Scalable Synthesis of Trifluoromethylpyridines via Molybdenum Oxide Catalysis for Global Agrochemical Supply Chains
The global demand for fluorinated heterocycles, particularly trifluoromethylpyridines, has surged due to their pivotal role as key intermediates in the synthesis of high-performance agrochemicals and pharmaceuticals. Patent CN101092393A introduces a transformative manufacturing methodology that addresses long-standing inefficiencies in producing these critical building blocks. This technology utilizes a robust molybdenum oxide-catalyzed gas-phase or liquid-phase fluorination process, converting readily available trichloromethylpyridine precursors into valuable trifluoromethyl derivatives. For R&D Directors and Supply Chain Heads, this represents a significant opportunity to optimize the production of essential intermediates used in herbicides like fluazifop and fungicides such as fluazinam. The process operates under relatively mild conditions compared to historical methods, utilizing anhydrous hydrogen fluoride and a stable, non-hygroscopic catalyst system that ensures consistent quality and operational safety.
The strategic value of this patent lies in its ability to bypass the severe limitations of prior art, which often relied on toxic heavy metals or hazardous reagents. By shifting to a molybdenum-based catalytic cycle, manufacturers can achieve yields typically ranging from 70% to 80% with exceptional purity levels exceeding 99.5%. This level of efficiency is not merely a laboratory curiosity but a commercially viable pathway designed for large-scale industrial implementation. The following analysis details the mechanistic advantages, operational parameters, and supply chain benefits of adopting this novel synthetic route for the reliable production of high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethylpyridines has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex fluorinated heterocycles. Traditional methods described in patents such as US4680406 and US4567273 relied heavily on mercurous or silver-based catalysts supported on reaction beds. These heavy metal systems present profound disadvantages, including extreme sensitivity to reaction conditions, high equipment corrosion rates, and stringent environmental disposal requirements due to mercury toxicity. Furthermore, alternative routes utilizing sulfur tetrafluoride (SF4), as seen in US007518, involve reagents that are notoriously difficult to synthesize and handle due to their high toxicity and reactivity, posing severe safety risks to plant personnel. Other methods employing antimony trifluoride (SbF3) or iron-based catalysts often suffer from unacceptably low yields or require excessively high temperatures and prolonged reaction times, leading to increased energy consumption and the formation of difficult-to-remove by-products. These legacy processes create bottlenecks in cost reduction in agrochemical intermediate manufacturing, as the downstream purification and waste treatment costs often outweigh the value of the final product.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN101092393A leverages a novel catalytic system based on molybdenum oxides (MoO2, MoO3, or combinations thereof) to drive the selective fluorination of trichloromethyl groups. This approach fundamentally alters the economic and operational landscape of trifluoromethylpyridine production. The core innovation involves reacting trichloromethylpyridine compounds, represented by Formula (I), with anhydrous hydrogen fluoride in the presence of the molybdenum catalyst at temperatures between 100°C and 250°C. 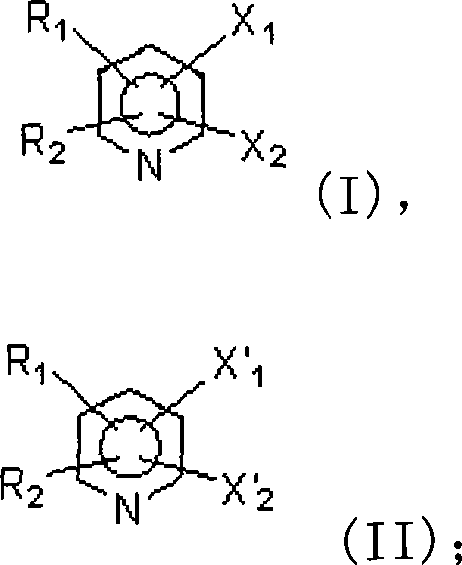 This transformation efficiently replaces the chlorine atoms of the trichloromethyl group with fluorine atoms to yield the target trifluoromethylpyridine structures shown in Formula (II), without affecting other sensitive functional groups on the pyridine ring. The use of molybdenum oxide is particularly advantageous because, unlike molybdenum pentachloride or other high-valency metal halides, it is not hygroscopic and does not decompose upon exposure to air, ensuring catalyst stability during storage and handling. This stability translates directly into process reliability, allowing for consistent batch-to-batch performance and eliminating the need for specialized inert atmosphere handling equipment required by moisture-sensitive catalysts.
