Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Production
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and safer synthetic routes to bioactive scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN112480015B, which discloses a robust multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. These privileged structures are ubiquitous in medicinal chemistry, serving as the core framework for numerous pharmaceutical agents exhibiting antifungal, antiviral, anti-inflammatory, and anticancer properties. The strategic incorporation of the trifluoromethyl group further enhances the metabolic stability and lipophilicity of these molecules, making them highly desirable candidates for drug discovery programs. As a leading entity in fine chemical manufacturing, understanding and leveraging such patented methodologies allows us to offer superior reliable pharmaceutical intermediate supplier capabilities to our global partners.
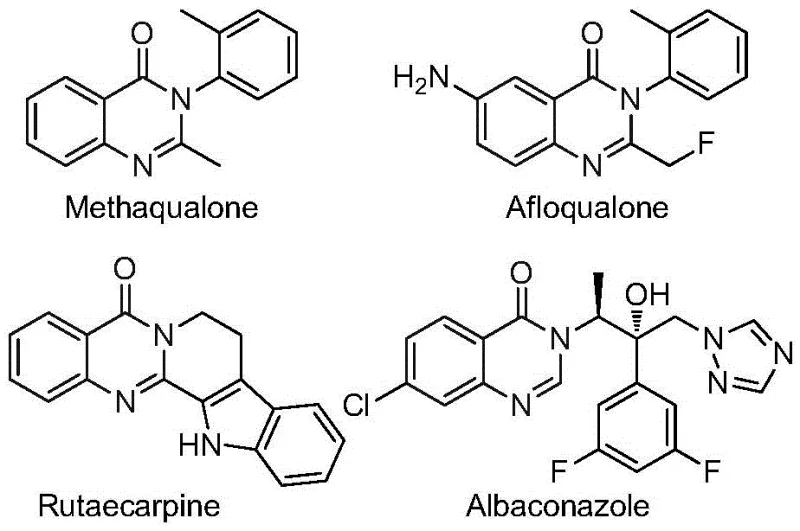
Quinazolinone compounds represent a critical class of condensed nitrogen-containing six-membered heterocycles found in many functional molecules. The biological significance of this scaffold cannot be overstated, with market-approved drugs like methaqualone and various kinase inhibitors relying on this core structure. However, the traditional synthesis of these derivatives often involves multi-step sequences with poor atom economy. The innovation presented in CN112480015B addresses these historical bottlenecks by utilizing cheap and readily available nitro compounds and trifluoroethylimidoyl chloride as starting materials. This approach not only simplifies the synthetic route but also opens new avenues for cost reduction in API manufacturing by minimizing waste and operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone ring system has been plagued by significant technical and economic hurdles. Conventional synthetic strategies often rely on harsh reaction conditions that pose severe safety risks in an industrial setting. For instance, earlier methods frequently necessitated the use of high-pressure carbon monoxide gas, which requires specialized autoclaves and rigorous safety protocols to prevent leakage and exposure. Furthermore, many established protocols utilize expensive noble metal catalysts such as ruthenium or platinum, which drastically inflate the raw material costs and complicate the downstream purification process due to heavy metal residue concerns. Additionally, some routes require pre-activated substrates like acid anhydrides or 2-bromoformylanilines, which are not only costly to produce but also generate stoichiometric amounts of waste, thereby negatively impacting the overall environmental footprint of the synthesis.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN112480015B introduces a paradigm shift through a palladium-catalyzed carbonylation cascade reaction. This novel approach elegantly bypasses the need for gaseous carbon monoxide by employing molybdenum hexacarbonyl (Mo(CO)6) as a solid, easy-to-handle CO surrogate. The reaction proceeds under relatively mild thermal conditions (120°C) in a common organic solvent like 1,4-dioxane, eliminating the need for high-pressure equipment. By merging the reduction of the nitro group, the formation of the amidine intermediate, and the final cyclization into a single pot, this method achieves remarkable step economy. This streamlined process directly translates to commercial scale-up of complex pharmaceutical intermediates, offering a practical solution for producing high-purity targets with minimal operational overhead.
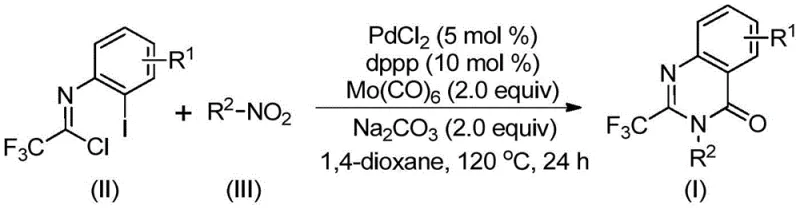
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
To fully appreciate the sophistication of this synthesis, one must delve into the intricate mechanistic pathway that drives the transformation. The reaction is believed to initiate with the reduction of the nitro compound to the corresponding amine by Mo(CO)6, which simultaneously serves as the carbon monoxide source. Following this reduction, a base-promoted intermolecular carbon-nitrogen bond coupling occurs between the newly formed amine and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine derivative in situ. The palladium catalyst then plays a pivotal role by inserting into the carbon-iodine bond of the imidoyl chloride moiety, forming a reactive divalent palladium intermediate. Subsequently, carbon monoxide released from the molybdenum complex inserts into the carbon-palladium bond to create an acyl-palladium species.
