Optimized Manufacturing of Clopidogrel Bisulfate: A Technical Breakthrough for Global Supply Chains
Optimized Manufacturing of Clopidogrel Bisulfate: A Technical Breakthrough for Global Supply Chains
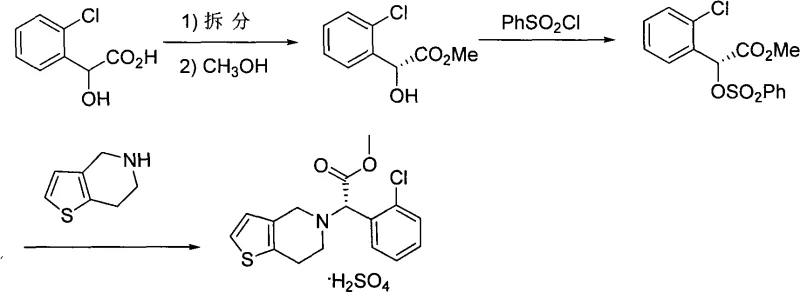
The global demand for antiplatelet agents remains steadfast, driven by the increasing prevalence of cardiovascular diseases worldwide. At the forefront of this therapeutic class is Clopidogrel, a molecule whose efficacy relies heavily on its stereochemical purity. Recent advancements in process chemistry have sought to address the limitations of early synthetic routes, particularly focusing on yield optimization and impurity control. A pivotal development in this arena is detailed in patent CN101348490A, which outlines a refined methodology for the preparation of S-type Clopidogrel and its bisulfate salt. This technical insight report analyzes the strategic value of this patented route, specifically examining how the integration of catalytic sulfonylation and optimized resolution strategies offers a compelling value proposition for R&D directors and procurement leaders seeking a reliable pharmaceutical intermediate supplier. By shifting away from racemic synthesis and low-yielding sulfonylation steps, this approach delivers a robust pathway that balances high purity with operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Clopidogrel has been plagued by significant inefficiencies that impact both cost and supply continuity. Early methodologies, such as those described in US4529596, relied on synthesizing the racemate first followed by chiral resolution. This approach is inherently flawed from an atom-economy perspective, as the theoretical maximum yield is capped at 50%, necessitating the disposal or recycling of the unwanted enantiomer. Furthermore, alternative routes involving condensation and cyclization often require harsh reagents like sodium cyanide, posing severe safety and environmental hazards that complicate regulatory compliance. Even within the "resolution-first" strategies, prior art such as US5132435 demonstrated critical bottlenecks; specifically, the sulfonylation of the chiral intermediate suffered from abysmal yields of approximately 45%, creating a major material loss point in the synthesis. Additionally, the reliance on solvents like dichloromethane with low boiling points limited the thermal window for reaction acceleration, forcing extended reaction times exceeding 20 hours which drastically reduces reactor throughput.
The Novel Approach
The methodology presented in CN101348490A represents a strategic evolution of the resolution-first pathway, specifically engineered to overcome the yield barriers of previous generations. By maintaining the chiral integrity from the starting material (R-o-chloromandelic acid), the process avoids the 50% yield ceiling associated with late-stage racemate resolution. The core innovation lies in the modification of the sulfonylation step, where the introduction of a specific catalyst system transforms a sluggish transformation into a high-efficiency reaction. Instead of accepting the 45% yield benchmark of the past, this novel approach leverages a synergistic combination of base and catalyst to drive the formation of the sulfonate leaving group to completion. This not only maximizes the utilization of the expensive chiral starting material but also simplifies the downstream purification burden. The result is a streamlined synthetic line that is not only chemically superior but also operationally simpler, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Catalytic Sulfonylation and Stereoinversion
To fully appreciate the technical merit of this synthesis, one must delve into the mechanistic nuances of the catalytic sulfonylation step. In traditional protocols, the reaction between the chiral alcohol and benzenesulfonyl chloride is often kinetically slow and prone to side reactions, leading to the poor yields observed in prior art. The patented process introduces trimethylamine hydrochloride as a catalyst alongside a tertiary amine base. Mechanistically, this likely facilitates the formation of a more reactive sulfonyl species or stabilizes the transition state, thereby lowering the activation energy required for the esterification of the sulfonic acid. This catalytic enhancement allows the reaction to proceed thoroughly even at reduced temperatures (-10 to 15°C), which is crucial for preserving the optical purity of the chiral center. Following this, the subsequent nucleophilic substitution by the tetrahydrothienopyridine moiety proceeds via an SN2 mechanism. This step is critical as it induces a Walden inversion, converting the R-configuration of the starting mandelic acid derivative into the desired S-configuration of the final Clopidogrel molecule. The precise control of reaction parameters, including the molar ratio of base to substrate, ensures that this inversion occurs with minimal racemization, safeguarding the enantiomeric excess required for biological activity.
Impurity control is another pillar of this mechanistic design. By optimizing the stoichiometry of the base and the sulfonyl chloride, the formation of bis-sulfonylated byproducts or hydrolysis products is minimized. The use of specific solvents like ethyl acetate in the final substitution step further aids in impurity management. Unlike polar aprotic solvents that might solubilize a wide range of polar impurities, ethyl acetate offers a selective solubility profile that facilitates the crystallization of the product while keeping inorganic salts and polar side-products in the aqueous phase during workup. This inherent "self-purifying" characteristic of the reaction medium reduces the need for extensive chromatographic purification, which is often a bottleneck in GMP manufacturing. Consequently, the final product exhibits high purity levels directly from the crystallization step, meeting the stringent specifications demanded by regulatory bodies for high-purity API intermediates.
