Industrial Scale Synthesis of 3,5-Dihydroxyacetophenone for Asthma Medication Intermediates
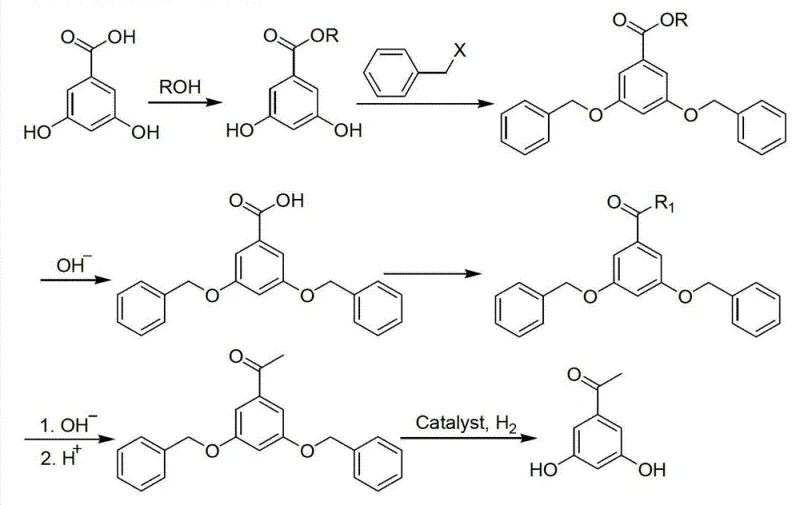
The pharmaceutical industry continuously seeks robust synthetic pathways for critical intermediates used in respiratory medications, specifically targeting compounds like 3,5-dihydroxyacetophenone which serves as a foundational building block for beta-agonists such as terbutaline and fenoterol. Patent CN102675075B introduces a refined preparation method that significantly diverges from traditional methodologies by utilizing 3,5-dihydroxybenzoic acid as the primary starting material. This specific intellectual property outlines a comprehensive six-step sequence involving esterification, benzylation, hydrolysis, nucleophilic substitution, hydrolytic decarboxylation, and final debenzylation. For R&D directors and procurement specialists evaluating supply chain resilience, this patent represents a pivotal shift towards more manageable reaction conditions that do not rely on sensitive organometallic reagents. The technical breakthrough lies in the strategic use of benzyl protection groups which allow for selective functionalization while maintaining the integrity of the phenolic rings throughout the harsher acylation stages. By adopting this methodology, manufacturers can achieve a more consistent impurity profile, which is crucial for regulatory compliance in the production of active pharmaceutical ingredients. The transition from laboratory scale to commercial manufacturing is facilitated by the use of common industrial solvents and catalysts, reducing the barrier to entry for large-scale production facilities aiming to secure a reliable pharmaceutical intermediate supplier for asthma medication components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dihydroxyacetophenone has been plagued by significant operational challenges associated with Grignard reagent methodologies. Conventional routes often require the use of highly reactive organomagnesium compounds which necessitate strictly anhydrous conditions and inert atmospheres, thereby increasing the complexity and cost of reactor setup and maintenance. Literature reviews indicate that previous attempts using 3,5-dihydroxybenzoic acid via etherification and subsequent Grignard ketonation have resulted in total yields ranging merely from 47% to 65%, which is suboptimal for cost-sensitive commercial manufacturing. These older processes frequently suffer from harsh reaction conditions that can lead to the formation of difficult-to-remove byproducts, complicating the downstream purification processes and potentially compromising the purity specifications required for pharmaceutical applications. Furthermore, the handling of Grignard reagents poses inherent safety risks on a large scale, including exothermic runaway potentials and sensitivity to moisture, which can disrupt production schedules and impact supply chain continuity. The environmental footprint of these traditional methods is also considerable, often generating substantial metal-containing waste streams that require specialized treatment before disposal, adding another layer of operational expense and regulatory burden for chemical manufacturers.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a protective group strategy that circumvents the need for sensitive organometallic chemistry, thereby simplifying the operational workflow significantly. The process initiates with a straightforward esterification followed by benzylation, which effectively masks the reactive hydroxyl groups, allowing for subsequent nucleophilic substitution under much milder conditions. This pathway enables the use of common nucleophiles such as diethyl malonate or ethyl acetoacetate, which are readily available and cost-effective compared to specialized Grignard reagents. The reaction conditions are notably温和 (mild), typically operating at reflux temperatures in standard solvents like acetone or ethanol, which are easier to recover and recycle in a closed-loop manufacturing system. By eliminating the requirement for ultra-dry environments, the novel approach reduces the capital expenditure needed for specialized drying equipment and nitrogen blanketing systems. This shift not only enhances the safety profile of the manufacturing plant but also improves the overall atom economy of the synthesis. The final steps involve hydrolytic decarboxylation and catalytic hydrogenation, both of which are well-understood unit operations in the fine chemical industry, ensuring that the technology transfer from pilot plant to full-scale production is seamless and reliable for any cost reduction in pharmaceutical intermediate manufacturing initiative.
