Revolutionizing Carboxylic Acid Production: A Green Catalytic Route for Industrial Scale-Up
Revolutionizing Carboxylic Acid Production: A Green Catalytic Route for Industrial Scale-Up
The global demand for high-purity carboxylic acids, essential building blocks in the synthesis of active pharmaceutical ingredients (APIs) and agrochemicals, necessitates manufacturing processes that balance efficiency with environmental stewardship. Patent CN113292417A introduces a transformative methodology for the preparation of carboxylic acids through the oxidative cleavage of carbon-carbon bonds in vicinal diols. This technology addresses critical bottlenecks in traditional synthesis by employing a novel inorganic ligand-supported metal catalyst, specifically Anderson-type polyoxometalates, which facilitate the reaction under mild conditions using green oxidants. For R&D directors and procurement strategists, this patent represents a significant leap forward, offering a pathway to eliminate toxic heavy metal waste streams associated with conventional stoichiometric oxidants while achieving exceptional conversion rates exceeding 99% and yields approaching quantitative levels. The integration of this catalytic system into existing production lines promises not only regulatory compliance but also substantial operational efficiencies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of carboxylic acids via the oxidative cleavage of vicinal diols has relied heavily on stoichiometric amounts of high-valence oxidants such as sodium periodate, chromium trioxide, manganese dioxide, or sodium hypochlorite. These traditional reagents present severe drawbacks for modern sustainable manufacturing; they are inherently toxic, generate vast quantities of hazardous waste sludge, and often require complex downstream purification steps to remove heavy metal residues from the final product. Furthermore, earlier attempts to utilize heterogeneous metal catalysts like Na-Mn, Co, V, or W often resulted in insufficient conversion rates and poor yields, failing to meet the rigorous purity specifications required for pharmaceutical intermediates. The reliance on non-recyclable reagents drives up raw material costs and creates significant supply chain liabilities regarding waste disposal and environmental compliance, making these legacy processes increasingly untenable for large-scale commercial operations.
The Novel Approach
In stark contrast, the methodology disclosed in CN113292417A utilizes a sophisticated inorganic ligand-supported metal catalyst that operates efficiently with environmentally benign oxidants like hydrogen peroxide or oxygen. This approach fundamentally shifts the reaction paradigm from a wasteful stoichiometric process to a catalytic cycle where the metal center is regenerated. The process allows for the oxidative cleavage of diverse substrates, including hydrogenated benzoin and phenyl glycol, under relatively mild thermal conditions ranging from 50°C to 100°C. By avoiding the use of toxic chromium or manganese reagents, the novel approach drastically simplifies the workup procedure and eliminates the generation of hazardous heavy metal wastewater. The ability to achieve near-quantitative conversion and high isolation yields using this green chemistry platform positions it as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing, aligning perfectly with the industry's shift towards greener synthetic routes.
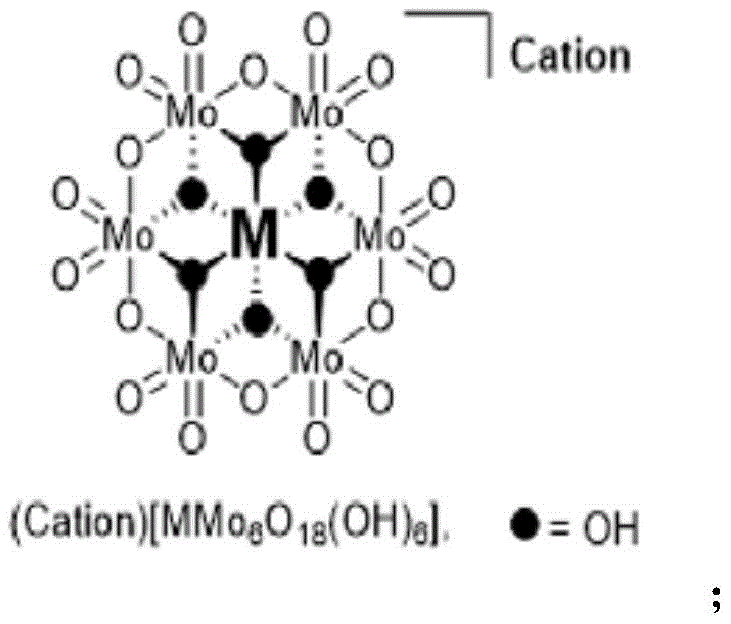
Mechanistic Insights into Anderson-Type Polyoxometalate Catalysis
The core innovation of this technology lies in the specific structure and electronic properties of the Anderson-type polyoxometalate catalyst, represented by the general formula $(Cation)[MMo_6O_{18}(OH)_6]$. As illustrated in the catalyst structure diagram, the central metal atom M (such as Iron, Nickel, Cobalt, or Chromium) is coordinated within a robust molybdenum-oxide framework. This unique architecture provides exceptional stability against oxidative degradation, allowing the catalyst to withstand the harsh conditions of peroxide oxidation without decomposing. The mechanism likely involves the activation of hydrogen peroxide by the metal center to form a high-valent metal-oxo species, which then engages in a ligand exchange reaction with the vicinal diol substrate. This interaction facilitates the cleavage of the C-C bond through a cyclic ester intermediate, ultimately releasing the carboxylic acid products and regenerating the catalyst for the next turnover. The presence of the inorganic ligand framework ensures that the active metal species remains soluble during the reaction but can be precipitated out in the aqueous phase post-reaction, enabling efficient recovery.
