Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Iron Catalysis for Pharmaceutical Intermediates
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Iron Catalysis for Pharmaceutical Intermediates
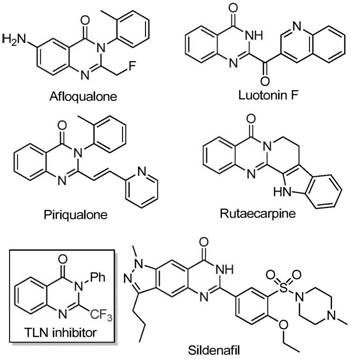
The pharmaceutical industry continuously seeks robust synthetic pathways for nitrogen-containing heterocycles, particularly quinazolinones, which serve as critical scaffolds in numerous bioactive molecules. As illustrated in the structural diversity of known bioactive compounds such as Afloqualone and Luotonin F, the quinazolinone core is ubiquitous in drug discovery programs targeting cancer, inflammation, and fungal infections. The strategic introduction of a trifluoromethyl group into this scaffold further enhances metabolic stability and lipophilicity, properties essential for modern drug design. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds that addresses long-standing challenges in heterocyclic synthesis. This technology leverages a cost-effective iron-catalyzed system to transform readily available starting materials into high-value intermediates, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate with substrates like anthranilamide or isatoic anhydride. While chemically feasible, these traditional routes suffer from significant drawbacks that hinder efficient cost reduction in API manufacturing. The reaction conditions are often severe, requiring harsh reagents that limit functional group tolerance and lead to complex impurity profiles. Furthermore, the reliance on specialized trifluoromethyl synthons often results in narrow substrate scope and lower overall yields, creating bottlenecks in the supply chain for high-purity OLED material or pharmaceutical precursors. These inefficiencies translate directly into higher production costs and extended lead times, making conventional methods less attractive for large-scale commercial operations where margin compression is a constant pressure.
The Novel Approach
In stark contrast, the methodology described in CN111675662B utilizes a novel combination of trifluoroethylimidoyl chloride and isatin as the primary building blocks. This approach fundamentally shifts the economic and operational landscape of quinazolinone synthesis by employing earth-abundant iron catalysts instead of precious metals. The reaction proceeds through a streamlined sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This pathway not only simplifies the synthetic route but also dramatically improves the atom economy and substrate versatility. By avoiding expensive and sensitive reagents, this novel approach enables the design and synthesis of quinazolinone compounds with trifluoromethyl substitution at various positions, providing a flexible platform for generating diverse chemical libraries required for advanced drug discovery and development pipelines.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the intricate interplay between the iron catalyst and the base promoter. The reaction mechanism initiates with the formation of a trifluoroacetamidine intermediate through the nucleophilic attack of the isatin nitrogen on the imidoyl chloride. Subsequently, the ferric chloride catalyst facilitates a crucial decarbonylation step, driving the rearrangement of the intermediate into the stable quinazolinone ring system. This iron-catalyzed cycle is highly efficient, operating effectively under aerobic conditions, which simplifies the reactor setup by eliminating the need for stringent inert gas protection typically required for sensitive organometallic transformations. The use of sodium hydride as a base ensures the deprotonation necessary for the initial coupling, while the specific choice of DMF as a solvent optimizes the solubility of both organic substrates and inorganic salts, ensuring homogeneous reaction kinetics throughout the process.
