Advanced Two-Component Chiral Resolution for High-Purity (S)-(+)-Ibuprofen Manufacturing
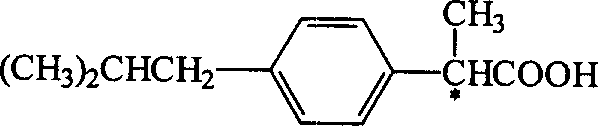
The pharmaceutical industry continuously seeks more efficient pathways to produce high-value chiral intermediates, and the technology disclosed in patent CN1314336A represents a significant leap forward in the manufacturing of non-steroidal anti-inflammatory drugs (NSAIDs). This patent details a novel method for splitting racemic ibuprofen mixtures using a sophisticated two-component chiral resolving agent system, specifically targeting the isolation of the biologically active (S)-(+)-enantiomer. Unlike traditional methods that rely on single-component agents and suffer from long cycle times and moderate purity, this innovation utilizes a synergistic combination of chiral amino alcohols and chiral arylamines or amino acids to drive the formation of diastereomeric salts with superior selectivity. The structural integrity and chiral center of the target molecule, as illustrated below, are critical for its pharmacological efficacy, being approximately 160 times more active than its (R)-(-)-counterpart.
For R&D directors and process chemists, the significance of this patent lies in its ability to deliver optical purities ranging from 95% to 99% in a single crystallization step, with yields around 40% of the starting racemate. This level of purity is essential for meeting the stringent regulatory requirements of modern pharmaceutical markets, where the presence of the inactive or potentially toxic enantiomer is increasingly scrutinized. By leveraging this two-component strategy, manufacturers can bypass the complexities of asymmetric synthesis, which often involves expensive catalysts and difficult-to-source chiral pools, opting instead for a robust resolution process that is easier to control and scale within existing infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of racemic ibuprofen has been plagued by inefficiencies inherent to single-component resolving agents, such as (S)-phenylethylamine or L-lysine, which often require extended processing times ranging from 20 to 40 hours to achieve acceptable separation. These traditional processes frequently result in suboptimal optical purities, often hovering between 76% and 90%, necessitating multiple recrystallization steps that erode overall yield and increase solvent consumption. Furthermore, the slow crystallization kinetics associated with single-agent systems can lead to operational bottlenecks in large-scale reactors, limiting batch turnover rates and increasing energy costs associated with prolonged heating and stirring. The complexity of recovering and recycling these single agents is also often high, leading to increased waste generation and higher raw material costs per kilogram of final product.
The Novel Approach
In stark contrast, the method described in CN1314336A introduces a dual-agent system that fundamentally alters the thermodynamics and kinetics of the crystallization process, reducing the total resolution cycle to merely 1-7 hours. By sequentially or simultaneously introducing a chiral amino alcohol (Component A) and a chiral arylamine or amino acid (Component B), the process creates a unique chemical environment where the diastereomeric salt of the desired (S)-(+)-enantiomer precipitates rapidly and with high selectivity. This approach not only simplifies the operational workflow by eliminating the need for multiple purification stages but also enhances the physical properties of the precipitate, making filtration and drying significantly more efficient. The ability to complete the entire resolution in under a day represents a transformative improvement in throughput, allowing manufacturing facilities to maximize asset utilization and respond more agilely to market demand fluctuations.
Mechanistic Insights into Two-Component Chiral Resolution
The core mechanism driving this enhanced performance is the cooperative interaction between the two distinct chiral resolving agents and the carboxylic acid group of the ibuprofen molecule. Component A, typically a chiral amino alcohol like norephedrine or a nitrophenyl-propanediol derivative, acts as a primary selector that initiates the formation of a hydrogen-bonded network, while Component B, such as alpha-phenylethylamine or an amino acid, reinforces the stereochemical discrimination through additional steric and electronic interactions. This dual-interaction model stabilizes the crystal lattice of the (S)-(+)-ibuprofen salt far more effectively than the (R)-(-)-salt, creating a large solubility differential that drives the selective precipitation of the target isomer from the mixed solvent system. The precise tuning of the molar ratios, typically between 0.5 to 1 equivalent of each agent relative to the substrate, allows chemists to fine-tune the supersaturation levels, ensuring that nucleation occurs exclusively for the desired diastereomer.
Impurity control is inherently built into this mechanistic framework, as the rapid and selective crystallization prevents the occlusion of the unwanted (R)-enantiomer within the growing crystal lattice, a common issue in slower, diffusion-limited processes. The use of a mixed solvent system, comprising a non-polar hydrocarbon like petroleum ether or cyclohexane combined with a polar modifier like an amine or alcohol, further refines the solubility profile, ensuring that the impurities remain in the mother liquor while the pure product crashes out. Post-crystallization treatment with saturated sodium carbonate solution effectively liberates the free acid from the salt complex, and subsequent acidification with dilute hydrochloric acid precipitates the final (S)-(+)-ibuprofen with minimal contamination. This robust mechanism ensures that the final product consistently meets the high purity standards required for downstream API synthesis without the need for extensive chromatographic purification.