This transformation efficiently replaces the chlorine atoms of the trichloromethyl group with fluorine atoms to yield the target trifluoromethylpyridine structures shown in Formula (II), without affecting other sensitive functional groups on the pyridine ring. The use of molybdenum oxide is particularly advantageous because, unlike molybdenum pentachloride or other high-valency metal halides, it is not hygroscopic and does not decompose upon exposure to air, ensuring catalyst stability during storage and handling. This stability translates directly into process reliability, allowing for consistent batch-to-batch performance and eliminating the need for specialized inert atmosphere handling equipment required by moisture-sensitive catalysts.
Mechanistic Insights into MoO3-Catalyzed Halogen Exchange
The efficacy of this process stems from the unique Lewis acidic properties of molybdenum oxides, which facilitate the activation of the carbon-chlorine bonds in the trichloromethyl side chain. In the presence of anhydrous hydrogen fluoride, the molybdenum catalyst likely acts to polarize the C-Cl bond, making the carbon atom more susceptible to nucleophilic attack by fluoride ions. This mechanism promotes a stepwise halogen exchange where the trichloromethyl group (-CCl3) is sequentially converted first to a chlorodifluoromethyl group (-CClF2), then to a dichlorofluoromethyl group (-CCl2F), and finally to the thermodynamically stable trifluoromethyl group (-CF3). The reaction conditions, specifically the temperature range of 150°C to 200°C and the use of excess HF (molar ratio of 1:5 to 1:10), are optimized to drive this equilibrium towards the fully fluorinated product while minimizing the accumulation of partially fluorinated intermediates. Crucially, the catalyst demonstrates high chemoselectivity; it targets the aliphatic C-Cl bonds of the side chain while leaving aromatic C-Cl or C-Br bonds on the pyridine ring intact. This selectivity is vital for R&D teams aiming to synthesize specific derivatives like 2,3-dichloro-5-trifluoromethylpyridine, where preserving the ring chlorines is essential for subsequent coupling reactions in pesticide synthesis.
Impurity control is another critical aspect of this mechanistic pathway. The patent data indicates that maintaining the reaction temperature below 250°C is essential to prevent over-fluorination or ring degradation, which can lead to the formation of high-boiling impurities that complicate downstream purification. By operating within the optimal window of 150°C to 200°C, the process minimizes secondary reactions and ensures that the crude product profile is dominated by the desired trifluoromethyl species. Post-reaction workup is straightforward, involving neutralization of residual acid, phase separation, and fractional distillation. The non-volatile nature of the molybdenum oxide catalyst allows it to remain in the reactor residue or be easily separated, preventing metal contamination of the final distillate. This results in a product with purity specifications often exceeding 99.5%, meeting the rigorous standards required for pharmaceutical and agrochemical applications without the need for extensive chromatographic purification.
How to Synthesize Trifluoromethylpyridine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology at pilot or commercial scales. The process begins with the charging of the trichloromethylpyridine substrate and the molybdenum oxide catalyst into a corrosion-resistant reactor capable of withstanding hydrogen fluoride. The reaction is initiated by introducing anhydrous HF gas, and the system is maintained at the target temperature with continuous monitoring via gas chromatography to track the conversion of intermediates. Detailed standardized synthesis steps follow below, outlining the precise stoichiometry and operational parameters required to replicate the high yields reported in the patent examples.
- Charge a reactor with trichloromethylpyridine substrate (Formula I) and 0.1-10 wt% molybdenum oxide catalyst (MoO2 or MoO3).
- Heat the mixture to 100-250°C (preferably 150-200°C) and introduce anhydrous hydrogen fluoride gas at a molar ratio of 1: (3-15).
