Optimizing Phenylethanolamine Production: A Technical Breakthrough in Patent CN1237574A
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing critical bioactive scaffolds, particularly phenylethanolamine derivatives which serve as the backbone for numerous beta-adrenergic agonists and cardiovascular therapeutics. Patent CN1237574A introduces a transformative two-step precipitation method that fundamentally alters the economic and operational landscape of producing these valuable intermediates. By bypassing the traditionally cumbersome protection and deprotection sequences, this technology offers a direct route to high-purity phenylethanolamine hydrochlorides. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes for drugs like Denopamine and Deterenol. The core innovation lies in the clever utilization of triethylamine not just as a base, but as a precipitating agent for the byproduct, simplifying the workup significantly. As a reliable phenylethanolamine supplier, understanding the nuances of such patented methodologies is crucial for maintaining a competitive edge in the global supply chain.
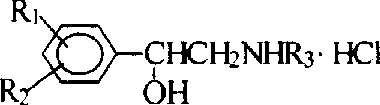
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylethanolamine compounds has been plagued by inefficiencies that drive up costs and extend lead times, creating bottlenecks for procurement managers. Conventional routes typically necessitate the protection of sensitive functional groups, such as phenolic hydroxyls, to prevent unwanted side reactions during the condensation with omega-bromoacetophenone. Furthermore, to suppress dialkylation side reactions on primary amines, manufacturers are often forced to employ a large excess of the amine starting material, sometimes up to three or four equivalents. This wasteful practice not only inflates raw material costs but also complicates downstream purification, requiring extensive chromatography or recrystallization to remove unreacted amines and byproducts. Additionally, traditional reduction steps often rely on expensive hydride reagents like sodium borohydride, which generate significant chemical waste and pose safety hazards on a large scale. These cumulative inefficiencies render many legacy processes unsuitable for modern, cost-sensitive industrial production.
The Novel Approach
In stark contrast, the methodology outlined in CN1237574A presents a streamlined, two-step protocol that elegantly circumvents these historical hurdles. The novel approach employs a direct nucleophilic substitution between the amine and omega-bromoacetophenone in the presence of triethylamine, eliminating the need for protective group chemistry entirely. A key feature of this process is the in-situ precipitation of triethylamine hydrobromide, which can be simply filtered off, leaving the desired intermediate in the solution. This allows for the use of stoichiometric amounts of amine (1:1 molar ratio), drastically reducing raw material consumption compared to traditional excess methods. The subsequent reduction is achieved via catalytic hydrogenation using standard catalysts like Pd/C or Raney Ni under normal pressure, replacing hazardous hydride reagents with clean hydrogen gas. This shift not only enhances safety but also aligns with green chemistry principles, offering substantial cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Triethylamine-Mediated Alkylation and Hydrogenation
From a mechanistic perspective, the success of this synthesis relies on the precise control of nucleophilic substitution and the thermodynamics of salt precipitation. In the first step, the amine acts as a nucleophile attacking the alpha-carbon of the omega-bromoacetophenone. The presence of triethylamine serves a dual purpose: it neutralizes the hydrogen bromide generated during the reaction, driving the equilibrium forward, and simultaneously forms triethylamine hydrobromide. Due to the low solubility of this salt in tetrahydrofuran (THF), it precipitates out of the reaction mixture, effectively removing the byproduct and preventing it from interfering with the product or reversing the reaction. This precipitation-driven equilibrium shift is a masterstroke of process chemistry, allowing the reaction to proceed to completion with high conversion rates even at moderate temperatures ranging from -10°C to 50°C. The resulting intermediate, a keto-amine hydrochloride, is then isolated via acidification and filtration, ensuring high purity before the reduction step.
The second phase involves the stereoselective or non-selective reduction of the ketone moiety to a secondary alcohol, depending on the specific catalyst and conditions employed. The patent specifies the use of palladium on activated carbon (5% Pd/C) or Raney Nickel in alcoholic solvents like ethanol or methanol. Under mild heating and normal hydrogen pressure, the catalyst facilitates the addition of hydrogen across the carbonyl double bond. This catalytic hydrogenation is highly chemoselective, reducing the ketone without affecting the aromatic rings or the amine functionality, which is critical for maintaining the structural integrity of complex drug molecules. For high-purity phenylethanolamine production, this step is vital as it avoids the formation of over-reduced byproducts often seen with stronger chemical reducing agents. The final product is obtained as a stable hydrochloride salt after solvent removal, ready for formulation or further chiral resolution if necessary.
