Advanced Imatinib Purification: Achieving Ultra-Low Genotoxic Impurities for Commercial Scale
Advanced Imatinib Purification: Achieving Ultra-Low Genotoxic Impurities for Commercial Scale
The pharmaceutical industry faces increasingly stringent regulatory scrutiny regarding genotoxic impurities in oncology drugs, particularly for tyrosine kinase inhibitors like Imatinib. Patent CN107245070B introduces a breakthrough purification methodology that addresses these critical safety concerns by reducing the content of the genotoxic impurity N-(5-amino-2-methylphenyl)-4-(3-pyridyl)-2-aminopyrimidine to below 2ppm, fully compliant with European and US Pharmacopeia standards. This technical advancement represents a significant leap forward in process chemistry, offering a route that combines high yield (>96%) with exceptional purity (>99.8%) through a simplified acid-base extraction and crystallization protocol. For R&D directors and procurement specialists, this patent outlines a robust pathway to secure supply chains for high-quality API intermediates while mitigating the risks associated with mutagenic contaminants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the removal of genotoxic impurity B from Imatinib has been a persistent bottleneck in manufacturing, often requiring complex, multi-step recrystallization sequences that compromise overall yield and operational efficiency. Prior art methods, such as those described in WO2012015999 and EP2927223, frequently rely on harsh pH adjustments using strong bases like sodium hydroxide, which can induce rapid precipitation and "oiling out," effectively trapping impurities within the crystal lattice. These conventional approaches often struggle to consistently achieve the rigorous <2ppm threshold, with many reported processes yielding products containing between 5ppm and 140ppm of the genotoxic species, necessitating further costly reprocessing. Furthermore, traditional methods often involve multiple salting-out steps with methanesulfonic acid, creating a cumbersome workflow that increases solvent consumption, waste generation, and production lead times, thereby inflating the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
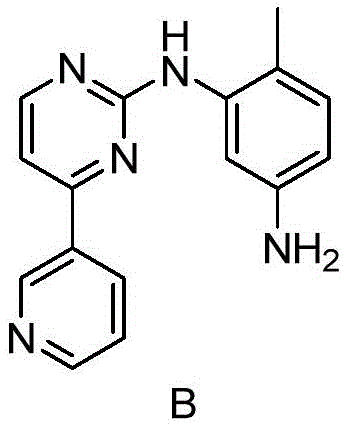
The innovative process disclosed in CN107245070B fundamentally re-engineers the purification logic by optimizing the partitioning behavior of Imatinib versus its impurities through precise pH control and targeted adsorption. Instead of aggressive basification, this method employs a gentle pH swing, initially dissolving the crude material in an acidic aqueous environment (pH 2.0-3.0) to separate it from organic-soluble contaminants, followed by a strategic decolorization step using activated carbon or diatomite mixtures. This approach effectively strips away the stubborn genotoxic impurity B, whose chemical structure is shown below, before the final crystallization event. By utilizing ammonia water for the final pH adjustment to 8.0-10.0, the process ensures slow, controlled crystal growth that excludes impurities, resulting in a fine product with purity exceeding 99.8% and impurity levels consistently below 1.8ppm, demonstrating superior selectivity compared to legacy techniques.
Mechanistic Insights into Acid-Base Extraction and Controlled Crystallization
The core mechanistic advantage of this purification strategy lies in the differential solubility and ionization states of Imatinib and its related substances across specific pH ranges. At a pH of 2.0 to 3.0, the basic nitrogen atoms on the Imatinib molecule are protonated, rendering the drug highly soluble in the aqueous phase, whereas many neutral or less basic organic impurities remain preferentially dissolved in the immiscible organic solvent layer (such as 2-methyltetrahydrofuran or ethyl acetate). This liquid-liquid extraction step acts as a primary bulk purification, removing a significant portion of synthetic by-products and starting materials before the critical crystallization phase begins. The subsequent addition of activated carbon serves as a secondary polishing mechanism, leveraging physical adsorption to capture planar aromatic impurities and colored bodies that might otherwise co-crystallize, ensuring the visual and chemical quality of the intermediate.
Furthermore, the choice of ammonia water as the basifying agent is critical for controlling the supersaturation profile during crystallization. Unlike strong inorganic bases that cause instantaneous pH spikes and chaotic nucleation, ammonia provides a buffered, gradual increase in pH, allowing the Imatinib free base to precipitate in a thermodynamically stable crystalline form. This controlled kinetics minimizes the inclusion of mother liquor within the crystal lattice, which is the primary vector for impurity entrapment. The structural integrity of the target molecule, depicted below, is maintained throughout this mild process, avoiding degradation pathways that can occur under harsh alkaline conditions, thus preserving the overall mass balance and yield of the valuable API intermediate.