Mechanistic Insights into Benzyl-Protected Nucleophilic Substitution
The core chemical innovation of this synthesis lies in the meticulous management of reactivity through the temporary installation of benzyl protecting groups on the phenolic hydroxyls. In the second step of the process, the 3,5-dihydroxybenzoic acid ester reacts with a benzylation reagent, such as benzyl chloride or bromide, in the presence of an acid-binding agent like potassium carbonate. This transformation is critical because free phenolic hydroxyl groups are highly nucleophilic and could interfere with the subsequent acylation or nucleophilic substitution steps, leading to polymerization or oligomerization side products. By converting the hydroxyls to benzyl ethers, the electron density of the aromatic ring is modulated, and the oxygen atoms are rendered inert to the basic and nucleophilic conditions employed in the fourth step. The nucleophilic substitution involves the reaction of the activated acid chloride derivative with a carbon nucleophile, specifically a malonate or acetoacetate derivative, facilitated by a Lewis acid catalyst such as magnesium chloride hexahydrate. This specific catalytic system promotes the formation of the beta-keto ester or beta-diketone intermediate with high regioselectivity, ensuring that the acyl group attaches precisely at the desired position without affecting the protected ether linkages. The mechanistic precision here is vital for R&D teams focused on impurity谱 (impurity profiles), as it minimizes the generation of structural isomers that are notoriously difficult to separate during crystallization.
Following the construction of the carbon skeleton, the removal of the protecting groups and the carboxyl moiety is achieved through a controlled hydrolytic decarboxylation sequence. The fifth step involves refluxing the intermediate in an aqueous alkali solution, which hydrolyzes the ester functionality and facilitates the loss of carbon dioxide from the beta-keto acid intermediate. This decarboxylation is thermodynamically driven and proceeds cleanly under the specified pH conditions, typically adjusted to acidic levels after the initial base hydrolysis. The final debenzylation is accomplished via catalytic hydrogenation using metals such as Pd/C or Raney nickel under hydrogen pressure. This step is particularly elegant as it simultaneously removes the benzyl protecting groups to regenerate the free phenolic hydroxyls without reducing the ketone functionality, provided the catalyst and pressure are carefully selected. The selectivity of this hydrogenation is a key quality attribute, ensuring that the final 3,5-dihydroxyacetophenone retains its keto group intact while restoring the pharmacophore necessary for downstream coupling reactions. Understanding these mechanistic nuances allows process chemists to optimize reaction times and catalyst loadings, further driving down the cost of goods sold while maintaining the high-purity pharmaceutical intermediate standards required by global regulatory bodies.
How to Synthesize 3,5-Dihydroxyacetophenone Efficiently
The efficient synthesis of this critical intermediate relies on a disciplined adherence to the six-step protocol outlined in the patent, beginning with the esterification of 3,5-dihydroxybenzoic acid in a low molecular weight alcohol solvent. Operators must ensure that the molar ratio of alcohol to acid is maintained within the specified range to drive the equilibrium towards the ester product, followed by a rigorous workup involving extraction and drying to remove residual acid catalyst. The subsequent benzylation step requires careful temperature control during the addition of the benzyl halide to manage the exotherm and ensure complete conversion of the phenolic groups. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Esterification of 3,5-dihydroxybenzoic acid using low molecular alcohol and concentrated sulfuric acid under reflux.
- Benzylation of the ester using benzyl halides and acid-binding agents in acetone to protect hydroxyl groups.