The final stages of the catalytic cycle involve the intramolecular nucleophilic attack of the nitrogen atom onto the acyl-palladium center, facilitated by the base, which closes the ring to form a seven-membered cyclic palladium intermediate. The cycle concludes with a reductive elimination step that releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This mechanism highlights the exquisite chemoselectivity of the system, as it tolerates a wide array of functional groups without interference. The ability to accommodate diverse substituents on both the nitro compound and the imidoyl chloride precursor ensures that this methodology is versatile enough for the synthesis of complex libraries required in modern drug discovery.
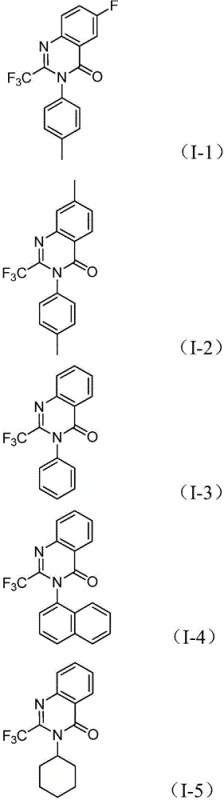
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to ensure maximum yield and purity. The process is designed to be user-friendly, utilizing standard Schlenk techniques and commercially available reagents. The key to success lies in the precise stoichiometric balance of the catalyst system and the choice of solvent, which profoundly influences the reaction kinetics. While the procedure is robust, maintaining an inert atmosphere and controlling the temperature profile are critical for reproducibility. For detailed operational instructions, please refer to the standardized guide below which breaks down the exact quantities and timing required for optimal results.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers compelling advantages that extend far beyond simple chemical transformation. The primary driver for value creation here is the drastic simplification of the supply chain logistics. By utilizing nitro compounds and trifluoroethylimidoyl chloride as starting materials, manufacturers can source inputs from a broad base of commodity chemical suppliers, thereby mitigating the risk of single-source dependency. This abundance of raw materials ensures a stable supply chain, which is crucial for maintaining continuous production schedules in the face of global market volatility. Furthermore, the elimination of high-pressure gas cylinders reduces the regulatory burden and insurance costs associated with hazardous material storage.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the replacement of expensive noble metal catalysts with a more economical palladium system and the use of solid CO surrogates. By avoiding the capital expenditure associated with high-pressure reactors and the operational costs of handling toxic gases, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the one-pot nature of the reaction minimizes solvent usage and workup time, leading to reduced utility consumption and labor costs. These efficiencies accumulate to provide a highly competitive pricing structure for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of this chemistry translates directly into supply chain resilience. Because the reaction tolerates a wide range of functional groups and impurities better than sensitive multi-step sequences, the quality of incoming raw materials is less critical, allowing for greater flexibility in vendor selection. The scalability of the method from gram to kilogram scales without significant re-optimization means that production can be ramped up quickly to meet sudden spikes in demand. This agility is a vital asset for pharmaceutical companies aiming to accelerate their time-to-market for new drug candidates.
- Scalability and Environmental Compliance: In an era of increasing environmental scrutiny, the green chemistry attributes of this method are a major selling point. The avoidance of stoichiometric heavy metal waste and the use of a closed system for CO generation align with strict environmental, health, and safety (EHS) regulations. The simplified post-treatment process, involving basic filtration and chromatography, reduces the volume of chemical waste generated per kilogram of product. This not only lowers waste disposal costs but also enhances the sustainability profile of the manufacturing process, appealing to eco-conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for R&D teams evaluating this route for their specific projects. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What are the advantages of this method over traditional high-pressure carbonylation?
A: Unlike conventional methods requiring hazardous high-pressure carbon monoxide gas and expensive ruthenium or platinum catalysts, this protocol utilizes solid Mo(CO)6 as a safe CO surrogate and operates under standard atmospheric pressure, significantly reducing safety risks and equipment costs.
Q: What is the substrate scope for the nitro compounds in this reaction?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents on the nitro compound including alkyl groups, halogens (F, Cl, Br), and trifluoromethyl groups at ortho, meta, or para positions, allowing for diverse library synthesis.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method is scalable to gram levels and beyond. The use of commercially available, cheap starting materials and a simple one-pot operation without specialized high-pressure reactors makes it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN112480015B. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate performs consistently in your downstream applications. Our infrastructure is designed to handle complex heterocyclic chemistry with the highest standards of safety and quality.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive efficiency and reliability in your supply chain.
