Advanced Catalytic Route for 2-Oxindole: Scaling High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for critical heterocyclic scaffolds, and Patent CN1078590C presents a transformative approach for the preparation of 2-oxindole and N-hydroxy-2-oxindole. This specific intellectual property details a practical process that begins with the catalytic hydrogenation of 2-nitroaryl malonate diesters, leading to the formation of key intermediates such as 2-(N-hydroxyamino)aryl malonate or 2-aminoaryl malonate. Unlike conventional multi-step syntheses that require the isolation of unstable intermediates, this method facilitates intramolecular aminolysis and subsequent decarboxylation in situ. For R&D directors and procurement specialists, this represents a significant opportunity to enhance the purity profile of pharmaceutical intermediates while simultaneously addressing the growing demand for cost reduction in API manufacturing. The ability to generate these valuable structures from readily available 2-halo nitro-aromatic starting materials underscores the commercial viability and strategic importance of this technology for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-oxindole derivatives has relied heavily on methods that are fraught with inefficiencies and environmental burdens, such as the Wolff-Kishner reduction or reductions using stoichiometric amounts of reactive metals like iron and tin. These traditional pathways often necessitate the preparation of isatin derivatives or 2-nitrophenyl acetic esters as isolated solid intermediates, which introduces multiple unit operations including filtration, drying, and purification. The use of polyvoltine metering iron or large chemical doses of tin not only generates substantial quantities of hazardous metal waste but also complicates the downstream processing due to the need for extensive waste treatment. Furthermore, these older methods frequently suffer from poor regiocontrol, leading to mixtures of isomeric oxindoles that are difficult to separate, thereby reducing the overall yield and increasing the cost of goods. The cumulative effect of these limitations is a manufacturing process that is neither economically sustainable nor environmentally compliant for modern large-scale production.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent leverages a streamlined sequence where catalytic hydrogenation, cyclization, hydrolysis, and decarboxylation occur within a single reaction vessel or continuous flow system without isolating intermediates. By utilizing 2-nitroaryl diester malonate as the starting substrate, the process capitalizes on the presence of two ester groups to facilitate intramolecular cyclization through aminolysis, a mechanism that is surprisingly efficient even without the addition of external acid catalysts. This in situ methodology eliminates the need for isolating the 2-nitrophenyl acetic ester, which is a common bottleneck in prior art, and avoids the generation of heavy metal waste associated with stoichiometric reductions. The result is a drastically simplified workflow that enhances the overall yield and purity of the final 2-oxindole product, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates. This shift from stoichiometric to catalytic reagents fundamentally alters the economic model of production, offering substantial cost savings and improved supply chain reliability.
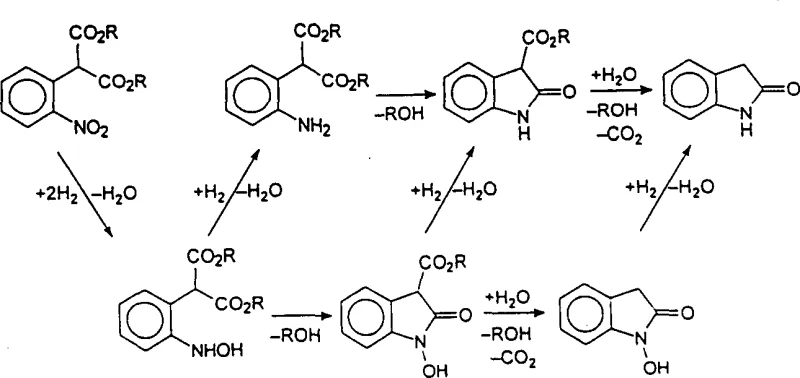
Mechanistic Insights into Catalytic Hydrogenation and Intramolecular Cyclization
The core chemical innovation of this process lies in the unique reactivity of the 2-aminoaryl diester malonate and 2-(N-hydroxyamino)aryl diester malonate intermediates generated during the hydrogenation phase. The presence of the second ester group plays a critical role in promoting cyclization, potentially through intramolecular hydrogen bonding or by stabilizing the negative charge developed in the tetrahedral intermediate via conjugation with the aromatic pi-system. This tautomeric stabilization effectively lowers the activation energy for the intramolecular aminolysis step, allowing the ring closure to proceed smoothly under the reaction conditions without requiring harsh reagents. The mechanism suggests that the ester group acts similarly to an acid catalyst within the tautomeric form, polarizing the carbonyl group and making it susceptible to nucleophilic attack by the adjacent amino or hydroxylamino functionality. This deep understanding of the electronic effects governing the cyclization allows chemists to fine-tune reaction conditions to maximize selectivity and minimize the formation of by-products, ensuring a high-purity output suitable for sensitive pharmaceutical applications.