![Chemical structure of Imatinib (Compound A): 4-[(4-methyl-1-piperazinyl)methyl]-N-[4-methyl-3-[[4-(3-pyridinyl)-2-pyrimidinyl]amino]phenyl]-benzamide](/insights/img/imatinib-purification-genotoxic-impurity-removal-supplier-20260309152934-01.png)
How to Synthesize High-Purity Imatinib Efficiently
Implementing this purification protocol requires strict adherence to the specified pH windows and temperature controls to maximize the rejection of genotoxic impurities. The process begins with the suspension of crude Imatinib in a biphasic system, followed by acidification to transfer the product into the aqueous layer, leaving lipophilic impurities behind. Detailed operational parameters, including solvent ratios and decolorization times, are critical for reproducibility. For process chemists looking to adopt this technology, the following standardized synthesis steps outline the exact procedure validated in the patent examples to achieve pharmaceutical-grade purity.
- Suspend crude imatinib in a water-organic solvent mixture and adjust pH to 2.0-3.0 using acid to dissolve the solid while separating layers.
- Treat the aqueous layer with activated carbon or a carbon-diatomite mixture at 40-60°C to adsorb colored impurities and residual organics.
- Adjust the filtrate pH to 8.0-10.0 using ammonia water at controlled temperatures to crystallize high-purity imatinib free base.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this purification method offers substantial strategic benefits for procurement managers and supply chain heads focused on cost efficiency and risk mitigation. The simplification of the workflow eliminates the need for multiple, iterative salting and recrystallization cycles that characterize older methods, directly translating to reduced processing time and lower utility consumption per kilogram of output. By streamlining the unit operations to a single effective extraction and crystallization sequence, manufacturers can significantly decrease the burden on production scheduling and equipment occupancy, allowing for higher throughput without capital expansion. This operational efficiency is a key driver for cost reduction in pharmaceutical intermediate manufacturing, enabling more competitive pricing structures for downstream API producers.
- Cost Reduction in Manufacturing: The elimination of complex multi-step salting procedures and the use of recoverable, common organic solvents drastically lowers the variable costs associated with production. By avoiding the repeated use of expensive reagents and minimizing solvent exchange steps, the overall material cost per batch is optimized. Additionally, the high yield (>96%) ensures that raw material utilization is maximized, reducing waste disposal costs and improving the overall economic viability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of this purification method against variations in crude feedstock quality ensures a consistent supply of high-purity material. Because the process is designed to handle crude inputs with impurity levels as high as 250ppm and still deliver <2ppm output, it provides a buffer against upstream synthesis fluctuations. This reliability reduces the risk of batch failures and supply interruptions, securing the continuity of supply for critical oncology medications.
- Scalability and Environmental Compliance: The process utilizes environmentally friendlier solvents like 2-methyltetrahydrofuran and ethyl acetate, aligning with modern green chemistry principles and reducing the regulatory burden associated with hazardous waste management. The simplicity of the equipment requirements—standard reactors and filtration units—facilitates easy scale-up from pilot to commercial production (100 kgs to 100 MT), ensuring that the technology can meet global demand without requiring specialized or exotic infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms traditional approaches in terms of impurity control and operational simplicity.
Q: How does this purification method reduce genotoxic impurity B below 2ppm?
A: The method utilizes a precise pH swing strategy. By dissolving the crude product at a highly acidic pH (2.0-3.0), the imatinib transfers to the aqueous phase while many organic impurities remain in the organic layer or are removed during extraction. Subsequent decolorization with activated carbon specifically targets trace aromatic impurities, and the gentle basification with ammonia ensures selective crystallization of pure imatinib, leaving the genotoxic impurity B in the mother liquor.
Q: Why is ammonia water preferred over sodium hydroxide for crystallization?
A: Using strong bases like sodium hydroxide can cause rapid precipitation, leading to the occlusion of impurities within the crystal lattice (oiling out). Ammonia water provides a more controlled and gradual increase in pH, allowing for the formation of well-defined crystals with higher purity and significantly lower entrapment of the genotoxic impurity N-(5-amino-2-methylphenyl)-4-(3-pyridyl)-2-aminopyrimidine.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids complex multi-step salting procedures and uses common, environmentally friendly solvents like 2-methyltetrahydrofuran or ethyl acetate. The operational simplicity, involving standard acid-base extraction and filtration equipment, ensures good reproducibility and safety for manufacturing at the 100 MT scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity and safety of oncology intermediates like Imatinib are non-negotiable priorities for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex purification challenges are met with rigorous scientific precision. Our facilities are equipped with state-of-the-art rigorous QC labs capable of detecting genotoxic impurities at the ppm level, guaranteeing that every batch meets stringent purity specifications and complies with international pharmacopeial standards.
We invite procurement leaders and R&D teams to collaborate with us to leverage this advanced purification technology for your supply chain. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can deliver high-quality Imatinib intermediates with superior cost-performance ratios and reliable delivery schedules.
