieties. This robustness ensures that the high-purity imatinib specifications are met consistently, batch after batch.
How to Synthesize Imatinib Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and solvent selection to maximize yield and minimize side reactions. The patent outlines a versatile protocol where the molar ratio of the amine substrate to the alkali base ranges from 1:2 to 1:10, ensuring excess base is available to drive the deprotonation to completion. The halopyrimidine is typically added in a slight excess (1 to 5 equivalents) to ensure full consumption of the valuable amine intermediate. The reaction can be performed in polar aprotic solvents like DMF for faster kinetics or in less polar solvents like toluene for easier product isolation. Phase-transfer catalysts, such as crown ethers or quaternary ammonium salts, can be employed to further accelerate the reaction rate, particularly in heterogeneous systems.
- Preparation of the amine precursor: React 3-nitro-4-methyl-aniline with 4-(4-methyl-piperazine-1-ylmethyl) benzoyl chloride, followed by reduction of the nitro group to an amine.
- Base-mediated activation: Dissolve the amino-benzamide intermediate in an organic solvent (e.g., DMF, THF, or Toluene) and treat with an alkali metal compound such as sodium hydride or potassium tert-butoxide.
- Nucleophilic coupling: Add 2-halo-4-(3-pyridyl)-pyrimidine to the reaction mixture at elevated temperatures (30-150°C) to form the final imatinib structure without requiring palladium catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond simple unit cost savings. The primary advantage is the decoupling of production from the volatile market of precious metals. Palladium prices are subject to significant geopolitical and mining supply fluctuations; by eliminating the need for palladium catalysts and phosphine ligands, manufacturers insulate their cost structure from these external shocks. Additionally, the removal of pyrophoric reagents like trimethylaluminum reduces the need for specialized storage facilities and hazardous material handling protocols, lowering insurance premiums and facility maintenance costs associated with high-risk chemical operations.
- Cost Reduction in Manufacturing: The elimination of expensive catalytic systems and chromatographic purification steps results in substantial operational savings. Without the need for column chromatography, solvent consumption is drastically reduced, and batch cycle times are shortened, leading to higher throughput per reactor volume. The use of commodity bases and solvents further drives down the Bill of Materials (BOM) cost, making the final API more competitive in generic markets while maintaining healthy margins for innovators.
- Enhanced Supply Chain Reliability: Sourcing alkali metal bases and simple halopyrimidines is far more reliable than sourcing specialized catalysts which may have limited suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Furthermore, the simplified purification process reduces the dependency on specialized resin or silica gel supplies, ensuring that production schedules remain uninterrupted even during broader supply chain disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram to multi-ton production without the engineering complexities of handling air-sensitive catalysts. From an environmental standpoint, the reduction in heavy metal waste simplifies effluent treatment and regulatory compliance. The ability to produce high-purity material without chromatography significantly lowers the E-factor (mass of waste per mass of product), aligning with corporate sustainability goals and reducing waste disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in CN101016293A, offering clarity on how this technology outperforms legacy methods in terms of safety, efficiency, and purity. Understanding these nuances is critical for technical teams evaluating process transfers or capacity expansions.
Q: How does this method improve upon traditional palladium-catalyzed routes?
A: This method eliminates the need for expensive palladium catalysts and ligands (like BINAP), which significantly reduces raw material costs and removes the complex step of heavy metal residue removal, thereby simplifying the purification process.
Q: What are the safety advantages regarding reagents used?
A: Unlike prior art methods that utilize pyrophoric and dangerous reagents like trimethylaluminum, this process relies on stable alkali metal bases (e.g., NaH, t-BuOK) and common organic solvents, drastically improving operational safety for large-scale production.
Q: Does this synthesis route require column chromatography?
A: No, one of the key benefits of this invention is that it yields high-purity imatinib directly through crystallization or standard extraction, avoiding the time-consuming and solvent-intensive column chromatography purification required by older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the modern pharmaceutical supply chain. Our technical team has extensively analyzed the methodology presented in CN101016293A and possesses the expertise to implement this catalyst-free nucleophilic substitution at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Imatinib intermediate meets the highest global regulatory standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and economically viable chemistry available.