- Hydrolysis, nucleophilic substitution with malonate derivatives, hydrolytic decarboxylation, and final catalytic debenzylation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits primarily driven by the simplification of raw material sourcing and the reduction of hazardous waste handling costs. The elimination of Grignard reagents removes the dependency on suppliers of sensitive organometallic chemicals, which often command premium pricing and require specialized logistics for safe transport and storage. Instead, the process relies on commodity chemicals such as ethanol, acetone, and benzyl chloride, which are available from multiple vendors globally, thereby enhancing supply chain reliability and reducing the risk of single-source bottlenecks. The mild reaction conditions also translate to lower energy consumption, as the process does not require cryogenic cooling or extreme heating, contributing to a reduced carbon footprint and alignment with modern sustainability goals in chemical manufacturing. Furthermore, the high yields reported in the patent examples suggest a more efficient utilization of starting materials, which directly correlates to lower raw material costs per kilogram of finished product. For supply chain heads, the robustness of this method means fewer batch failures and less downtime associated with cleaning reactors contaminated by metal residues, ensuring a more consistent flow of materials to downstream API production lines.
- Cost Reduction in Manufacturing: The economic advantage of this process is largely derived from the avoidance of expensive and hazardous reagents while utilizing standard industrial equipment that does not require exotic metallurgy or passivation. By replacing complex organometallic steps with conventional acid-base chemistry and catalytic hydrogenation, manufacturers can significantly reduce the operational expenditure associated with safety protocols and waste disposal. The ability to recycle solvents like acetone and ethanol further amplifies these savings, creating a circular economy within the production facility that minimizes fresh solvent purchases. Additionally, the higher overall yield means that less raw material is wasted, effectively lowering the cost basis for every unit of 3,5-dihydroxyacetophenone produced. This efficiency allows companies to offer more competitive pricing to their clients while maintaining healthy profit margins, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical intermediates is paramount for pharmaceutical companies, and this synthesis route enhances reliability by diversifying the input material base. Since the starting material, 3,5-dihydroxybenzoic acid, is a widely produced chemical, the risk of supply disruption due to raw material scarcity is minimized compared to routes relying on niche reagents. The process tolerance to minor variations in reaction conditions also means that manufacturing can continue smoothly even if there are slight fluctuations in utility quality or ambient conditions, reducing the likelihood of batch rejections. This robustness ensures that procurement managers can forecast inventory levels with greater confidence, knowing that the production timeline is less susceptible to technical delays. Consequently, lead times for high-purity pharmaceutical intermediates can be optimized, allowing downstream manufacturers to maintain leaner inventory buffers without risking production stoppages.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden complexities, but this method is designed with commercial scale-up of complex pharmaceutical intermediates in mind. The unit operations involved, such as reflux, filtration, and distillation, are standard across the industry, meaning that existing infrastructure can often be utilized without major capital investment. From an environmental standpoint, the absence of heavy metal waste from Grignard reagents simplifies effluent treatment and reduces the regulatory burden associated with hazardous waste disposal. The use of catalytic hydrogenation in the final step is also cleaner than stoichiometric reduction methods, generating water as the primary byproduct rather than toxic sludge. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for environmentally conscious global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis route over Grignard methods?
A: This route avoids harsh Grignard conditions, offering milder reaction parameters, higher overall yields, and simpler industrial operation without requiring strict anhydrous environments typical of organometallic reagents.
Q: How is product purity ensured during the decarboxylation step?
A: Purity is maintained through precise pH control during hydrolysis and the use of benzyl protecting groups which prevent side reactions on the phenolic hydroxyls until the final deprotection stage.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights strong industrial application value due to the use of common solvents like ethanol and acetone, and standard unit operations such as reflux and filtration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dihydroxyacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that 3,5-dihydroxyacetophenone plays in the global supply chain for respiratory therapeutics, and we are committed to delivering this intermediate with uncompromising quality and consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities for clinical trials or metric tons for commercial launch, we can meet your demands seamlessly. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our commitment to excellence extends beyond mere compliance; we actively engage in continuous process improvement to enhance yield and reduce environmental impact, aligning our operations with the long-term goals of our partners. By choosing us, you gain access to a supply chain partner that understands the nuances of fine chemical manufacturing and prioritizes your project's success through proactive communication and technical support.
We invite you to collaborate with us to optimize your sourcing strategy and achieve significant efficiencies in your production costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate our capability to support your development timelines. Let us help you secure a stable and cost-effective supply of this vital intermediate, enabling you to focus on innovation and market expansion while we handle the complexities of chemical synthesis and logistics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
