Advanced Heterogeneous Catalysis for Scalable 2-Trifluoromethyl Benzimidazole Production
Advanced Heterogeneous Catalysis for Scalable 2-Trifluoromethyl Benzimidazole Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes for synthesizing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN113429349A, which discloses a novel preparation method for 2-trifluoromethyl substituted benzimidazole compounds. This technology leverages a heterogeneous copper-doped carbon nitride catalyst to drive the cyclization of trifluoroethylimidoyl chloride and amines. For R&D directors and procurement specialists, this represents a pivotal shift away from traditional homogeneous catalysis, offering a pathway to high-purity intermediates with simplified downstream processing. The introduction of the trifluoromethyl group is critical for enhancing the metabolic stability and lipophilicity of drug candidates, making this synthetic route highly valuable for the development of next-generation active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzimidazole core, particularly with trifluoromethyl substitution, has relied on methodologies that present significant operational and economic challenges. Traditional routes often involve the condensation of o-phenylenediamine with trifluoroacetic acid or aldehydes, or the reductive cyclization of o-nitroanilines. These processes frequently suffer from severe reaction conditions, requiring high temperatures or strong acidic environments that can degrade sensitive functional groups. Furthermore, earlier reports utilizing homogeneous copper catalysts, while effective in promoting cascade cyclizations, introduce a major bottleneck in manufacturing: catalyst removal. Separating trace amounts of soluble copper species from the final product requires expensive scavenging resins or complex extraction protocols, which drastically increases the cost of goods sold (COGS) and complicates the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in the patent data overcomes these hurdles by employing a robust heterogeneous catalytic system. By utilizing copper-doped graphitic carbon nitride (Cu/g-C3N4), the reaction proceeds under remarkably mild conditions, typically between 70°C and 90°C. This thermal flexibility allows for the preservation of delicate substituents on the aromatic ring, thereby widening the scope of accessible chemical space. The heterogeneous nature of the catalyst means it can be physically separated from the reaction mixture via simple filtration, eliminating the need for costly metal removal steps. This innovation not only streamlines the workflow but also aligns with green chemistry principles by enabling catalyst recycling, thus reducing the overall consumption of precious metal resources and minimizing hazardous waste generation in the manufacturing of complex polymer additives or electronic chemicals.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic substitution and transition metal catalysis. Initially, the trifluoroethylimidoyl chloride undergoes a nucleophilic addition-elimination reaction with the amine substrate to form an amidine intermediate. This step is crucial as it sets the stage for the subsequent ring closure. The copper species embedded within the carbon nitride matrix then plays a pivotal role by inserting into the carbon-iodine bond of the aryl ring. This oxidative addition generates a reactive copper-aryl complex. Under the basic conditions provided by potassium carbonate, a molecule of hydrogen iodide is eliminated, facilitating the formation of a bivalent or trivalent cyclic copper intermediate. Finally, a reductive elimination step releases the desired 2-trifluoromethyl-substituted benzimidazole product and regenerates the active copper catalytic site, completing the cycle.
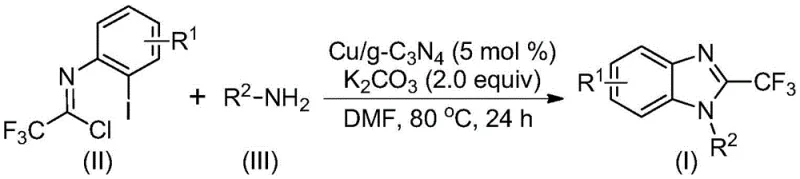
Understanding this mechanism is vital for impurity control. The use of a solid-supported catalyst helps to moderate the reactivity of the copper species, preventing over-reaction or non-selective coupling that often plagues homogeneous systems. The carbon nitride support acts not merely as a carrier but potentially participates in stabilizing the copper oxidation states, ensuring a consistent turnover frequency throughout the reaction duration of 18 to 30 hours. This controlled environment minimizes the formation of side products such as dehalogenated species or polymeric byproducts, resulting in a cleaner crude reaction profile that simplifies the final purification via column chromatography or crystallization.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
Implementing this synthesis requires careful attention to the preparation of the catalyst and the stoichiometry of the reagents. The process begins with the synthesis of the Cu/g-C3N4 catalyst, involving the dispersion of carbon nitride in DMF followed by doping with copper sulfate. Once the catalyst is prepared, the reaction is set up by combining the trifluoroethylimidoyl chloride, the chosen amine, potassium carbonate, and the catalyst in a solvent like DMF. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated by experimental data, are outlined below to ensure reproducibility and high yield.