![Chemical structure of Denopamine intermediate 1-(4-hydroxyphenyl)-2-[2-(3,4-dimethoxyphenyl)ethylamino]ethanol hydrochloride](/insights/img/phenylethanolamine-synthesis-patent-pharma-supplier-20260308221441-02.png)
How to Synthesize Phenylethanolamine Compounds Efficiently
Implementing this synthesis requires careful attention to solvent quality and temperature control to maximize the efficiency of the precipitation steps. The process begins with the dissolution of reactants in anhydrous THF, followed by controlled addition to manage exotherms. Detailed standard operating procedures regarding filtration rates and hydrogenation safety protocols are essential for safe scale-up. The following guide outlines the critical operational parameters derived from the patent examples to ensure reproducible results.
- Dissolve amine, omega-bromoacetophenone, and triethylamine (molar ratio 1: 1:1-2) in tetrahydrofuran (THF) at -10°C to 50°C to precipitate triethylamine bromide.
- Filter the reaction mixture to remove the precipitate, then acidify the filtrate with hydrogen chloride in ethanol to precipitate the intermediate keto-amine hydrochloride.
- Hydrogenate the purified intermediate using Pd/C or Raney Ni catalyst in ethanol under normal pressure and mild heat to obtain the final phenylethanolamine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this patented methodology translates directly into tangible operational benefits and risk mitigation. By eliminating multiple synthetic steps associated with protection and deprotection, the overall cycle time for production is significantly compressed, enhancing the responsiveness of the supply chain to market demands. The ability to use stoichiometric amounts of expensive amine starting materials, rather than large excesses, results in substantial raw material savings, directly impacting the cost of goods sold (COGS). Furthermore, the reliance on catalytic hydrogenation instead of stoichiometric metal hydrides reduces the burden on waste treatment facilities and lowers the cost of hazardous waste disposal. These factors combined create a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of protecting group reagents and the reduction in amine usage from multi-equivalent excess to a 1:1 molar ratio dramatically lowers material costs. Additionally, replacing expensive reducing agents like sodium borohydride with hydrogen gas and reusable catalysts further drives down operational expenses. The simplified workup involving filtration rather than complex extractions or chromatography reduces labor and solvent consumption, contributing to a leaner manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of the precipitation-based purification method ensures consistent product quality batch after batch, reducing the risk of supply disruptions caused by failed purification steps. The use of common, commercially available solvents like THF and ethanol minimizes the risk of raw material shortages. Moreover, the shorter synthetic route reduces the number of potential failure points in the process, thereby increasing the overall reliability of delivery schedules for high-purity phenylethanolamine compounds.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful synthesis of various derivatives in the patent examples. The generation of solid byproducts (triethylamine hydrobromide) that can be easily filtered and potentially recycled or disposed of safely simplifies waste management. The avoidance of heavy metal waste from hydride reductions and the use of catalytic hydrogenation align with increasingly stringent environmental regulations, ensuring long-term compliance and sustainability for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide clarity on feasibility and performance.
Q: How does this method improve upon traditional phenylethanolamine synthesis?
A: Traditional methods often require protecting sensitive groups like phenolic hydroxyls and using excess amines to prevent dialkylation, leading to long steps and waste. This patent utilizes a direct alkylation strategy with triethylamine as a scavenger, eliminating protection steps and allowing for stoichiometric amine usage.
Q: What catalysts are suitable for the reduction step in this process?
A: The patent specifies the use of palladium on activated carbon (Pd/C) or Raney Nickel as effective catalysts for the hydrogenation step, operating under normal pressure and mild heating conditions to reduce the ketone group to the corresponding alcohol.
Q: Is this process scalable for industrial production of cardiac drug intermediates?
A: Yes, the process is highly scalable due to its reliance on simple precipitation for purification rather than complex chromatography. The use of common solvents like THF and ethanol, along with standard hydrogenation equipment, facilitates easy transition from lab to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylethanolamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like CN1237574A in delivering high-quality intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised in the lab are fully realized in our manufacturing plants. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenylethanolamine compound meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to technical excellence allows us to navigate complex chemistries with precision and reliability.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall manufacturing costs.
