Scalable Production of 2-Trifluoromethyl Quinazolinones via Iron-Catalyzed Cyclization
Scalable Production of 2-Trifluoromethyl Quinazolinones via Iron-Catalyzed Cyclization
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Quinazolinones, in particular, represent a privileged scaffold found in numerous drugs exhibiting anticancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into these structures further enhances their metabolic stability, lipophilicity, and bioavailability, making them highly desirable targets for drug discovery programs. A groundbreaking preparation method disclosed in patent CN111675662B addresses the historical challenges associated with synthesizing these complex motifs. This innovation utilizes a cost-effective iron-catalyzed system to couple readily available isatins with trifluoroethylimidoyl chlorides, offering a streamlined pathway that bypasses the limitations of traditional methodologies.
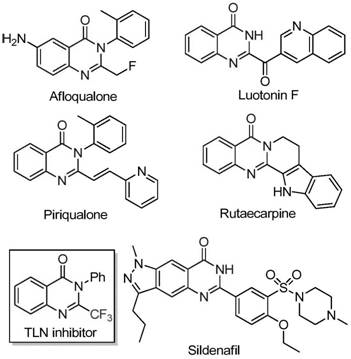
This technological advancement is particularly relevant for R&D directors and procurement specialists looking to optimize the supply chain for high-purity pharmaceutical intermediates. By leveraging earth-abundant iron catalysts instead of expensive transition metals, the process significantly lowers the barrier to entry for producing these valuable scaffolds. The method not only simplifies the operational workflow but also ensures high yields across a broad substrate scope, including various halogenated and alkyl-substituted derivatives. For global supply chain heads, the ability to source key starting materials like isatin and aromatic amines from established markets ensures continuity and reduces the risk of bottlenecks often associated with exotic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on cyclization reactions involving specialized synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. While chemically feasible, these conventional routes suffer from several critical drawbacks that hinder their industrial applicability. The reaction conditions are often severe, requiring harsh reagents that pose safety risks and complicate waste management protocols. Furthermore, the starting materials, particularly the fluorinated synthons, can be prohibitively expensive and difficult to source in bulk quantities, leading to inflated production costs. Yields in these traditional methods are frequently inconsistent and generally low, necessitating extensive purification steps that erode overall process efficiency. The narrow substrate scope of these older methods also limits the chemical diversity accessible to medicinal chemists, restricting the exploration of structure-activity relationships.
The Novel Approach
In stark contrast, the novel methodology described in the patent introduces a paradigm shift by utilizing trifluoroethylimidoyl chloride and isatin derivatives as the primary building blocks. This approach capitalizes on the high reactivity of the imidoyl chloride moiety, which facilitates efficient bond formation under much milder conditions. The core of this innovation lies in the use of ferric chloride (FeCl3) as a catalyst, promoted by sodium hydride (NaH) and 4A molecular sieves in a polar aprotic solvent like DMF. This catalytic system enables a tandem sequence involving initial carbon-nitrogen bond formation followed by an iron-catalyzed decarbonylation and cyclization. The result is a direct, atom-economical route to the target quinazolinone core. As illustrated in the reaction scheme below, this process tolerates a wide array of functional groups, allowing for the modular synthesis of diverse derivatives with high precision and reliability.
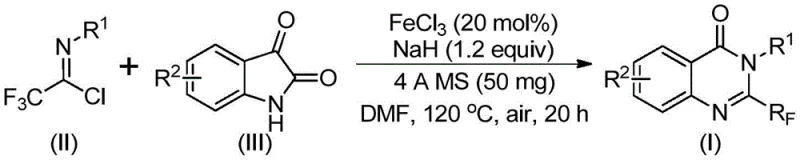
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a testament to the versatility of iron catalysis in organic synthesis. The reaction initiates with the base-promoted nucleophilic attack of the isatin nitrogen on the electrophilic carbon of the trifluoroethylimidoyl chloride, forming a trifluoroacetamidine intermediate. This step is crucial as it establishes the necessary connectivity for the subsequent ring closure. Following this, the ferric chloride catalyst plays a pivotal role in activating the carbonyl group of the isatin moiety, facilitating a decarbonylation event that drives the thermodynamic equilibrium towards the formation of the stable quinazolinone ring system. The presence of 4A molecular sieves is instrumental in sequestering moisture and potentially trapping small molecule byproducts, thereby pushing the reaction to completion. This intricate interplay between the base, the Lewis acid catalyst, and the drying agent ensures high conversion rates even with sterically demanding substrates.
