Optimizing Beta-2 Agonist Production: A Technical Deep Dive into Streamlined Phenylethanolamine Synthesis
Optimizing Beta-2 Agonist Production: A Technical Deep Dive into Streamlined Phenylethanolamine Synthesis
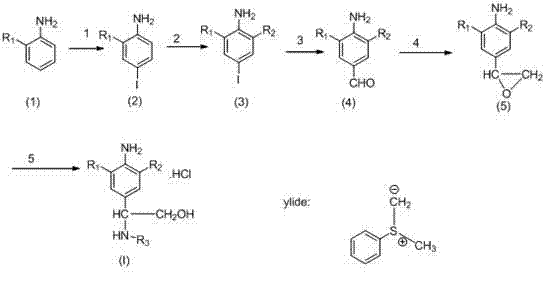
The pharmaceutical industry's relentless pursuit of more efficient synthetic routes for respiratory therapeutics has led to significant advancements in the preparation of phenylethanolamine compounds. As detailed in the Chinese patent CN102477000A, filed in May 2012, a novel methodology has been established that drastically reduces the complexity of synthesizing beta-2 receptor agonists, which are critical for treating asthma and chronic obstructive pulmonary disease (COPD). This technical insight report analyzes the breakthrough 5-step synthesis pathway, contrasting it with legacy methods to highlight its value for R&D directors seeking higher purity and procurement managers aiming for cost optimization. By leveraging palladium-catalyzed carbonylation and selective ylide chemistry, this process offers a robust framework for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations disclosed in CN102477000A, the industry standard for producing these vital beta-2 agonist intermediates relied on a cumbersome and inefficient synthetic strategy, often exemplified by the route described in Chinese patent 01128234.7. This legacy approach necessitates a staggering ten distinct reaction steps to reach the final active pharmaceutical ingredient (API) precursor. Such excessive step count inherently amplifies the risk of yield erosion at every stage, leading to a significantly lower overall throughput. Furthermore, the conventional route employs potassium borohydride (KBH4) for the reduction of ketone intermediates, a step that is notoriously difficult to control regarding stereoselectivity and impurity profiles. The generation of unwanted isomers and side products during this reduction phase not only complicates downstream purification but also results in a dismal final yield of merely 20-30% for the critical ring-opening step. For supply chain heads, this translates to higher waste generation, increased solvent consumption, and unpredictable delivery timelines due to the fragility of the long process chain.
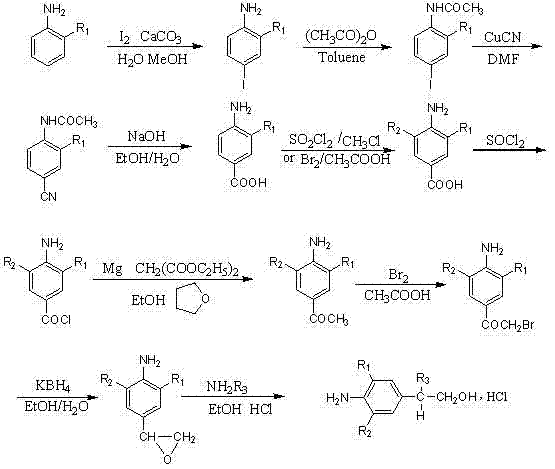
The Novel Approach
In stark contrast, the methodology presented in CN102477000A introduces a highly convergent 5-step synthesis that fundamentally reimagines the construction of the phenylethanolamine core. By strategically utilizing regioselective iodination followed by a palladium-catalyzed reductive carbonylation, the new route bypasses the need for multiple protection and deprotection cycles inherent in the older method. The process begins with the efficient conversion of commercially available substituted anilines into 4-iodo intermediates with yields exceeding 90%, establishing a high-yield foundation immediately. Subsequent halogenation and formylation steps are optimized to maintain high purity, while the introduction of a sulfur ylide-mediated epoxidation replaces the problematic borohydride reduction. This modern approach not only halves the number of unit operations but also significantly enhances the atom economy of the process. For a reliable phenylethanolamine supplier, this streamlined pathway represents a paradigm shift towards lean manufacturing, enabling faster time-to-market and reduced operational expenditures without compromising on the structural integrity of the final molecule.