How to Synthesize (S)-(+)-Ibuprofen Efficiently
The practical implementation of this resolution technology involves a straightforward sequence of mixing, heating, and crystallization steps that are highly amenable to standard chemical engineering equipment. The process begins by suspending the racemic ibuprofen with the first resolving agent in a heated mixed solvent, followed by the addition of the second agent to trigger the selective salt formation. Detailed operational parameters, including specific temperature ramps, stirring speeds, and filtration protocols, are critical for reproducing the high yields and purities reported in the patent data. For process engineers looking to adopt this technology, understanding the precise interplay between solvent polarity and agent stoichiometry is key to optimizing the batch cycle time and minimizing solvent waste.
- Suspend racemic ibuprofen with Component A (chiral amino alcohol) in a mixed solvent system and heat to reflux for 0.5-3 hours to initiate interaction.
- Add Component B (chiral arylamine or amino acid) to the mixture and continue refluxing for 0.5-2 hours to form the less soluble diastereomeric salt of (S)-(+)-ibuprofen.
- Cool the solution to precipitate crystals, filter, and treat the salt with saturated Na2CO3 followed by acidification to isolate pure (S)-(+)-ibuprofen.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this two-component resolution technology offers profound advantages for procurement managers and supply chain leaders focused on cost optimization and reliability. The drastic reduction in processing time from days to hours translates directly into lower utility costs and higher facility throughput, allowing manufacturers to produce more material with the same fixed assets. Additionally, the patent explicitly highlights the recyclability of both the resolving agents and the solvents, which significantly reduces the variable cost of goods sold (COGS) by minimizing the need for fresh raw material purchases and lowering waste disposal fees. This circular approach to solvent and reagent management aligns perfectly with modern sustainability goals while providing a tangible buffer against volatile raw material pricing in the global chemical market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to recycle chiral auxiliaries create a leaner cost structure that is less sensitive to fluctuations in precious metal markets. By avoiding the complex workup procedures associated with asymmetric synthesis, such as heavy metal scavenging, manufacturers can realize substantial savings in both consumables and labor hours. The high yield of the primary crystallization step further contributes to cost efficiency by maximizing the output from each batch of starting racemic material, ensuring that capital is not tied up in low-yielding processes.
- Enhanced Supply Chain Reliability: The reliance on domestically available and chemically stable resolving agents, such as simple amino alcohols and arylamines, mitigates the risk of supply disruptions often associated with exotic chiral ligands. The robustness of the process, characterized by its wide operating window and tolerance to minor variations in conditions, ensures consistent production schedules and reliable delivery timelines for downstream customers. This stability is crucial for maintaining continuous supply lines to API manufacturers who cannot afford interruptions in their own production schedules due to intermediate shortages.
- Scalability and Environmental Compliance: The simplicity of the unit operations involved—primarily reflux, filtration, and acid-base extraction—makes this technology exceptionally easy to scale from pilot plant to multi-ton commercial production without significant re-engineering. Furthermore, the ability to recover and reuse solvents reduces the overall environmental footprint of the manufacturing process, facilitating compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and liquid waste discharge. This ease of scale-up and regulatory alignment makes it an attractive option for companies looking to expand their capacity for high-purity chiral intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, derived directly from the patent specifications and industrial best practices. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the two-component system over legacy methods, focusing on purity, speed, and economic viability.
Q: What is the optical purity achievable with this two-component resolution method?
A: According to patent CN1314336A, this method consistently achieves an optical purity of 95-99% for (S)-(+)-ibuprofen after a single crystallization step, significantly outperforming traditional single-agent methods which often yield lower purities around 76-90%.
Q: How does this process improve production efficiency compared to conventional resolution?
A: The novel two-component approach drastically reduces the resolution cycle time from the conventional 20-40 hours down to just 1-7 hours. This acceleration is due to faster crystallization kinetics and simplified operational steps, allowing for higher throughput in manufacturing facilities.
Q: Can the resolving agents and solvents be recovered to reduce costs?
A: Yes, a key economic advantage of this technology is the recyclability of both the resolving agents and the solvent systems. The mother liquor containing the unwanted enantiomer can also be racemized and reprocessed, maximizing material utilization and minimizing waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-(+)-Ibuprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral resolution technologies in the modern pharmaceutical supply chain and possess the technical expertise to bring patents like CN1314336A to life on an industrial scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high purity and rapid cycle times are fully realized in actual manufacturing runs. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-(+)-ibuprofen intermediate meets the exacting standards required by global regulatory bodies, providing our partners with absolute confidence in product quality.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our optimized version of this two-component resolution process, we can help you secure a stable supply of high-purity intermediates while significantly reducing your overall manufacturing costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced capabilities can enhance your supply chain resilience.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
