Advanced Manufacturing of High-Purity Fluorophenol via Optimized Grignard Exchange Technology
Introduction to Next-Generation Fluorophenol Synthesis
The global demand for high-purity fluorinated aromatics has surged, driven by their critical role as building blocks in advanced pharmaceutical intermediates and electronic chemical materials. Patent CN101445431B introduces a transformative methodology for the preparation of fluorophenol, specifically targeting the efficient synthesis of trifluoromethyl phenol derivatives. This technology represents a significant departure from legacy manufacturing protocols, addressing long-standing challenges related to environmental impact, operational safety, and final product quality. By leveraging a sophisticated Grignard exchange mechanism, the process achieves exceptional control over regioselectivity and impurity profiles. For R&D directors and procurement specialists seeking a reliable fluorophenol supplier, understanding the nuances of this patented route is essential for securing a competitive advantage in the supply chain. The following analysis dissects the technical merits and commercial viability of this innovation, highlighting its potential to redefine standards in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of trifluoromethyl phenol has relied heavily on classical diazotization pathways or harsh nucleophilic substitution reactions. Traditional methods often necessitate the use of 1-halo-3,4,5-trifluorobenzene as a starting material, subjecting it to rigorous ammonification followed by diazotization and hydrolysis. These processes are fraught with significant drawbacks, including the requirement for high temperature and high pressure (HTHP) conditions which pose substantial safety risks in a plant environment. Furthermore, the reliance on copper-based catalysts in these older routes introduces heavy metal contamination issues, complicating downstream purification and waste treatment. The generation of excessive wastewater and the use of stoichiometric amounts of hazardous reagents render these conventional methods increasingly untenable in the face of modern environmental regulations. Additionally, the selectivity of these reactions is often poor, leading to complex mixtures that require energy-intensive separation techniques, thereby inflating the overall cost of production and reducing the effective yield of the desired API intermediate.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a mild and highly efficient Grignard exchange strategy that circumvents the pitfalls of traditional synthesis. This method initiates with fluorobromobenzene, reacting it with a specific alkyl magnesium chloride to generate a highly reactive aryl magnesium chloride intermediate under controlled, low-temperature conditions. This intermediate is then seamlessly converted into a boronic acid ester through reaction with a borate ester, followed by a gentle acidic hydrolysis to yield the corresponding boronic acid. The final transformation involves a clean oxidation step to produce the target fluorophenol. This pathway is characterized by its operational simplicity and compatibility with standard reactor setups, eliminating the need for specialized high-pressure equipment. 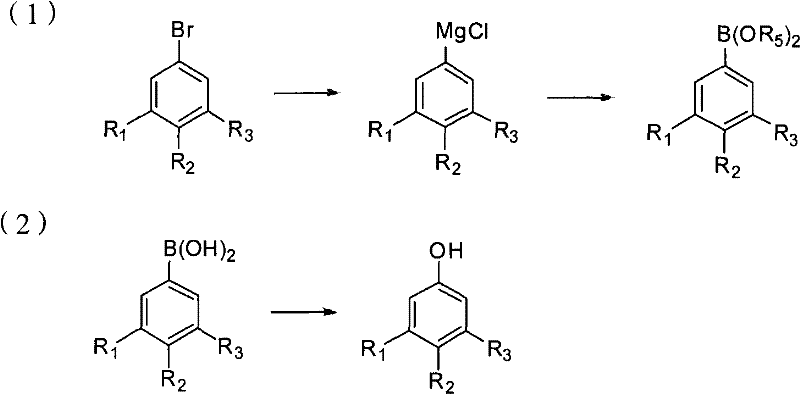 As illustrated in the reaction scheme, the process flows logically from halogen exchange to functionalization, ensuring a streamlined workflow that minimizes unit operations. For manufacturers focused on cost reduction in electronic chemical manufacturing, this route offers a compelling alternative that balances high performance with operational efficiency.
As illustrated in the reaction scheme, the process flows logically from halogen exchange to functionalization, ensuring a streamlined workflow that minimizes unit operations. For manufacturers focused on cost reduction in electronic chemical manufacturing, this route offers a compelling alternative that balances high performance with operational efficiency.