- Prepare the copper-doped carbon nitride catalyst by dispersing carbon nitride in DMF, reacting with isonicotinic acid chloride, and subsequently doping with copper sulfate pentahydrate.
- Combine the trifluoroethylimidoyl chloride substrate, amine reactant, potassium carbonate base, and the prepared Cu/g-C3N4 catalyst in an organic solvent such as DMF.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter the heterogeneous catalyst and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this heterogeneous catalytic method offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the post-reaction workflow. By removing the necessity for heavy metal scavengers, manufacturers can significantly reduce the number of unit operations required, leading to shorter batch cycles and lower utility consumption. This efficiency translates directly into improved margin potential for high-value intermediates used in agrochemical or pharmaceutical applications. Furthermore, the reliance on cheap and commercially available starting materials, such as copper sulfate and carbon nitride, insulates the production cost from the volatility often seen with specialized ligand prices in homogeneous catalysis.
- Cost Reduction in Manufacturing: The economic impact of switching to this heterogeneous system is profound. Since the catalyst can be recovered and reused multiple times with only a partial reduction in efficiency, the effective cost per kilogram of catalyst usage drops precipitously. Additionally, the elimination of expensive metal scavenging resins and the reduction in solvent usage for extra wash steps contribute to substantial cost savings. The mild reaction conditions also imply lower energy requirements for heating and cooling, further optimizing the operational expenditure (OPEX) associated with large-scale production runs.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, non-sensitive reagents. Unlike air-sensitive homogeneous catalysts that require inert atmosphere handling and special storage, the copper-doped carbon nitride is stable and easy to handle. This robustness reduces the risk of batch failures due to catalyst degradation during storage or transport. Moreover, the broad substrate tolerance means that a single catalytic platform can be used to produce a wide variety of derivatives, allowing for greater flexibility in inventory management and faster response times to changing market demands for specific benzimidazole analogues.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat transfer and mixing challenges, but the mild thermal profile of this reaction (70-90°C) makes it highly amenable to scale-up in standard stainless steel reactors. From an environmental perspective, the ability to recycle the catalyst aligns with increasingly stringent global regulations regarding heavy metal discharge. Reducing the copper load in the waste stream simplifies wastewater treatment compliance, mitigating regulatory risks and avoiding potential fines, thereby ensuring a smoother, uninterrupted supply of critical intermediates to downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on catalyst performance, substrate compatibility, and purification strategies. Understanding these nuances is essential for process chemists aiming to integrate this technology into their existing manufacturing pipelines.
Q: Can the copper-doped carbon nitride catalyst be recycled?
A: Yes, the heterogeneous nature of the Cu/g-C3N4 catalyst allows for easy recovery via filtration. Experimental data indicates that the catalyst retains significant catalytic efficiency even after three recycling runs, with yields remaining high compared to the initial run.
Q: What is the substrate scope for this benzimidazole synthesis?
A: The method demonstrates broad substrate tolerance. It effectively accommodates various substituents on the aromatic ring (R1) such as hydrogen, methyl, chloro, bromo, and trifluoromethyl groups. Furthermore, it accepts diverse amine inputs (R2) including alkyl chains like n-butyl and n-pentyl, as well as benzyl and phenyl groups.
Q: Why is this method superior to traditional homogeneous copper catalysis?
A: Traditional methods often rely on homogeneous copper catalysts which are difficult to separate from the final product, leading to potential heavy metal contamination. This heterogeneous approach eliminates the need for complex metal scavenging steps, simplifies purification, and reduces the environmental footprint associated with metal waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in modern drug discovery and development. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing rigorous QC labs to verify every batch. Our capability to implement heterogeneous catalysis allows us to offer competitive pricing without compromising on quality or delivery timelines.
We invite you to collaborate with us to leverage this innovative synthesis for your project needs. Whether you require custom synthesis of specific benzimidazole derivatives or scale-up of existing routes, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can best support your supply chain goals.