Impurity control is another critical aspect where this catalytic system excels compared to traditional methods. In conventional oxidations using periodate or chromic acid, side reactions often lead to over-oxidation or the formation of complex byproducts that are difficult to separate. The highly selective nature of the polyoxometalate catalyst minimizes these side reactions, ensuring a clean impurity profile in the crude product. The patent data indicates that by optimizing the solvent system, specifically using mixtures of acetonitrile and acetic acid, the solubility of the substrate and the stability of the transition states are finely tuned. This optimization suppresses the formation of oligomeric byproducts and ensures that the reaction proceeds cleanly to the desired carboxylic acid. For quality control teams, this means fewer chromatographic purification steps are required, directly translating to higher throughput and reduced solvent consumption in the final isolation stages.
How to Synthesize Carboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this reaction from laboratory benchtop to pilot plant operations. The process begins with the preparation of a suspension containing the diol substrate, the polyoxometalate catalyst, and hydrogen peroxide in a tailored solvent system. A key operational parameter identified is the two-stage heating profile, where an initial higher temperature phase (70-85°C) kickstarts the reaction, followed by a prolonged lower temperature phase (60-70°C) to drive the conversion to completion without degrading the product. This nuanced thermal control is essential for maximizing yield while maintaining catalyst integrity. The detailed standardized synthesis steps below outline the precise molar ratios, solvent choices, and workup procedures required to replicate the high performance reported in the patent examples.
- Prepare a suspension by mixing the vicinal diol substrate, hydrogen peroxide oxidant, and the Anderson-type polyoxometalate catalyst in a solvent mixture of acetonitrile and acetic acid.
- Heat the suspension using a two-stage protocol: first at 70-85°C for 6-10 hours, followed by a second stage at 60-70°C for 16-20 hours to maximize conversion.
- Quench the reaction, extract the organic phase to isolate the carboxylic acid product, and recover the solid catalyst from the aqueous phase via filtration for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic oxidative cleavage technology offers compelling economic and logistical advantages over legacy methods. The primary value driver is the drastic reduction in raw material costs associated with eliminating expensive and toxic stoichiometric oxidants. By switching to hydrogen peroxide, a commodity chemical with a stable supply chain, manufacturers can insulate themselves from the price volatility of specialized oxidizing agents. Furthermore, the ability to recover and recycle the expensive polyoxometalate catalyst multiple times significantly lowers the cost of goods sold (COGS) per kilogram of product. This recyclability reduces the frequency of catalyst procurement and minimizes the inventory burden, creating a more lean and responsive supply chain operation that is less susceptible to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal oxidants removes the need for costly waste treatment and disposal services, which often constitute a hidden but significant portion of manufacturing expenses. Additionally, the simplified workup procedure, which relies on phase separation and filtration rather than complex extractions or scavenging resins, reduces labor hours and solvent usage. The high conversion rates mean that less starting material is wasted, improving the overall atom economy of the process and ensuring that every dollar spent on raw materials translates efficiently into saleable product inventory.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents like hydrogen peroxide and acetonitrile ensures a stable supply chain that is not dependent on niche chemical suppliers. The robustness of the catalyst allows for consistent batch-to-batch reproducibility, reducing the risk of production delays caused by failed reactions or off-spec material. This reliability is crucial for meeting strict delivery deadlines for downstream API manufacturers, fostering stronger long-term partnerships and reducing the need for safety stock buffers that tie up working capital.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of standard heating and filtration equipment, avoiding the need for specialized high-pressure or cryogenic reactors. From an environmental perspective, the absence of chromium and manganese waste simplifies regulatory compliance and reduces the risk of environmental fines or shutdowns. This green credential is increasingly valuable for securing contracts with multinational corporations that have strict sustainability mandates, positioning the manufacturer as a preferred vendor in a competitive market.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this oxidative cleavage technology. These answers are derived directly from the experimental data and embodiments described in patent CN113292417A, providing clarity on catalyst selection, reaction conditions, and substrate compatibility. Understanding these nuances is vital for process engineers evaluating the feasibility of integrating this route into their current production portfolios.
Q: What are the advantages of using Anderson-type polyoxometalates over traditional oxidants?
A: Unlike stoichiometric toxic oxidants like chromium trioxide or periodate which generate heavy metal waste, Anderson-type polyoxometalates act as reusable heterogeneous catalysts. They enable the use of green oxidants like hydrogen peroxide, significantly reducing environmental impact and waste disposal costs while maintaining high conversion rates above 99%.
Q: Can the catalyst be recovered and reused in this process?
A: Yes, a key feature of this patented process is the recoverability of the catalyst. After the reaction and extraction of the organic product, the catalyst precipitates in the aqueous phase and can be separated by simple filtration, allowing it to be returned to the reactor for subsequent batches, thereby lowering long-term operational costs.
Q: What types of substrates are compatible with this oxidative cleavage method?
A: The method demonstrates broad substrate scope, effectively converting various vicinal diols and ketols such as hydrogenated benzoin, phenyl glycol, ethylene glycol, and cyclohexane-o-diol into their corresponding carboxylic acids with high selectivity and yield under mild thermal conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic oxidative cleavage technology described in CN113292417A for the production of high-value carboxylic acid intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of carboxylic acid meets the exacting standards required for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this catalytic process for maximum efficiency and cost-effectiveness for our global clients.
We invite you to collaborate with us to evaluate the feasibility of this advanced synthetic route for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic method. Please contact our technical procurement team today to request specific COA data for similar compounds and to discuss detailed route feasibility assessments tailored to your supply chain requirements.