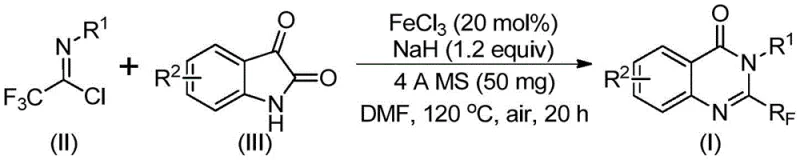
Impurity control is another critical aspect where this mechanism excels, particularly for R&D directors focused on purity specifications. The inclusion of 4A molecular sieves in the reaction mixture plays a pivotal role in scavenging trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride or deactivate the catalyst. By maintaining anhydrous conditions in situ, the process minimizes the formation of hydrolysis byproducts and ensures high conversion rates. The tolerance for various substituents on the aryl rings, including halogens, alkyl groups, and methoxy functionalities, demonstrates the robustness of the catalytic cycle. This broad functional group tolerance means that complex analogues can be synthesized without requiring extensive protecting group strategies, thereby reducing the number of synthetic steps and associated waste generation, aligning perfectly with green chemistry principles and environmental compliance standards.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and industrial scale-up of complex polymer additives or pharma intermediates. The protocol involves a straightforward one-pot procedure where all reagents are combined in a specific molar ratio, typically utilizing a slight excess of the imidoyl chloride to drive the reaction to completion. The detailed standardized synthetic steps outlined below provide a clear roadmap for replicating the high yields reported in the patent data, ensuring consistency across different batches. This operational simplicity reduces the training burden on technical staff and minimizes the risk of operator error, which is a common source of variability in fine chemical manufacturing.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in DMF solvent within a reaction vessel.
- Initiate the reaction by stirring at 40°C for approximately 10 hours to facilitate initial bond formation, then increase temperature to 120°C.
- Maintain heating at 120°C for an additional 20 hours under air atmosphere to complete the decarbonylation and cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology offers tangible strategic benefits beyond mere technical feasibility. The primary advantage stems from the drastic simplification of the raw material portfolio, replacing costly and potentially supply-constrained precious metal catalysts with commodity-grade ferric chloride. This shift not only lowers the direct material cost but also mitigates the geopolitical and market volatility risks associated with rare earth and precious metal sourcing. Furthermore, the use of isatin and aromatic amines as starting materials leverages well-established global supply chains, ensuring consistent availability and competitive pricing for bulk purchases. This stability is crucial for maintaining uninterrupted production schedules and meeting the rigorous delivery commitments expected by downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or rhodium results in substantial cost savings, as these metals often account for a significant portion of the raw material budget in heterocyclic synthesis. Additionally, the simplified post-treatment process, which avoids complex heavy metal scavenging steps, reduces the consumption of auxiliary chemicals and waste disposal costs. The high reaction efficiency and yield reported in the patent data imply less raw material waste per kilogram of product, further enhancing the overall economic viability of the process for commercial scale-up.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals like ferric chloride, sodium hydride, and DMF ensures that production is not vulnerable to the supply disruptions that frequently plague specialty reagent markets. The robustness of the reaction conditions, which tolerate air and moisture to a reasonable extent thanks to the molecular sieves, allows for more flexible manufacturing scheduling and reduces the need for specialized infrastructure. This resilience translates into shorter lead times for high-purity pharmaceutical intermediates, enabling suppliers to respond more agilely to fluctuating market demands and urgent customer orders.
- Scalability and Environmental Compliance: The protocol's demonstrated success from gram to potential ton scales indicates a low barrier to technology transfer, allowing manufacturers to rapidly ramp up production capacity without extensive re-engineering. The use of iron, a non-toxic and environmentally benign metal, aligns with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical wastewater. This eco-friendly profile simplifies the permitting process for new production lines and enhances the corporate sustainability credentials of the manufacturer, a factor that is becoming increasingly important in vendor selection criteria for major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing manufacturing frameworks or sourcing strategies for next-generation therapeutic agents.
Q: What are the advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: Using ferric chloride eliminates the need for expensive palladium or rhodium catalysts, significantly reducing raw material costs and simplifying the removal of toxic heavy metal residues from the final API intermediate.
Q: How does this method improve substrate tolerance compared to traditional anhydride routes?
A: The use of trifluoroethylimidoyl chloride and isatin allows for broader functional group compatibility, enabling the synthesis of diverse derivatives with halogen, alkyl, or methoxy substituents without severe side reactions.
Q: Is this synthesis protocol suitable for large-scale industrial production?
A: Yes, the protocol utilizes commercially available reagents and standard solvents like DMF, and the patent explicitly demonstrates scalability from gram levels to potential multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis route for the production of high-value quinazolinone derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest quality standards required for clinical and commercial applications. We are committed to leveraging this innovative chemistry to drive value for our partners in the global pharmaceutical sector.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your journey from early-stage development to full-scale commercial manufacturing with efficiency and precision.
