Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Intermediates for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Intermediates for Scalable Pharmaceutical Manufacturing
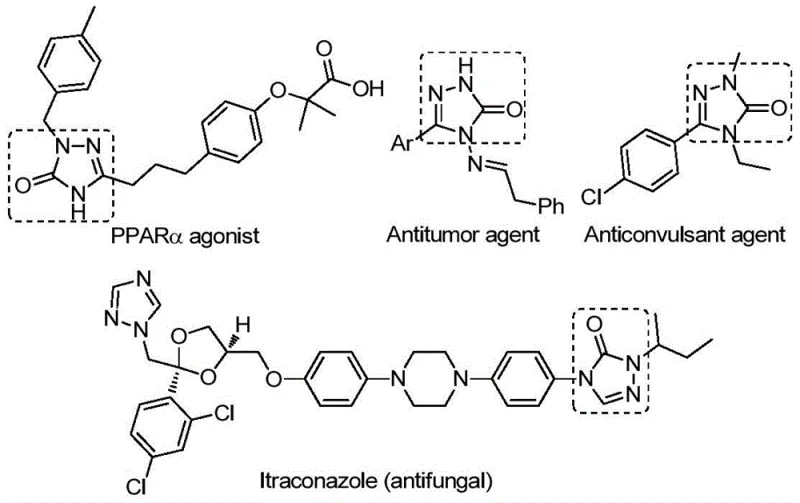
The pharmaceutical industry continuously seeks robust synthetic methodologies to access privileged scaffolds efficiently, and the recent disclosure in patent CN112538054B represents a significant leap forward in the construction of 1,2,4-triazole-3-one derivatives. This specific class of five-membered nitrogen-containing heterocycles is ubiquitous in medicinal chemistry, serving as the core structural motif for a diverse array of bioactive molecules including potent antifungal, anti-inflammatory, antitumor, and anticonvulsant agents. As depicted in the structural diversity shown below, these compounds function as critical tyrosinase inhibitors, CB receptor modulators, and angiotensin AT1 receptor antagonists, underlining their immense value in drug discovery pipelines. The patented methodology introduces a transition metal palladium-catalyzed carbonylation tandem cyclization reaction that fundamentally alters the accessibility of these high-value intermediates.
By leveraging inexpensive and commercially available starting materials such as chlorohydrazones and sodium azide, this novel approach circumvents the historical bottlenecks associated with heterocycle synthesis. The process operates under relatively mild thermal conditions and exhibits exceptional functional group tolerance, allowing for the rapid generation of libraries substituted with various alkyl, aryl, and halogen groups. For R&D directors and process chemists, this translates to a versatile platform technology that can be adapted for the late-stage functionalization of complex drug candidates. Furthermore, the operational simplicity of the reaction, which avoids the handling of hazardous gaseous carbon monoxide, positions this technology as a prime candidate for seamless technology transfer from laboratory benchtop to commercial manufacturing suites.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazol-3-one compounds has been plagued by significant synthetic inefficiencies that hinder both research throughput and commercial viability. Traditional protocols often rely on the cyclization of benzoyl hydrazides with urea under strongly basic conditions, or the tandem reaction of hydrazides with isocyanates, which necessitates the use of highly toxic and volatile reagents. Other established routes involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines, all of which suffer from severe drawbacks. These legacy methods are frequently characterized by harsh reaction environments that degrade sensitive functional groups, multi-step sequences that require tedious pre-activation of substrates, and generally low overall yields that inflate the cost of goods. Additionally, the narrow substrate scope of these conventional techniques often fails to accommodate the diverse structural requirements of modern drug design, forcing chemists to resort to circuitous synthetic detours.
The Novel Approach
In stark contrast to these archaic strategies, the methodology described in CN112538054B offers a streamlined, one-pot solution that dramatically simplifies the molecular architecture assembly. The core innovation lies in the use of a palladium catalyst system combined with a solid carbon monoxide surrogate, TFBen, which releases CO in situ upon heating. This clever design eliminates the need for specialized high-pressure equipment required for gaseous CO handling, thereby enhancing laboratory safety and reducing capital expenditure. As illustrated in the general reaction scheme below, the transformation proceeds via a tandem cyclization where chlorohydrazone and sodium azide converge to form the triazole ring with high atom economy. The reaction conditions are remarkably forgiving, utilizing common organic solvents like 1,4-dioxane and tolerating a wide range of substituents on both the hydrazone and the nitrogen atom, thus enabling the rapid synthesis of diverse analogues for structure-activity relationship studies.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and impurity control. The reaction is postulated to initiate with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the thermal decomposition of TFBen releases carbon monoxide directly into the reaction medium, which subsequently inserts into the carbon-palladium bond to form an acyl-palladium species. This acyl intermediate then reacts with sodium azide to generate an acyl azide compound, which undergoes a classic Curtius rearrangement to yield an isocyanate intermediate. The final ring closure occurs via an intramolecular nucleophilic addition, where the pendant hydrazine nitrogen attacks the electrophilic isocyanate carbon, forging the five-membered 1,2,4-triazole-3-one ring. This intricate cascade ensures high regioselectivity and minimizes the formation of side products, resulting in a clean reaction profile that simplifies downstream purification.