How to Synthesize Clopidogrel Bisulfate Efficiently
The execution of this synthesis requires precise adherence to the optimized conditions defined in the patent to replicate the high yields and purity profiles. The process is divided into three distinct operational stages: esterification, catalytic sulfonylation, and final coupling. Each stage demands specific attention to temperature control and reagent addition rates to maintain safety and quality. For R&D teams looking to implement this technology, understanding the interplay between the catalyst loading and the reaction temperature in the second step is paramount. The following guide outlines the standardized workflow derived from the patent examples, serving as a foundational protocol for process development.
- Esterify R-o-chloromandelic acid with methanol under acidic conditions at 60-80°C to form the methyl ester intermediate.
- React the methyl ester with benzenesulfonyl chloride in the presence of a base and trimethylamine hydrochloride catalyst at -10 to 15°C.
- Perform nucleophilic substitution with 4,5,6,7-tetrahydrothienopyridine hydrochloride under alkaline conditions, followed by salt formation with concentrated sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical elegance. For procurement managers and supply chain heads, the primary value drivers are cost stability, raw material accessibility, and operational safety. The shift towards a high-yielding, resolution-first strategy fundamentally alters the cost structure of Clopidogrel manufacturing by maximizing the output per kilogram of chiral starting material. This efficiency gain translates directly into improved margin potential and reduced sensitivity to fluctuations in raw material pricing. Furthermore, the elimination of hazardous reagents like sodium cyanide removes a significant layer of logistical complexity and regulatory overhead associated with the transport and storage of controlled substances. This makes the supply chain more resilient and less prone to disruptions caused by safety audits or environmental compliance issues.
- Cost Reduction in Manufacturing: The economic advantage of this process is rooted in the dramatic improvement of yield at the critical sulfonylation stage. By elevating the yield from the historical baseline of roughly 45% to over 85%, the consumption of the chiral precursor is effectively halved for the same output volume. This reduction in material intensity is a powerful lever for cost reduction in pharmaceutical intermediate manufacturing. Additionally, the ability to recycle solvents such as ethyl acetate further diminishes the variable costs associated with production. The process avoids the need for expensive transition metal catalysts or complex chromatographic separations, relying instead on standard unit operations like extraction and crystallization which are capital efficient and widely available in existing manufacturing facilities.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche or hazardous reagents. This synthesis route utilizes commodity chemicals like methanol, benzenesulfonyl chloride, and potassium carbonate, which are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency. Moreover, the simplified reaction conditions—operating at normal pressure and moderate temperatures—reduce the likelihood of equipment failure or batch deviations that can halt production lines. The robustness of the process ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable, allowing manufacturers to respond more agilely to market demand spikes without compromising on quality standards.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a synthesis route becomes a critical selection criterion. This method is inherently greener, avoiding the generation of cyanide-containing waste streams that require costly treatment protocols. The use of ethyl acetate, a solvent with a favorable toxicity profile, aligns with the industry's push towards safer solvent guides. The process is designed for scalability, with exotherms that are manageable through standard cooling systems, ensuring that the transition from pilot plant to multi-ton commercial production is smooth and predictable. This alignment with environmental, social, and governance (ESG) goals adds intangible value to the supply chain, appealing to partners who prioritize sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Clopidogrel synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic benefits for potential partners. Understanding these details is essential for making informed decisions about technology transfer and long-term supply agreements.
Q: How does the catalytic sulfonylation step improve yield compared to conventional methods?
A: Conventional methods often suffer from low yields (around 45%) in the sulfonylation step. The patented process introduces a specific catalyst, trimethylamine hydrochloride, which significantly accelerates the reaction rate and ensures more thorough conversion, boosting yields to over 85%.
Q: What are the environmental advantages of this synthesis route?
A: This route avoids the use of highly toxic reagents like sodium cyanide found in other pathways. Furthermore, it utilizes recyclable organic solvents such as ethyl acetate, reducing hazardous waste generation and aligning with green chemistry principles.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under normal pressure with mild reaction temperatures (mostly below 80°C) and uses readily available raw materials. The simplified operational steps and robust impurity control make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Bisulfate Supplier
The technical superiority of the CN101348490A route underscores the importance of partnering with a manufacturer who possesses the expertise to execute complex chiral syntheses at scale. NINGBO INNO PHARMCHEM stands ready to leverage this advanced methodology to deliver consistent, high-quality Clopidogrel Bisulfate to the global market. Our facility is equipped with the necessary infrastructure to handle chiral resolutions and catalytic reactions with precision, ensuring that every batch meets the rigorous standards required for downstream API production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, coupled with stringent purity specifications enforced by our rigorous QC labs. Our commitment to technical excellence ensures that the theoretical advantages of this patent are fully realized in commercial supply.
We invite procurement leaders and R&D directors to engage with us for a deeper discussion on how this optimized route can enhance your supply chain resilience. By collaborating with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest degree of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
