Advancing Benzoindolizine Production: A Cost-Effective Copper-Catalyzed Strategy for Global Supply Chains
Introduction to Next-Generation Benzoindolizine Synthesis
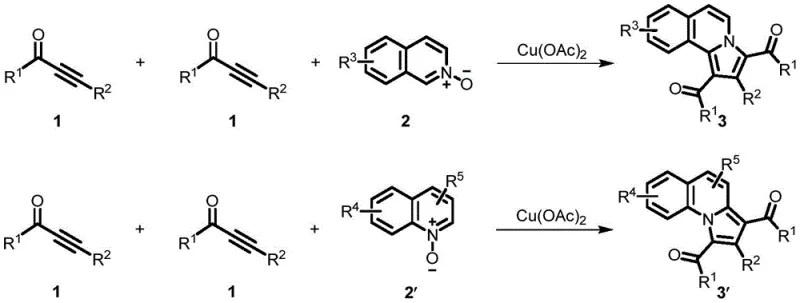
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and sustainable synthetic routes for biologically active scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN111662290B, which discloses a novel method for synthesizing benzoindolizine compounds. This technology addresses critical bottlenecks in the production of these valuable N-heterocycles, which are prevalent in numerous pharmaceutical and agrochemical candidates. By leveraging a copper-catalyzed [2+2+1] cyclization strategy, the patent outlines a pathway that transforms readily available alkynone compounds and quinoline or isoquinoline N-oxides into complex benzoindolizine derivatives. This approach is particularly noteworthy for its operational simplicity and robustness, offering a compelling alternative to legacy methods that often suffer from harsh reaction conditions and limited substrate scope.
For R&D directors and process chemists, the implications of this patent extend beyond mere academic interest; it represents a viable route for scaling up the production of high-purity intermediates. The methodology described eliminates the dependency on expensive noble metal catalysts and inert atmospheres, two factors that traditionally inflate the cost and complexity of fine chemical manufacturing. As we delve deeper into the technical specifics, it becomes evident that this innovation aligns perfectly with the industry's shift towards greener, more cost-effective synthesis protocols. The ability to utilize benign solvents like PEG-200 or water further underscores the environmental compatibility of this process, making it an attractive option for companies aiming to reduce their carbon footprint while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzoindolizine skeleton has relied on several established strategies, each carrying inherent drawbacks that hinder large-scale application. Traditional methods often involve the 1,3-dipolar cycloaddition of (iso)quinolinium salts with electron-deficient alkynes or alkenes, which typically requires the preparation of unstable salt precursors. Other approaches, such as the intramolecular cyclization of substituted (iso)quinolines or reactions involving diazoacetates, frequently necessitate the use of expensive transition metal catalysts like palladium, rhodium, or ruthenium. These noble metals not only drive up the raw material costs significantly but also introduce challenges related to residual metal removal, a critical quality attribute for pharmaceutical intermediates. Furthermore, many of these conventional protocols demand strict inert gas protection (nitrogen or argon) and elevated temperatures, increasing energy consumption and operational complexity. The limited tolerance for diverse functional groups in these older methods often restricts the structural diversity of the final products, forcing chemists to employ lengthy protection-deprotection sequences.
The Novel Approach
In stark contrast, the method disclosed in patent CN111662290B offers a streamlined and economically superior alternative. By utilizing copper(II) acetate (Cu(OAc)2) as the catalyst, the process achieves efficient cyclization under relatively mild conditions, typically between 80°C and 120°C. A defining feature of this novel approach is its operation under an air atmosphere, completely removing the need for inert gas shielding and the associated equipment costs. The reaction demonstrates excellent universality, accommodating a wide range of substituents on both the alkynone and the (iso)quinoline N-oxide substrates, including alkyl, phenyl, halogen, and ester groups. This broad substrate tolerance allows for the rapid generation of diverse libraries of benzoindolizine derivatives, accelerating the drug discovery process. Moreover, the use of green solvents such as PEG-200 or water enhances the safety profile and simplifies waste treatment, positioning this method as a leader in sustainable chemical manufacturing.
Mechanistic Insights into Cu(OAc)2-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the copper catalyst. The reaction is proposed to proceed via a [2+2+1] cyclization mechanism, where the alkynone and the N-oxide serve as the primary building blocks. The copper species likely activates the alkyne moiety of the alkynone, facilitating nucleophilic attack by the nitrogen oxide oxygen or the adjacent carbon framework. This activation lowers the energy barrier for the subsequent ring-closing steps, enabling the formation of the fused pyrrolo-isoquinoline or pyrrolo-quinoline system with high regioselectivity. Unlike radical-based mechanisms that might require harsh initiators, this copper-mediated pathway appears to be highly controlled, minimizing the formation of polymeric byproducts and ensuring a clean reaction profile. The ability of the catalyst to turnover efficiently in the presence of air suggests a robust catalytic cycle that is resistant to oxidation deactivation, a common pitfall for many base-metal catalysts.
