Advanced Manufacturing of Bosutinib via Mild Cyclization for Global Supply Chains
The pharmaceutical landscape for chronic myelogenous leukemia (CML) treatment relies heavily on the consistent supply of high-quality tyrosine kinase inhibitors, among which Bosutinib stands out as a critical therapeutic agent. Recent advancements in process chemistry, specifically detailed in patent CN111646939A, have introduced a transformative approach to synthesizing this complex molecule. This innovation addresses long-standing challenges in the final cyclization step, traditionally a bottleneck due to harsh reaction conditions and poor solubility profiles. By integrating an alkaline salt into the reaction system, manufacturers can now achieve superior kinetic control and material efficiency. 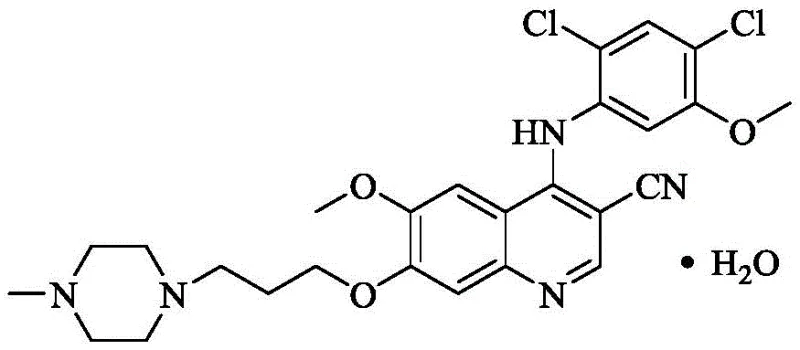 As depicted in the molecular structure, the intricate arrangement of the quinoline-3-carbonitrile core requires precise construction to maintain biological activity. This report analyzes the technical merits of this new methodology, offering strategic insights for R&D directors and procurement leaders seeking to optimize their API supply chains through robust, scalable chemistry.
As depicted in the molecular structure, the intricate arrangement of the quinoline-3-carbonitrile core requires precise construction to maintain biological activity. This report analyzes the technical merits of this new methodology, offering strategic insights for R&D directors and procurement leaders seeking to optimize their API supply chains through robust, scalable chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Bosutinib has been plagued by inefficient cyclization strategies that impose severe constraints on manufacturing throughput and cost effectiveness. Conventional routes, particularly those relying on direct dehydration with phosphorus oxychloride without additives, suffer from inadequate solubility of the starting materials within the reaction medium. This physical limitation necessitates the application of excessive thermal energy, often requiring temperatures as high as 115°C and prolonged reflux periods extending up to 16 hours to drive the reaction to completion. Such aggressive conditions not only escalate energy consumption but also increase the risk of thermal degradation and the formation of difficult-to-remove impurities. Furthermore, the reliance on linear synthesis strategies in older pathways often involves multiple protection and deprotection steps, which cumulatively reduce the overall yield to below 20% in some documented cases. These factors combine to create a fragile supply chain vulnerable to batch failures and inconsistent quality, posing significant risks for commercial partners dependent on reliable inventory levels for global distribution networks.
The Novel Approach
In stark contrast to these legacy methods, the novel technique disclosed in the referenced patent introduces a paradigm shift by incorporating alkaline salts directly into the cyclization reactor. This modification fundamentally alters the reaction environment, enhancing the solubility of the SM-1 intermediate and facilitating a smoother conversion to the target quinoline structure. By buffering the acidity generated during the chlorination and cyclization phases, the alkaline salt mitigates the formation of byproducts that typically arise from uncontrolled acid catalysis. The result is a process that operates effectively at significantly milder temperatures, ranging between 65°C and 70°C, while reducing the reaction time to a window of just 4 to 8 hours. This drastic reduction in thermal load and processing duration translates directly into enhanced operational safety and reduced utility costs. Moreover, the simplified workup procedure, which utilizes organic bases for crystallization rather than complex extraction sequences, streamlines the post-reaction processing, thereby minimizing solvent usage and waste generation while maximizing the recovery of high-purity product suitable for immediate downstream formulation.