- Maintain reaction pressure between 1-10 kPa for 8-23 hours, then neutralize, wash, and distill to isolate the trifluoromethylpyridine product (Formula II) with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this molybdenum-catalyzed fluorination technology offers substantial strategic advantages beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the supply chain for catalyst materials. Unlike the specialized and often regulated mercury or silver catalysts of the past, molybdenum oxides are commodity chemicals available globally in large quantities at low cost. This shift eliminates supply bottlenecks associated with scarce precious metals and reduces the volatility of raw material pricing. Furthermore, the stability of the catalyst means it can be stored under ambient conditions without degradation, reducing inventory management costs and the risk of material loss due to moisture sensitivity. The elimination of toxic heavy metals from the process also significantly lowers the barrier for environmental compliance, reducing the costs and complexities associated with hazardous waste disposal and effluent treatment.
- Cost Reduction in Manufacturing: The transition to a molybdenum oxide catalyst system drives cost reduction in agrochemical intermediate manufacturing through multiple channels. First, the catalyst itself is significantly cheaper than noble metal alternatives, directly lowering the bill of materials. Second, the process operates without the need for organic solvents in the fluorination step, utilizing HF as both reagent and solvent medium, which reduces solvent purchase and recovery costs. Third, the high selectivity of the reaction minimizes the formation of by-products, thereby increasing the overall mass efficiency and reducing the load on distillation columns. Finally, the absence of heavy metals removes the need for expensive scavenging resins or complex filtration steps to meet metal residue specifications, streamlining the post-reaction workflow and lowering operational expenditures.
- Enhanced Supply Chain Reliability: Reliability is paramount for the commercial scale-up of complex polymer additives and agrochemical intermediates. This method enhances supply chain reliability by utilizing raw materials that are already produced on a massive industrial scale. Trichloromethylpyridines are established commodities, and molybdenum oxides are widely mined and processed, ensuring a steady flow of inputs even during market fluctuations. The robustness of the catalyst against moisture and air exposure further secures the supply line, as there is no risk of catalyst deactivation during transport or storage. This resilience allows manufacturers to maintain consistent production schedules and meet tight delivery windows for downstream customers, reducing the risk of stockouts that can disrupt the production of finished herbicides or fungicides.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions (100-250°C, 1-10 kPa) that are easily manageable in standard industrial fluorination reactors. The simplicity of the workup—neutralization, washing, and distillation—facilitates easy scale-up from kilogram to multi-ton batches without requiring exotic equipment. From an environmental perspective, the method aligns with modern green chemistry principles by avoiding toxic reagents like SF4 and heavy metal catalysts. The reduced generation of hazardous waste and the potential for recycling unreacted starting materials contribute to a lower environmental footprint. This compliance advantage is increasingly critical as global regulations on chemical manufacturing tighten, positioning companies that adopt this technology as leaders in sustainable and responsible chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These answers are derived directly from the experimental data and claims presented in patent CN101092393A, providing a factual basis for evaluating the process viability. Understanding these details is essential for technical teams assessing the fit of this method within their existing manufacturing infrastructure.
Q: Why is molybdenum oxide preferred over traditional mercury or antimony catalysts?
A: Molybdenum oxide catalysts (MoO2/MoO3) are non-hygroscopic, chemically stable, and commercially inexpensive compared to moisture-sensitive mercury or antimony salts. They eliminate the need for complex heavy metal scavenging steps, significantly simplifying waste treatment and reducing environmental compliance costs.
Q: What is the expected yield and purity for this fluorination process?
A: According to patent data CN101092393A, the process consistently achieves yields between 70% and 80% with product purity exceeding 99.5% after standard distillation, making it highly suitable for high-specification agrochemical applications.
Q: Can this method tolerate other halogens on the pyridine ring?
A: Yes, the method exhibits high chemoselectivity. It selectively fluorinates the trichloromethyl side chain (CCl3 to CF3) while preserving ring substituents such as chlorine or bromine, which is critical for synthesizing diverse intermediates like 2,3-dichloro-5-trifluoromethylpyridine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation agrochemicals and pharmaceuticals. Our technical team has extensively analyzed the molybdenum-catalyzed fluorination pathway described in CN101092393A and possesses the expertise to implement this advanced chemistry at scale. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with state-of-the-art corrosion-resistant reactors and rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace halogenated impurities and residual metals. We are committed to delivering trifluoromethylpyridine derivatives that meet the highest industry standards for purity and performance.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific project requirements. Whether you need custom synthesis of novel fluorinated pyridines or reliable supply of established intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume and purity needs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us partner with you to optimize your supply chain and accelerate the development of your high-value fluorinated products.
