Revolutionizing Drospirenone Intermediate Production: A Deep Dive into Solvent-Free Mechanochemical Bromination
Revolutionizing Drospirenone Intermediate Production: A Deep Dive into Solvent-Free Mechanochemical Bromination
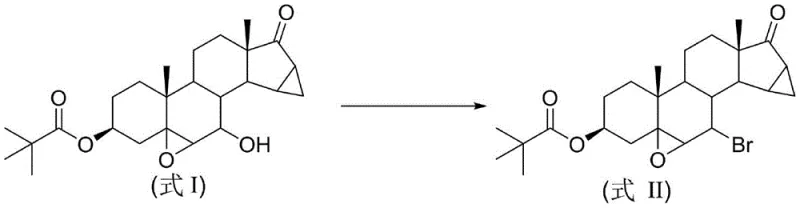
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more efficient manufacturing processes, particularly in the synthesis of complex steroidal intermediates. Patent CN114437169B, published in April 2023, introduces a groundbreaking methodology for the preparation of 3-pivaloyloxy-5β,6β-epoxy-7-bromo-15β,16β-dimethylene-pregn-17-one, a critical precursor in the production of drospirenone. This novel approach leverages mechanochemical ball milling technology to achieve high-yield bromination without the reliance on bulk organic solvents, addressing long-standing challenges regarding waste generation and process safety. By transitioning from traditional solution-phase chemistry to a solid-state mechanical activation regime, this innovation offers a compelling value proposition for manufacturers seeking to optimize their production lines for both economic and environmental performance.
The significance of this patent extends beyond mere academic interest; it represents a tangible solution for the commercial scale-up of complex pharmaceutical intermediates. The method utilizes a synergistic combination of Lewis acid catalysis and mechanical force to drive the substitution of the hydroxyl group with a bromine atom. This eliminates the need for hazardous reagents like sulfonyl chlorides or phosphorus-based catalysts that typically generate difficult-to-treat waste streams. For R&D directors and process chemists, this opens a new avenue for designing robust, scalable routes that align with modern green chemistry principles while maintaining the rigorous purity standards required for hormonal active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key drospirenone intermediate has relied heavily on solution-phase chemistry involving sulfonylation followed by bromination, or direct bromination catalyzed by triphenylphosphine. These traditional pathways suffer from inherent inefficiencies that burden both the operational budget and the environmental compliance team. The requirement for large volumes of organic solvents not only inflates raw material costs but also necessitates expensive recovery and distillation infrastructure to meet regulatory emission standards. Furthermore, the use of phosphorus-based reagents generates substantial amounts of phosphorus-containing wastewater, which poses significant treatment challenges and increases the overall ecological footprint of the manufacturing facility.
Another critical drawback of conventional methods is the issue of selectivity and purification. Solution-phase reactions often struggle with side reactions that lead to complex impurity profiles, requiring multiple chromatographic or recrystallization steps to achieve the necessary pharmaceutical grade purity. This multi-step purification process results in yield losses and extended cycle times, directly impacting the reducing lead time for high-purity pharmaceutical intermediates. The reliance on aqueous workups for product isolation further complicates the process, generating saline waste streams that require specialized disposal protocols. These cumulative factors make traditional synthesis routes increasingly unsustainable in a market that demands both cost-efficiency and environmental stewardship.
The Novel Approach
In stark contrast, the method disclosed in patent CN114437169B utilizes a solvent-free mechanochemical strategy that fundamentally redefines the reaction environment. By employing a ball mill to mechanically activate the solid reactants—specifically the steroid substrate, a Lewis acid, a brominating salt, and silica gel—the process achieves intimate molecular contact without the need for a liquid medium. This "dry" reaction condition effectively suppresses many of the side reactions common in solution chemistry, leading to improved selectivity and a cleaner crude product profile. The absence of bulk solvents during the reaction phase dramatically reduces the volume of hazardous waste generated, aligning perfectly with the principles of atom economy and waste prevention.