Mechanistic Insights into Alkaline Salt-Assisted Cyclization
The efficacy of this improved synthesis lies in the nuanced interaction between the alkaline salt and the phosphorus oxychloride reagent during the ring-closing event. In the absence of the salt, the reaction mixture tends to become heterogeneous, leading to mass transfer limitations that slow down the kinetics and promote side reactions at the interface of undissolved solids. The introduction of salts such as sodium acetate or potassium carbonate acts to solubilize the intermediate species, ensuring a homogeneous reaction phase where molecular collisions occur with optimal frequency and orientation. 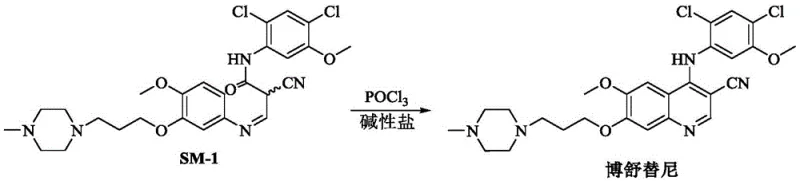 As illustrated in the reaction scheme, the transformation of SM-1 to Bosutinib proceeds through a activated intermediate that is stabilized by the basic environment. This stabilization prevents the premature decomposition of the nitrile group and protects the sensitive methoxy substituents from acid-catalyzed cleavage. Furthermore, the salt serves as an acid scavenger, neutralizing the hydrogen chloride gas evolved during the reaction, which otherwise could protonate the amine functionalities and inhibit the nucleophilic attack required for ring closure. This dual role of solubility enhancement and acid neutralization creates a thermodynamic driving force that favors the formation of the desired product over potential impurities, resulting in a cleaner reaction profile that is easier to purify on a multi-ton scale.
As illustrated in the reaction scheme, the transformation of SM-1 to Bosutinib proceeds through a activated intermediate that is stabilized by the basic environment. This stabilization prevents the premature decomposition of the nitrile group and protects the sensitive methoxy substituents from acid-catalyzed cleavage. Furthermore, the salt serves as an acid scavenger, neutralizing the hydrogen chloride gas evolved during the reaction, which otherwise could protonate the amine functionalities and inhibit the nucleophilic attack required for ring closure. This dual role of solubility enhancement and acid neutralization creates a thermodynamic driving force that favors the formation of the desired product over potential impurities, resulting in a cleaner reaction profile that is easier to purify on a multi-ton scale.
Controlling the impurity profile is paramount for any API intended for oncology applications, where regulatory standards for genotoxic impurities and heavy metals are exceptionally rigorous. The mild conditions afforded by this alkaline salt-assisted method inherently suppress the formation of high-molecular-weight oligomers and chlorinated byproducts that are common in high-temperature variants. By maintaining the reaction temperature below 70°C, the thermal energy available to overcome activation barriers for degradation pathways is insufficient, effectively locking the reaction into the desired productive channel. Additionally, the use of common inorganic salts eliminates the need for exotic catalysts that might introduce trace metal contaminants requiring costly removal steps later in the process. The crystallization step, triggered by the addition of organic bases like triethylamine to the filtrate, further refines the purity by selectively precipitating the target molecule while leaving soluble impurities in the mother liquor. This mechanism ensures that the final isolated solid meets the stringent specification of greater than 99.80% purity without the need for repetitive recrystallizations, thereby preserving yield and reducing solvent waste volumes significantly.
How to Synthesize Bosutinib Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and temperature control to fully realize the benefits of the alkaline salt additive. The process begins with the suspension of the SM-1 precursor and the selected alkaline salt in a polar aprotic solvent such as acetonitrile, which provides the necessary dielectric constant to support the ionic interactions involved. Phosphorus oxychloride is then added dropwise under strict temperature monitoring to prevent exothermic runaway, ensuring the reaction remains within the optimal 65°C to 70°C range throughout the addition and subsequent reflux period. Detailed standardized synthesis steps see the guide below.