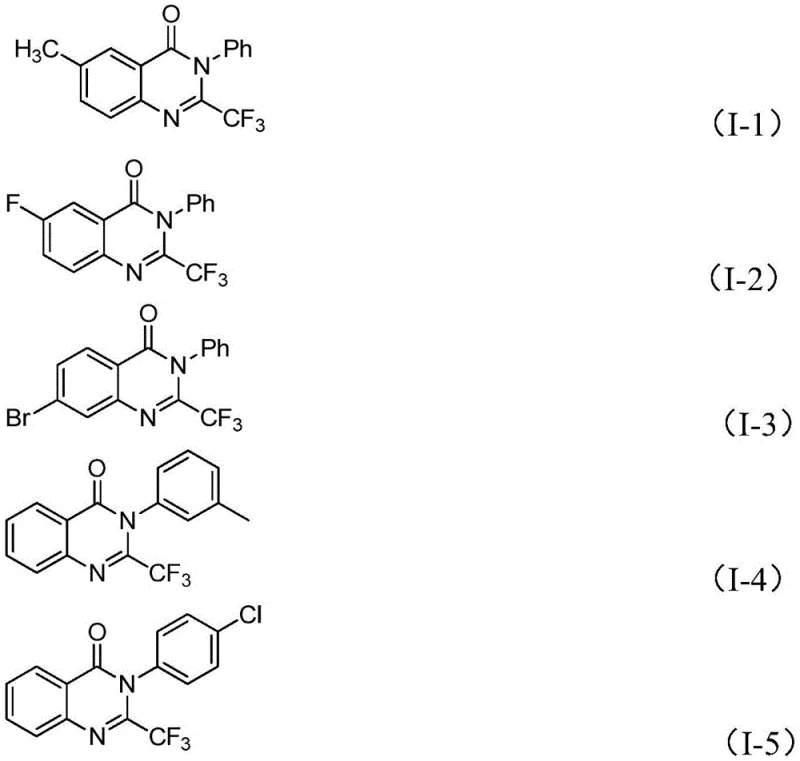
From an impurity control perspective, this mechanism offers distinct advantages. The specificity of the iron-catalyzed decarbonylation minimizes the formation of side products commonly seen in non-catalytic thermal cyclizations. The reaction conditions are tuned to favor the desired intramolecular cyclization over intermolecular polymerization or hydrolysis of the sensitive imidoyl chloride. Consequently, the crude reaction mixtures are cleaner, simplifying the downstream purification process. The patent data indicates that simple column chromatography is sufficient to isolate the products in high purity, which is a critical factor for meeting the stringent quality standards required for pharmaceutical intermediates. The ability to synthesize compounds like those shown below (I-1 to I-5) with consistent quality underscores the robustness of this mechanistic design.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and temperature profiling to maximize yield. The protocol is designed to be operationally simple, avoiding the need for inert atmosphere techniques beyond standard Schlenk line practices, which facilitates easier adoption in various manufacturing environments. The reaction proceeds in two distinct thermal stages: an initial lower temperature phase to form the intermediate, followed by a higher temperature phase to drive the cyclization. This staged heating profile is essential for controlling the reaction kinetics and preventing the decomposition of sensitive reagents. For detailed procedural specifics regarding reagent grades, exact addition rates, and workup parameters, operators should refer to the standardized synthesis steps provided in the technical documentation below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40 °C for approximately 10 hours to initiate the formation of intermediates.
- Heat the reaction to 120 °C and maintain for 20 hours under air to complete the decarbonylation and cyclization, followed by filtration and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this iron-catalyzed route presents a compelling value proposition centered on cost efficiency and supply security. The shift away from precious metal catalysts to ubiquitous iron salts drastically reduces the raw material cost per kilogram of product. Moreover, the starting materials—isatin and substituted aromatic amines—are commodity chemicals produced on a massive scale globally, ensuring a stable and competitive supply market. This contrasts sharply with older methods that rely on niche fluorinated reagents subject to volatile pricing and limited availability. The simplified post-treatment process, which avoids complex extraction or heavy metal scavenging steps, further contributes to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts in favor of ferric chloride represents a direct reduction in catalyst expenditure. Additionally, the high atom economy of the reaction means less waste generation, which lowers disposal costs. The use of DMF, a common industrial solvent, allows for easy recycling and recovery, enhancing the overall economic viability of the process. These factors combine to create a significantly leaner cost structure for the production of these high-value intermediates.
- Enhanced Supply Chain Reliability: Sourcing risk is minimized because the key precursors are widely available from multiple global suppliers. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental controls or reagent quality, leading to more predictable batch outcomes. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, reducing the likelihood of stockouts or production delays caused by synthetic failures.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram scales up to potential multi-ton production. The absence of toxic heavy metals simplifies regulatory compliance regarding residual metal limits in the final product. Furthermore, the reduced waste profile aligns with modern green chemistry principles, making it easier to obtain necessary environmental permits for large-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent literature, providing clarity on the practical aspects of adopting this method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for this synthesis?
A: The use of ferric chloride (FeCl3) offers significant economic and operational advantages compared to precious metal catalysts. It is inexpensive, readily available, and demonstrates high catalytic efficiency in promoting the decarbonylation cyclization required for quinazolinone formation, thereby reducing overall production costs.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the protocol exhibits excellent functional group tolerance. It successfully accommodates various substituents such as methyl, halogen (fluoro, bromo, chloro, iodo), methoxy, and nitro groups at different positions on the aromatic rings, allowing for the synthesis of a wide library of derivatives without compromising yield.
Q: Is the reaction scalable for industrial manufacturing?
A: The method is designed for scalability. The patent explicitly states that the process can be expanded from gram-level laboratory synthesis to industrial-scale application. The use of common solvents like DMF and robust reaction conditions supports reliable commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the potential of this iron-catalyzed cyclization route and is prepared to leverage it for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest international standards.
We invite you to collaborate with us to unlock the full potential of this technology for your pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to discuss your specific needs,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us be your trusted partner in delivering high-quality pharmaceutical intermediates with speed, precision, and economic efficiency.