Mechanistic Insights into Grignard Exchange and Boration
The core of this technological breakthrough lies in the precise execution of the Grignard exchange reaction, which serves as the foundation for the entire synthetic sequence. Unlike direct magnesium insertion which can be erratic and prone to side reactions, the exchange reaction using reagents such as isopropyl magnesium chloride (i-PrMgCl) offers superior kinetic control. This selectivity is crucial when dealing with poly-fluorinated substrates where multiple reactive sites might exist. The exchange proceeds rapidly at temperatures ranging from -50°C to 50°C, preferably between -20°C and 10°C, ensuring that the sensitive carbon-fluorine bonds remain intact while the bromine atom is selectively replaced by the magnesium species. 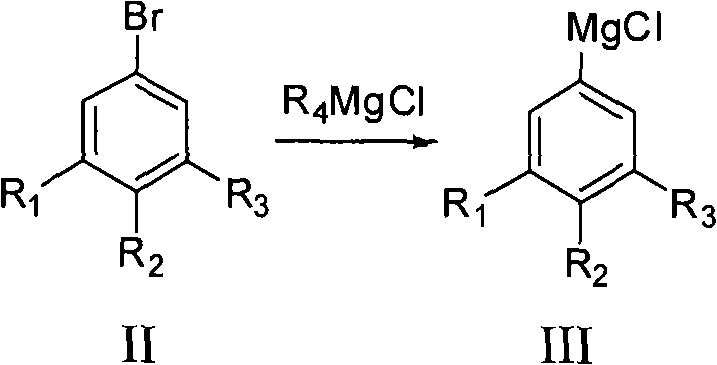 This step generates the key organometallic intermediate (Formula III) with high fidelity, setting the stage for the subsequent functionalization. The use of ethereal solvents like THF or glycol dimethyl ether further stabilizes the Grignard species, preventing decomposition and ensuring a homogeneous reaction mixture that is ideal for scale-up.
This step generates the key organometallic intermediate (Formula III) with high fidelity, setting the stage for the subsequent functionalization. The use of ethereal solvents like THF or glycol dimethyl ether further stabilizes the Grignard species, preventing decomposition and ensuring a homogeneous reaction mixture that is ideal for scale-up.
Following the formation of the Grignard reagent, the process moves to the boration step, where the organomagnesium species reacts with a trialkyl borate, such as trimethyl borate. This nucleophilic attack on the boron center is highly efficient, typically proceeding with a molar ratio of substrate to borate between 1:0.8 and 1:10. 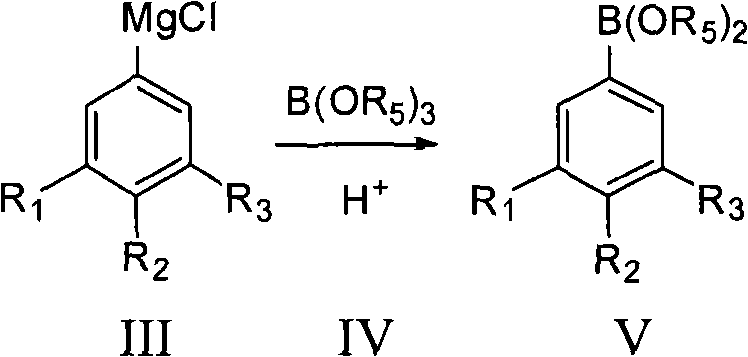 The resulting boronic acid ester (Formula V) is then subjected to acidic hydrolysis, a critical step that converts the ester into the free boronic acid (Formula VI). The patent specifies the use of mild acids like hydrochloric acid or acetic acid at concentrations of 5% to 100%, allowing for fine-tuning of the hydrolysis rate to prevent side reactions. This mechanistic pathway ensures that the boron functionality is installed with precision, creating a robust precursor for the final oxidative conversion to the phenol. The ability to control each step with such granularity allows for the production of intermediates with purity levels exceeding 99.8%, a metric that is vital for downstream pharmaceutical applications.
The resulting boronic acid ester (Formula V) is then subjected to acidic hydrolysis, a critical step that converts the ester into the free boronic acid (Formula VI). The patent specifies the use of mild acids like hydrochloric acid or acetic acid at concentrations of 5% to 100%, allowing for fine-tuning of the hydrolysis rate to prevent side reactions. This mechanistic pathway ensures that the boron functionality is installed with precision, creating a robust precursor for the final oxidative conversion to the phenol. The ability to control each step with such granularity allows for the production of intermediates with purity levels exceeding 99.8%, a metric that is vital for downstream pharmaceutical applications.
How to Synthesize Fluorophenol Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and reagent stoichiometry, to maximize yield and minimize impurity formation. The process begins with the preparation of the Grignard exchange reagent, followed by its addition to the fluorobromobenzene substrate in an inert atmosphere. Detailed operational protocols emphasize the importance of maintaining the reaction temperature within the optimal window to prevent thermal runaway or incomplete conversion. Following the boration and hydrolysis steps, the crude boronic acid is isolated and subjected to oxidation using hydrogen peroxide, a green oxidant that produces water as the only byproduct. The detailed standardized synthesis steps for this high-efficiency pathway are outlined below, providing a clear roadmap for technical teams aiming to replicate these results in a pilot or production setting.