The novelty of this approach also lies in its operational simplicity and versatility. The reaction proceeds efficiently at room temperature, eliminating the energy costs associated with heating or cooling large reaction vessels. The use of silica gel as a grinding auxiliary not only enhances the mechanical energy transfer but also serves as a convenient matrix for post-reaction processing. Instead of complex aqueous extractions, the product is recovered through a simple solvent soaking and filtration step, followed by recrystallization. This streamlined workflow significantly shortens the production cycle and reduces the demand for specialized equipment, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing while ensuring consistent product quality.
Mechanistic Insights into Lewis Acid-Catalyzed Mechanochemical Bromination
The core of this innovative synthesis lies in the unique interplay between mechanical force and Lewis acid catalysis within a solid-state matrix. In the absence of solvent molecules to solvate the ions, the mechanical energy provided by the colliding stainless steel balls serves to continuously expose fresh reactive surfaces and lower the activation energy barrier for the nucleophilic substitution. The Lewis acid catalyst, such as ferric chloride or ytterbium triflate, coordinates with the hydroxyl group of the steroid substrate, activating it for displacement by the bromide ion provided by reagents like lithium bromide or tribromopyridinium salts. This coordination is facilitated by the high local pressures and temperatures generated transiently at the impact sites of the milling media, promoting a reaction pathway that might be kinetically inaccessible under standard thermal conditions.
From an impurity control perspective, the solid-state nature of the reaction imposes a degree of steric constraint that favors the desired transformation over competing degradation pathways. The silica gel support plays a crucial role in this mechanism by adsorbing the reactants and preventing the aggregation of particles, thereby ensuring uniform reaction progress throughout the bulk material. This homogeneity is critical for minimizing the formation of regioisomers or elimination byproducts that often plague solution-phase brominations of sensitive steroidal skeletons. The result is a reaction profile characterized by high conversion rates and exceptional selectivity, yielding a product that requires minimal downstream purification to meet stringent specifications.
Furthermore, the choice of Lewis acid allows for fine-tuning of the reaction kinetics to match specific production requirements. The patent data indicates that rare earth triflates like ytterbium trifluoromethanesulfonate can achieve yields as high as 94% under optimized milling frequencies, demonstrating the robustness of the catalytic system. This mechanistic flexibility ensures that the process can be adapted to different scales and equipment configurations without compromising the integrity of the molecular structure. For technical teams, understanding this mechanism provides the confidence to implement the process with predictable outcomes, reducing the risk of batch failures and ensuring a stable supply of the critical intermediate.
How to Synthesize 3-pivaloyloxy-5β,6β-epoxy-7-bromo-15β,16β-dimethylene-pregn-17-one Efficiently
Implementing this mechanochemical route requires precise control over milling parameters and reagent ratios to maximize efficiency. The process begins with the careful loading of the ball mill tank with the stoichiometric amounts of the hydroxy-steroid precursor, the chosen Lewis acid catalyst, and the brominating source, along with a significant mass of silica gel to act as the grinding auxiliary. Stainless steel balls of specific diameters (typically 5-10mm) are added to provide the necessary impact energy. The detailed standardized synthesis steps, including specific molar ratios and milling frequencies derived from the patent examples, are outlined below to guide your process development team in replicating these high-yield results.
- Charge a ball milling tank with the starting material (Formula I), a Lewis acid catalyst (e.g., FeCl3 or Yb(OTf)3), a brominating reagent (e.g., LiBr), silica gel, and stainless steel grinding balls.
- Operate the ball mill at a frequency between 5Hz and 20Hz for a duration of 0.5 to 2 hours at room temperature to facilitate the solid-state substitution reaction.