Mechanistic Insights into Pd-Catalyzed Reductive Carbonylation and Ylide Epoxidation
The cornerstone of this improved synthetic strategy lies in the third step: the palladium-catalyzed reductive carbonylation of the aryl iodide to form the benzaldehyde derivative. This transformation utilizes a catalytic system comprising [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride (Pd(dppf)Cl2) in conjunction with triethylsilane as the reducing agent. Mechanistically, the palladium catalyst undergoes oxidative addition with the aryl iodide bond, followed by the insertion of carbon monoxide to form an acyl-palladium complex. The triethylsilane then acts as a hydride source, facilitating the reductive elimination to release the aldehyde product while regenerating the active palladium species. This reaction is typically conducted in DMF at temperatures ranging from 60-120°C under a saturated carbon monoxide atmosphere. The choice of triethylsilane over traditional hydrogen gas offers superior safety profiles and milder reaction conditions, which is crucial for preserving sensitive functional groups like the amino and trifluoromethyl moieties present on the aromatic ring. This step consistently achieves yields between 69% and 78%, demonstrating remarkable robustness across different substrate variations.
Following the formation of the aldehyde, the synthesis proceeds through a sophisticated epoxidation utilizing a sulfur ylide precursor. This involves the in situ generation of a sulfonium salt from iodoacetic acid, sulfide anisole, and silver trifluoromethanesulfonate, which is subsequently treated with cesium carbonate in THF. The resulting ylide attacks the carbonyl carbon of the benzaldehyde, forming the epoxide ring with high regioselectivity. This method is superior to traditional epoxidation techniques as it avoids the use of harsh oxidants that could degrade the electron-rich aniline core. The final ring-opening of the epoxide with various amines (R3-NH2) in refluxing ethanol proceeds via an SN2 mechanism, ensuring the correct stereochemical orientation required for beta-2 receptor binding. The entire sequence is designed to minimize impurity formation, with the final crystallization steps effectively removing trace metals and organic byproducts, thereby meeting the stringent purity specifications required for clinical-grade intermediates.
How to Synthesize Phenylethanolamine Derivatives Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity. The process initiates with the iodination of the starting aniline using sodium iodide and N-chlorosuccinimide (NCS) in glacial acetic acid at 50°C, a step that must be monitored closely to prevent over-halogenation. Following isolation, the intermediate undergoes electrophilic substitution with either sulfuryl chloride or bromine to install the second halogen atom ortho to the amino group. The subsequent carbonylation step demands a pressure-rated reactor capable of maintaining a CO-saturated environment, while the final amine coupling requires anhydrous conditions to prevent hydrolysis of the epoxide. Detailed standard operating procedures (SOPs) regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility. For comprehensive laboratory instructions and safety data sheets regarding these specific transformations, please refer to the standardized guide below.
- Regioselective iodination of substituted aniline using NaI and NCS in acetic acid at 50°C to generate the 4-iodo intermediate.
- Electrophilic halogenation at the ortho-position using SO2Cl2 or Br2 to introduce the second halogen substituent.
- Palladium-catalyzed reductive carbonylation using CO and triethylsilane to convert the aryl iodide into the corresponding benzaldehyde.
- Epoxidation of the aldehyde using a sulfur ylide precursor generated in situ from iodoacetic acid and sulfide anisole.
- Ring-opening of the epoxide with the desired amine (R3-NH2) in refluxing ethanol to yield the final phenylethanolamine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the CN102477000A synthesis route offers profound advantages for procurement managers and supply chain directors tasked with securing high-purity pharmaceutical intermediates. The most immediate benefit is the drastic reduction in process complexity, which directly correlates to lower manufacturing costs. By eliminating five reaction steps compared to the conventional method, the new route significantly reduces the consumption of solvents, reagents, and energy. Furthermore, the avoidance of expensive and hazardous reducing agents like potassium borohydride in favor of more benign amine ring-opening reactions simplifies waste treatment protocols and lowers environmental compliance costs. This efficiency gain allows for a more competitive pricing structure, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing without sacrificing quality.