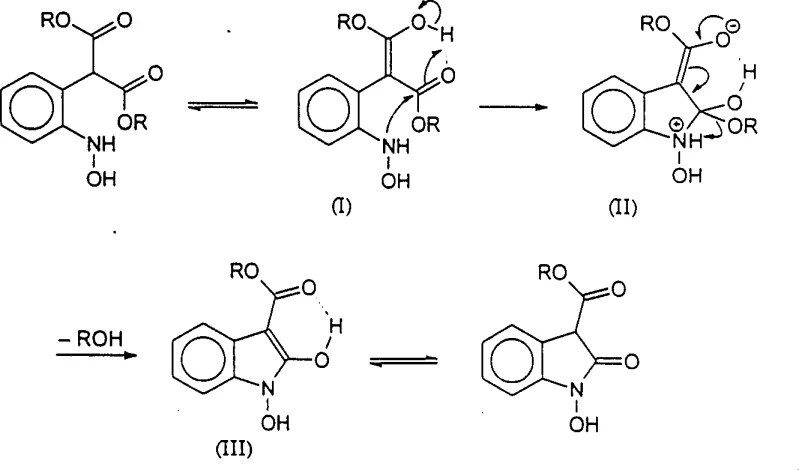
Following the cyclization event, the resulting 2-oxindole-3-carboxylic ester or N-hydroxy-2-oxindole-3-carboxylic ester undergoes hydrolysis and decarboxylation with remarkable ease, even in the absence of added water or acid in certain solvent systems. The patent data indicates that the water produced during the reduction of the nitro group is often sufficient to drive the hydrolysis, while the decarboxylation is facilitated by the electronic structure of the cyclized intermediate. This spontaneous decomposition of the carboxylic ester moiety is a critical advantage, as it removes the need for a separate hydrolysis step that typically requires strong acids or bases and elevated temperatures. By controlling the hydrogenation conditions, such as temperature and pressure, manufacturers can selectively produce either the N-hydroxy derivative or fully reduce it to the 2-oxindole, providing flexibility in synthesizing different downstream active pharmaceutical ingredients. This mechanistic elegance ensures that the process remains robust and scalable, reducing the risk of batch-to-batch variability.
How to Synthesize 2-Oxindole Efficiently
The synthesis of 2-oxindole via this patented route involves a sequence of highly integrated chemical transformations that begin with the preparation of the 2-nitroaryl diester malonate precursor from 2-halo nitro-aromatics. Once the precursor is formed, it is subjected to catalytic hydrogenation in a suitable solvent system, such as ethanol or acetic acid, using precious metal catalysts like palladium or platinum on carbon. The detailed standardized synthesis steps see the guide below, which outlines the specific parameters for temperature, pressure, and catalyst loading required to achieve optimal conversion and selectivity. This operational framework is designed to minimize manual intervention and maximize throughput, making it highly suitable for automated manufacturing environments. By adhering to these optimized conditions, production teams can ensure consistent quality and yield, thereby reducing lead time for high-purity oxindoles and enhancing the overall efficiency of the supply chain.