- Perform a Grignard exchange reaction between fluorobromobenzene and an alkyl magnesium chloride (such as i-PrMgCl) in an organic solvent like THF at low temperatures to generate the intermediate aryl magnesium chloride.
- React the resulting Grignard reagent with a borate ester (e.g., trimethyl borate) to form the fluorobenzene boronic acid ester, followed by acidic hydrolysis to obtain the fluorobenzene boronic acid.
- Oxidize the fluorobenzene boronic acid using hydrogen peroxide under controlled conditions, followed by extraction and distillation to isolate the final high-purity fluorophenol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Grignard-based synthesis route offers profound benefits for procurement managers and supply chain heads tasked with optimizing costs and ensuring continuity. The shift away from high-pressure ammonification and copper-catalyzed diazotization eliminates the need for expensive, corrosion-resistant reactors and complex waste treatment systems associated with heavy metal removal. This simplification of the infrastructure requirements translates directly into lower capital expenditure (CAPEX) and reduced operational expenditure (OPEX) for manufacturing facilities. Furthermore, the use of readily available starting materials like fluorobromobenzene and common solvents such as THF ensures a stable supply chain that is less susceptible to the volatility often seen with specialized reagents. The robustness of the process also means that production schedules can be maintained with greater reliability, reducing the risk of delays that could impact downstream API synthesis.
- Cost Reduction in Manufacturing: The elimination of costly phase-transfer catalysts and the reduction in solvent usage significantly lower the raw material costs per kilogram of finished product. By avoiding the generation of large volumes of saline wastewater typical of diazotization processes, facilities can also realize substantial savings in environmental compliance and waste disposal fees. The high yield reported in the embodiments, approaching quantitative conversion in the oxidation step, ensures that raw material utilization is maximized, further driving down the unit cost of production for high-purity fluorophenol.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions makes this process highly resilient to supply shocks. Unlike methods requiring bespoke catalysts or extreme conditions that limit the number of qualified contract manufacturers, this route can be executed by a broader range of fine chemical producers. This diversification of potential supply sources enhances security of supply for critical intermediates, ensuring that pharmaceutical and electronic material manufacturers can maintain consistent inventory levels without fear of interruption.
- Scalability and Environmental Compliance: The mild reaction conditions, operating primarily between -20°C and 50°C, are inherently safer and easier to scale from laboratory to multi-ton production than high-temperature alternatives. The process aligns with green chemistry principles by utilizing hydrogen peroxide as an oxidant and minimizing the use of toxic heavy metals, thereby simplifying regulatory approvals and reducing the environmental footprint of the manufacturing site. This sustainability profile is increasingly becoming a key differentiator for suppliers serving eco-conscious global corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorophenol synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply chains or R&D pipelines. The answers reflect the consensus on best practices for handling Grignard reagents and managing the oxidation steps to ensure product quality.
Q: What are the primary advantages of the Grignard exchange method over traditional diazotization for fluorophenol production?
A: The Grignard exchange method described in patent CN101445431B offers significantly milder reaction conditions compared to the high temperature and high pressure (HTHP) required for traditional ammonification and diazotization. It eliminates the need for toxic copper catalysts and reduces the generation of hazardous wastewater, aligning better with green chemistry principles while achieving higher product purity (>99.9%).
Q: How does this synthesis route impact the impurity profile of the final API intermediate?
A: By utilizing a specific Grignard exchange with reagents like i-PrMgCl, the process minimizes side reactions such as Wurtz coupling or halogen-metal exchange at unwanted positions. The subsequent boration and mild acidic hydrolysis steps further refine the intermediate, resulting in a crude product with exceptionally low foreign matter content before the final oxidation and distillation steps.
Q: Is this process suitable for large-scale commercial manufacturing of electronic chemicals?
A: Yes, the process is highly scalable due to its use of common organic solvents like THF and manageable temperature ranges (-20°C to 50°C). The avoidance of extreme pressures and the simplification of post-treatment procedures, such as straightforward extraction and distillation, make it economically viable and operationally safe for ton-scale production required in the electronic and pharmaceutical sectors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation pharmaceuticals and electronic materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex fluorination chemistry positions us as a strategic partner for companies seeking to secure a reliable fluorophenol supplier who can guarantee both quality and volume.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this greener, more efficient manufacturing process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to excellence and our ability to support your long-term supply chain goals with precision and reliability.