- Transfer the reaction mixture, soak in a minimal amount of organic solvent (e.g., ethyl acetate), filter to remove silica and balls, concentrate the filtrate, and recrystallize to obtain the pure bromide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this mechanochemical synthesis route offers transformative benefits that extend far beyond the laboratory bench. The elimination of bulk organic solvents during the reaction phase translates directly into a substantial reduction in raw material procurement costs and logistics overhead. Without the need to purchase, store, and handle large volumes of flammable solvents, facilities can lower their insurance premiums and reduce the complexity of their hazardous material management protocols. This simplification of the supply chain enhances overall operational resilience, making the production of this critical intermediate less vulnerable to fluctuations in solvent markets or supply disruptions.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the drastic simplification of the workflow and the removal of solvent recovery units. By avoiding the energy-intensive distillation steps required to recycle solvents in traditional methods, manufacturers can achieve significant savings in utility costs. Additionally, the high atom economy of the Lewis acid-catalyzed reaction minimizes the loss of valuable starting materials to side products, effectively increasing the overall yield per batch. The use of inexpensive and readily available reagents like silica gel and simple bromide salts further contributes to a lower cost of goods sold (COGS), providing a competitive edge in the pricing of the final API.
- Enhanced Supply Chain Reliability: The robustness of the solid-state reaction conditions ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream API manufacturers. The process is less sensitive to moisture and oxygen compared to some solution-phase alternatives, reducing the risk of batch rejection due to environmental factors. Furthermore, the simplified post-treatment procedure, which relies on filtration and recrystallization rather than complex extractions, shortens the manufacturing cycle time. This agility allows suppliers to respond more rapidly to changes in market demand, ensuring that customers receive their orders of high-purity intermediates without delay.
- Scalability and Environmental Compliance: From a regulatory standpoint, this method offers a clear path to sustainable manufacturing. The significant reduction in three-waste emissions (waste water, waste gas, and solid waste) simplifies compliance with increasingly stringent environmental regulations. The absence of phosphorus-containing waste streams removes a major hurdle in wastewater treatment, lowering the operational burden on environmental health and safety (EHS) teams. As the industry moves towards greener chemistry mandates, adopting this technology positions manufacturers as leaders in sustainability, potentially unlocking preferential status with eco-conscious partners and regulators alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this mechanochemical bromination technology. These answers are derived directly from the experimental data and claims presented in patent CN114437169B, providing a reliable foundation for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this process into existing production lines or for sourcing this intermediate from external partners who utilize this advanced methodology.
Q: What are the primary environmental benefits of this mechanochemical method compared to traditional solution-phase synthesis?
A: The mechanochemical method described in patent CN114437169B eliminates the need for large volumes of organic solvents during the reaction phase, drastically reducing VOC emissions and solvent waste. Furthermore, it avoids the generation of phosphorus-containing waste liquids associated with traditional triphenylphosphine-catalyzed bromination, making the process significantly greener and easier to treat.
Q: Which Lewis acid catalysts are compatible with this solid-state bromination process?
A: The patent specifies several effective Lewis acids that can catalyze this transformation under mechanical grinding conditions, including ferric chloride (FeCl3), aluminum chloride (AlCl3), ytterbium trifluoromethanesulfonate (Yb(OTf)3), and lanthanum trifluoromethanesulfonate (La(OTf)3), offering flexibility in catalyst selection based on availability and cost.
Q: How does the use of silica gel impact the reaction efficiency and post-treatment?
A: Silica gel acts as a solid support and grinding auxiliary, improving the dispersion of reactants and facilitating energy transfer during ball milling. Its presence simplifies post-treatment, as the product can be easily separated from the silica matrix via solvent soaking and filtration, avoiding complex aqueous workups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-pivaloyloxy-5β,6β-epoxy-7-bromo-15β,16β-dimethylene-pregn-17-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively analyzed the potential of mechanochemical processes like the one described in CN114437169B and possesses the expertise to translate these laboratory innovations into robust commercial realities. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot-scale quantities or full industrial volumes, our facilities are equipped to deliver. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how this green synthesis route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a sustainable, cost-effective, and reliable supply of this vital drospirenone intermediate.