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction conditions and the specific interaction between the copper center and the substrates reduce the likelihood of thermal decomposition or non-specific side reactions that often plague high-temperature processes. The patent data indicates that the resulting products can be isolated with high purity after standard workup procedures, such as extraction and silica gel chromatography. For process chemists, this implies a simpler downstream processing train, potentially reducing the number of purification steps required to meet stringent pharmaceutical specifications. The compatibility with various functional groups, such as methoxy, halogens, and esters, without significant degradation further attests to the chemoselectivity of this catalytic system, ensuring that sensitive moieties remain intact throughout the synthesis.
How to Synthesize Benzoindolizine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward, requiring standard equipment and readily available reagents. The general procedure involves dissolving the alkynone and the (iso)quinoline N-oxide in the chosen solvent, followed by the addition of the copper catalyst. The mixture is then heated and stirred for a defined period, typically ranging from 2 to 12 hours depending on the specific substrate reactivity. Upon completion, the reaction mixture undergoes a standard aqueous workup to remove inorganic salts and catalyst residues, followed by organic extraction. The crude product is then purified, often via column chromatography, to yield the target benzoindolizine compound. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Prepare the reaction mixture by adding alkynone compounds and quinoline or isoquinoline N-oxides into a solvent such as PEG-200 or water.
- Introduce Cu(OAc)2 as the catalyst (0.2 to 2 equivalents) under an air atmosphere without the need for inert gas protection.
- Heat the mixture to 80-120°C and stir for 2-12 hours, followed by extraction and purification to isolate the target benzoindolizine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology translates into tangible strategic benefits. The shift from precious metals to abundant copper represents a fundamental change in the cost structure of the raw materials. Copper acetate is orders of magnitude cheaper than palladium or rhodium catalysts, leading to a drastic reduction in the bill of materials for every kilogram of product produced. Additionally, the elimination of inert gas requirements simplifies the reactor setup, allowing for the use of standard glass-lined or stainless steel reactors without specialized sparging systems. This operational simplicity reduces capital expenditure for new production lines and lowers maintenance costs for existing facilities. The use of water or PEG-200 as solvents further mitigates the costs associated with volatile organic compound (VOC) management and disposal, aligning with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with inexpensive copper acetate results in substantial direct cost savings. Furthermore, the ability to run the reaction in air eliminates the ongoing expense of high-purity nitrogen or argon gases. The simplified downstream processing, driven by the clean reaction profile, reduces solvent consumption and labor hours associated with purification, collectively driving down the cost of goods sold (COGS) for these complex intermediates.
- Enhanced Supply Chain Reliability: Copper is a globally abundant commodity with a stable supply chain, unlike precious metals which are subject to geopolitical volatility and price spikes. By decoupling production from the precious metal market, manufacturers can secure more predictable pricing and availability for their catalyst needs. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by equipment failures related to inert atmosphere systems, ensuring consistent output and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The use of green solvents like water and PEG-200 significantly eases the burden of waste treatment and regulatory compliance. Scaling this process from gram to ton scale does not introduce new safety hazards related to pyrophoric reagents or high-pressure inert gases. The mild temperature range (80-120°C) is easily achievable with standard heating utilities, facilitating seamless technology transfer from R&D to commercial manufacturing. This environmental friendliness enhances the corporate sustainability profile, a key metric for modern supply chain evaluations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the detailed specifications and beneficial effects outlined in the patent documentation. These insights are intended to clarify the operational parameters and strategic value of adopting this technology for your specific application needs.
Q: What are the primary advantages of this copper-catalyzed method over traditional noble metal catalysis?
A: This method utilizes inexpensive copper acetate instead of costly transition metals like palladium or rhodium, significantly lowering raw material costs while maintaining high efficiency under mild conditions.
Q: Does this synthesis require specialized inert atmosphere equipment?
A: No, a key innovation of patent CN111662290B is that the reaction proceeds efficiently under an air atmosphere, eliminating the need for nitrogen or argon protection and simplifying operational requirements.
Q: What solvents are compatible with this green synthesis protocol?
A: The process supports environmentally friendly solvents, specifically Polyethylene Glycol-200 (PEG-200) or even water, enhancing the sustainability profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoindolizine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN111662290B for the production of high-value benzoindolizine derivatives. As a dedicated CDMO partner, we possess the technical expertise to adapt and optimize this copper-catalyzed protocol for your specific project requirements. Our facilities are equipped to handle diverse synthetic pathways, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch conforms to the highest industry standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this cost-effective and sustainable technology for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can improve your margins. Please contact our technical procurement team today to request specific COA data for our benzoindolizine portfolio and to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a reliable, high-quality supply of these critical intermediates.