- Prepare 2-nitroaryl malonate diester from 2-halo nitro-aromatic and diester malonate anion.
- Perform catalytic hydrogenation to generate amino or hydroxylamino intermediates without isolation.
- Execute in situ intramolecular aminolysis followed by hydrolysis and decarboxylation to yield 2-oxindole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring continuity of supply for critical chemical inputs. The elimination of stoichiometric metal reductants like iron and tin removes a significant cost center associated with raw material procurement and waste disposal, leading to a cleaner and more sustainable manufacturing footprint. Furthermore, the in situ nature of the reaction reduces the number of unit operations, which translates to lower energy consumption, reduced solvent usage, and decreased labor requirements per kilogram of product. These efficiencies collectively contribute to a more competitive pricing structure for the final 2-oxindole intermediate, allowing downstream pharmaceutical companies to manage their cost of goods sold more effectively. The robustness of the catalytic system also implies a lower risk of production delays due to equipment fouling or complex workup procedures, thereby enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The transition from stoichiometric metal reductions to catalytic hydrogenation fundamentally alters the cost structure by eliminating the need for expensive metal powders and the associated waste treatment costs. By avoiding the isolation of solid intermediates, the process reduces solvent consumption and energy usage related to drying and filtration, resulting in substantial cost savings. The higher overall yield achieved through the minimization of purification steps further drives down the unit cost, making this route economically superior to traditional methods. Additionally, the use of common solvents and standard hydrogenation equipment minimizes capital expenditure requirements for implementation.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-halo nitro-aromatics and diethyl malonate ensures a stable supply of raw materials that are not subject to the volatility of specialized reagent markets. The simplified process flow reduces the complexity of the manufacturing schedule, allowing for faster turnaround times and more responsive production planning. This agility is crucial for meeting the dynamic demands of the pharmaceutical industry, where delays in intermediate supply can impact the launch of new drugs. The robustness of the catalytic process also minimizes the risk of batch failures, ensuring a consistent flow of high-quality material to customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard catalytic hydrogenation technology, which is well-understood and widely deployed in the fine chemical industry. The significant reduction in metal waste aligns with increasingly stringent environmental regulations, reducing the regulatory burden and potential liability associated with hazardous waste disposal. This environmental advantage not only improves the corporate sustainability profile but also facilitates smoother regulatory approvals for new manufacturing sites. The ability to scale from laboratory to commercial production without significant process re-engineering ensures a seamless transition to high-volume manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity on its operational feasibility and strategic benefits. These insights are derived directly from the patent specifications and are intended to assist technical teams in evaluating the suitability of this process for their specific manufacturing needs. Understanding these details is essential for making informed decisions about process adoption and supply chain integration. The answers reflect the core innovations of the patent, focusing on yield, purity, and operational simplicity.
Q: How does this patent improve upon traditional iron powder reduction methods?
A: Traditional methods utilize stoichiometric amounts of iron or tin in acidic conditions, generating substantial metal waste and requiring complex filtration. This patent employs catalytic hydrogenation, eliminating heavy metal waste and simplifying the workup process significantly.
Q: Is isolation of the intermediate 2-nitrophenyl acetic ester required?
A: No. A key innovation of this process is the ability to perform cyclization, hydrolysis, and decarboxylation in situ. This avoids the isolation and purification of solid intermediates, thereby reducing processing time and material loss.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes. The process utilizes standard catalytic hydrogenation equipment and common solvents like ethanol or acetic acid. The elimination of isolation steps and the use of robust catalytic conditions make it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving medications, and we are committed to delivering excellence in every batch. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global pharmaceutical partners without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of 2-oxindole meets the exacting standards required for API synthesis. Our infrastructure is designed to support the complex chemistry described in Patent CN1078590C, allowing us to offer a reliable supply of this valuable intermediate to the market.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that you have all the information necessary to make a confident sourcing decision. Together, we can drive innovation and efficiency in the production of essential pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