- Dissolve SM-1 intermediate and an alkaline salt such as sodium acetate in an organic solvent like acetonitrile under inert gas protection.
- Add phosphorus oxychloride dropwise while maintaining temperature between 65°C and 70°C to initiate the cyclization reaction.
- After refluxing for 4 to 8 hours, cool the mixture, filter the solid, and recrystallize from a water-ethanol mixture with organic base adjustment.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized manufacturing process represents a substantial opportunity to enhance margin stability and supply security. The elimination of extreme thermal conditions reduces the strain on reactor vessels and heating systems, leading to lower maintenance costs and extended equipment lifecycles within the production facility. By shortening the batch cycle time from over 16 hours to under 8 hours, manufacturers can effectively double the throughput capacity of existing assets without requiring capital expenditure on new hardware. This increased velocity allows for more responsive inventory management, enabling suppliers to react swiftly to fluctuations in market demand for this critical oncology medication. Furthermore, the simplified purification workflow reduces the consumption of organic solvents and the volume of hazardous waste requiring disposal, aligning production practices with increasingly strict environmental regulations and sustainability goals demanded by global pharmaceutical partners.
- Cost Reduction in Manufacturing: The qualitative shift towards milder reaction conditions eliminates the need for high-energy heating protocols and specialized high-temperature resistant equipment, resulting in direct operational expenditure savings. The improved yield profile means that less raw material is wasted per kilogram of finished product, optimizing the cost of goods sold and allowing for more competitive pricing structures in tender negotiations. Additionally, the reduction in processing steps and solvent usage lowers the overall variable costs associated with each production batch, creating a more resilient economic model that can withstand fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The robustness of this chemical process minimizes the risk of batch failures due to thermal excursions or impurity spikes, ensuring a consistent and predictable output of qualified material. This reliability is crucial for maintaining continuous supply to formulation partners who cannot afford interruptions in the production of life-saving cancer therapies. The use of readily available inorganic salts and common solvents also mitigates the risk of supply disruptions associated with specialty reagents, securing the upstream supply chain against geopolitical or logistical volatility.
- Scalability and Environmental Compliance: The inherent safety of operating at lower temperatures and pressures facilitates easier scale-up from pilot plant to commercial multi-ton production without encountering the heat transfer limitations typical of exothermic cyclizations. The reduced generation of acidic waste streams and chlorinated byproducts simplifies effluent treatment requirements, ensuring compliance with environmental discharge standards and reducing the administrative burden of regulatory reporting. This eco-efficient profile enhances the corporate social responsibility standing of the supply chain, appealing to end-clients who prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkaline salt-assisted synthesis method for Bosutinib. These answers are derived directly from the experimental data and beneficial effects reported in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios or sourcing strategies for high-purity intermediates.
Q: How does the alkaline salt improve the Bosutinib synthesis yield?
A: The addition of alkaline salts significantly enhances the solubility of reactants in the phosphorus oxychloride system, preventing localized overheating and side reactions that typically lower yield in conventional high-temperature processes.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the process operates at mild temperatures between 65°C and 70°C with shorter reaction times, reducing energy consumption and equipment stress, which makes it highly scalable for industrial manufacturing compared to traditional routes requiring 115°C.
Q: What purity levels can be achieved with this novel route?
A: This optimized synthetic pathway consistently delivers Bosutinib with purity exceeding 99.80%, minimizing the need for extensive downstream purification and ensuring compliance with stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bosutinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven scaling capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in patent examples are replicated consistently in our manufacturing suites. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of Bosutinib meets the highest international standards for identity, assay, and impurity profiles. Our commitment to quality assurance means that clients can rely on us for a stable supply of this critical API intermediate, supporting their own regulatory filings and market launch timelines with confidence and precision.
We invite forward-thinking pharmaceutical companies to collaborate with us on optimizing their supply chains for oncology therapeutics. By leveraging our technical proficiency in this novel cyclization chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate how our manufacturing advantages can translate into tangible value for your organization. Together, we can ensure the uninterrupted availability of high-quality Bosutinib for patients worldwide while achieving mutual economic and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