From an impurity management perspective, the use of well-defined catalytic cycles and stable intermediates significantly reduces the complexity of the crude reaction mixture. Unlike radical-based or high-temperature thermal cyclizations that often produce polymeric tars or isomeric byproducts, this palladium-mediated pathway is highly specific. The choice of ligands, such as Xantphos, further stabilizes the active catalytic species, preventing palladium black precipitation and ensuring consistent turnover numbers throughout the reaction duration. For quality control teams, this means that the impurity profile is predictable and manageable, with the primary contaminants likely being unreacted starting materials or simple hydrolysis products that are easily removed during standard workup procedures. The ability to tune the electronic properties of the substrate without disrupting the catalytic cycle also allows for the synthesis of high-purity intermediates required for stringent regulatory filings.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible even to laboratories with standard equipment. The protocol involves charging a reaction vessel with the palladium source, ligand, CO surrogate, chlorohydrazone, and sodium azide in an aprotic solvent, followed by heating to 100-120°C. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and safety compliance.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), CO surrogate (TFBen), chlorohydrazone, and sodium azide in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to allow the tandem cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical elegance. The shift from hazardous gaseous reagents to stable solid surrogates drastically reduces the safety infrastructure costs associated with manufacturing, while the use of commodity chemicals ensures a resilient supply chain unaffected by niche reagent shortages. The high efficiency and broad substrate scope mean that a single manufacturing line can be utilized to produce a wide variety of derivatives, maximizing asset utilization and reducing the need for dedicated campaign setups. Furthermore, the simplified workup procedure minimizes solvent consumption and waste generation, aligning with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous carbon monoxide gas cylinders in favor of the solid surrogate TFBen represents a significant operational cost saving. Additionally, the starting materials, specifically chlorohydrazones and sodium azide, are commodity chemicals available in bulk quantities at low prices, which drives down the raw material cost per kilogram of the final API intermediate. The high reaction efficiency and yield reduce the amount of starting material wasted, further optimizing the cost structure. By avoiding complex multi-step sequences and pre-activation stages, the overall processing time and labor costs are substantially reduced, leading to a more competitive pricing model for the final product.
- Enhanced Supply Chain Reliability: Relying on widely available, non-proprietary reagents mitigates the risk of supply disruptions that often plague specialized chemical syntheses. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating market demands without lengthy lead times. The stability of the reagents also simplifies storage and transportation logistics, removing the need for specialized containment or temperature-controlled shipping. This reliability ensures a continuous flow of high-quality intermediates to downstream formulation partners, safeguarding the integrity of the entire pharmaceutical supply network.
- Scalability and Environmental Compliance: The transition from batch to continuous flow or large-scale batch production is facilitated by the homogeneous nature of the reaction and the absence of gas-liquid mass transfer limitations. The use of aprotic solvents like dioxane, which can be recovered and recycled, minimizes the environmental footprint of the process. The straightforward purification via filtration and chromatography reduces the volume of aqueous waste generated, easing the burden on wastewater treatment facilities. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the primary advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods requiring harsh conditions or pre-activated substrates, this protocol uses cheap, readily available chlorohydrazones and sodium azide under mild conditions with high substrate compatibility.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes stable solid reagents instead of toxic CO gas, simplifies the workup to filtration and chromatography, and demonstrates excellent scalability from milligram to multi-kilogram batches.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen acts as a safe carbon monoxide surrogate, releasing CO in situ under heating conditions to facilitate the carbonylation step without the need for high-pressure gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and risk-free. We are committed to delivering high-purity 1,2,4-triazole-3-one intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch conforms to the highest international standards, providing our clients with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this advanced palladium-catalyzed technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your time-to-market with our reliable, scalable, and cost-effective manufacturing solutions for complex pharmaceutical intermediates.