- Cost Reduction in Manufacturing: The streamlined 5-step process inherently lowers the cost of goods sold (COGS) by minimizing material throughput and labor hours. The high yields observed in the initial iodination step (over 90%) ensure that expensive starting materials are utilized efficiently, reducing the financial impact of raw material price fluctuations. Additionally, the use of catalytic amounts of palladium, which can potentially be recovered and recycled, further optimizes the cost profile. The elimination of multiple purification stages between ten steps also reduces the loss of product, ensuring that a higher percentage of the input mass is converted into saleable output, thereby maximizing return on investment for large-scale production campaigns.
- Enhanced Supply Chain Reliability: A shorter synthetic route inherently reduces the risk of supply chain disruptions. With fewer unit operations, there are fewer points of failure where a batch could be rejected due to out-of-specification results. The reliance on commercially available starting materials, such as substituted anilines sourced from established chemical suppliers like Shanghai Jiachen Chemical Co., Ltd., ensures a stable upstream supply. This stability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand surges for asthma medications. The robustness of the chemistry also means that technology transfer to different manufacturing sites can be accomplished with greater speed and confidence.
- Scalability and Environmental Compliance: The chemistry described is well-suited for commercial scale-up of complex pharmaceutical intermediates, transitioning smoothly from kilogram-scale R&D batches to multi-ton annual production. The reaction conditions, such as the use of ethanol and THF, are compatible with standard stainless steel reactors found in most cGMP facilities. Moreover, the process generates less hazardous waste compared to the borohydride-heavy legacy route, aligning with modern green chemistry principles. This environmental compatibility simplifies regulatory filings and reduces the burden on wastewater treatment facilities, ensuring long-term operational sustainability and compliance with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic pathway. These answers are derived directly from the experimental data and claims within patent CN102477000A, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement teams assessing vendor capabilities. The data reflects the specific conditions optimized for substrates containing trifluoromethyl and halogen substituents, which are prevalent in modern beta-2 agonist drug candidates.
Q: How does the new 5-step route compare to the conventional 10-step synthesis in terms of yield?
A: The conventional method described in prior art requires ten reaction steps and suffers from low overall yields, particularly in the final reduction stage where yields drop to 20-30%. In contrast, the novel 5-step route utilizes a direct palladium-catalyzed formylation and a selective ylide epoxidation, significantly improving step economy and minimizing cumulative yield loss.
Q: What are the critical reaction conditions for the palladium-catalyzed formylation step?
A: The formylation step converts the aryl iodide to a benzaldehyde using carbon monoxide and triethylsilane. Critical parameters include the use of [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride as the catalyst, DMF as the solvent, and maintaining a temperature between 60-120°C under a saturated CO atmosphere for 18-20 hours to ensure complete conversion.
Q: Is this synthesis scalable for commercial production of asthma medication intermediates?
A: Yes, the route is designed for scalability. It avoids hazardous reagents like potassium borohydride in the final steps, replacing them with safer amine ring-opening reactions. The use of commercially available starting materials like substituted anilines and standard solvents like ethanol and THF facilitates easy scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylethanolamine Compound Supplier
The technical potential of the CN102477000A synthesis route is immense, offering a clear pathway to high-quality beta-2 agonist intermediates that meet the rigorous demands of the global pharmaceutical market. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless. Our facility is equipped with state-of-the-art rigorous QC labs and analytical instrumentation capable of verifying stringent purity specifications, including residual solvent analysis and heavy metal testing. We understand that consistency is key in API intermediate supply, and our process engineering team is dedicated to maintaining batch-to-batch reproducibility through advanced process control systems.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot batches and conduct detailed route feasibility assessments to demonstrate how our optimized phenylethanolamine synthesis can enhance your supply chain resilience. Let us be your partner in delivering life-saving respiratory therapies to patients worldwide through superior chemical manufacturing excellence.
