Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Applications
Introduction to Novel Quinazolinone Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN112480015B, which discloses a highly efficient multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This technology addresses long-standing challenges in heterocyclic chemistry by leveraging a palladium-catalyzed carbonylation cascade that transforms readily available nitro compounds and trifluoroethylimidoyl chlorides into complex bioactive structures. The introduction of the trifluoromethyl group is particularly strategic, as it enhances the metabolic stability, lipophilicity, and bioavailability of the resulting drug candidates, making this methodology highly relevant for the development of next-generation antifungal, antiviral, and anticancer agents. By integrating the carbonylation step directly into the cyclization process, this approach streamlines the synthetic pathway, reducing the number of unit operations required and minimizing waste generation compared to traditional stepwise protocols.
The significance of this invention extends beyond mere academic interest; it offers a tangible solution for reliable pharmaceutical intermediate supplier networks aiming to secure stable supplies of high-value scaffolds. Quinazolinones are ubiquitous in medicinal chemistry, found in established drugs such as methaqualone and various kinase inhibitors. However, their synthesis has often been hampered by the need for hazardous reagents or complex multi-step sequences. The method described in CN112480015B utilizes a transition metal palladium catalyst system that operates under relatively mild thermal conditions, facilitating the construction of the quinazolinone core with high atom economy. This technological advancement positions manufacturers to offer cost reduction in API manufacturing by simplifying the supply chain for these critical intermediates, ensuring that downstream drug developers can access high-purity materials without the bottlenecks associated with older, less efficient synthetic technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has relied on methodologies that impose significant operational burdens and safety risks on chemical manufacturing facilities. Traditional routes often involve the use of high-pressure carbon monoxide gas, which requires specialized autoclaves and rigorous safety protocols to prevent leakage and exposure, thereby increasing capital expenditure and operational complexity. Furthermore, many existing protocols necessitate the use of expensive pre-activated substrates, such as 2-bromoformylanilines or acid anhydrides, which not only drive up raw material costs but also generate stoichiometric amounts of halogenated or acidic waste streams that require costly disposal. Another critical limitation is the reliance on precious metal catalysts like ruthenium or platinum under harsh conditions, which can lead to poor substrate compatibility and low yields when sensitive functional groups are present. These factors collectively contribute to extended lead times and reduced flexibility in process development, making it difficult for procurement teams to secure consistent volumes of intermediates for clinical and commercial programs.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a cleverly designed multi-component reaction system that circumvents these historical hurdles by employing solid molybdenum hexacarbonyl as a safe and controllable carbon monoxide surrogate. This innovation allows the carbonylation to proceed under atmospheric pressure conditions within a standard reactor, drastically lowering the barrier to entry for scale-up and eliminating the need for high-pressure infrastructure. The reaction initiates with the reduction of inexpensive nitro compounds to amines in situ, followed immediately by condensation with trifluoroethylimidoyl chloride and subsequent palladium-catalyzed cyclization. This telescoped sequence not only improves overall yield by avoiding the isolation of unstable intermediates but also broadens the substrate scope to include a wide array of electronically diverse nitroarenes. The result is a streamlined process that delivers high-purity quinazolinone derivatives with exceptional efficiency, providing a compelling value proposition for partners seeking to optimize their synthetic routes for commercial viability.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
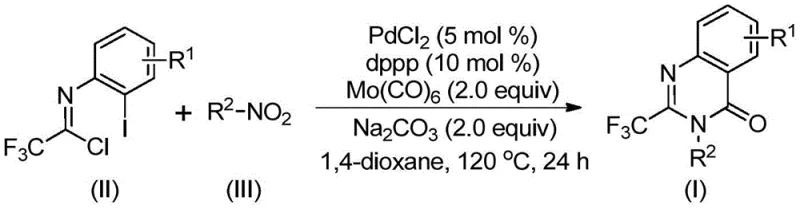
The mechanistic pathway of this transformation is a sophisticated example of transition metal catalysis, involving a sequential series of bond-forming events orchestrated by the palladium center. The cycle likely begins with the reduction of the nitro group on the aromatic ring by molybdenum hexacarbonyl, generating the corresponding aniline species which then undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst, coordinated by the dppp ligand, inserts into the carbon-iodine bond of the imidoyl chloride moiety, forming a reactive organopalladium species. As the temperature is maintained at 120°C, the molybdenum hexacarbonyl releases carbon monoxide, which inserts into the carbon-palladium bond to generate an acyl-palladium intermediate. This acyl species then undergoes an intramolecular nucleophilic attack by the nitrogen atom of the amidine, facilitated by the base, to close the seven-membered palladacycle before finally undergoing reductive elimination to release the desired 2-trifluoromethyl quinazolinone product and regenerate the active catalyst.
Understanding this mechanism is crucial for R&D directors focused on impurity control and process robustness. The choice of 1,3-bis(diphenylphosphino)propane (dppp) as the ligand is critical, as its bite angle and electronic properties stabilize the palladium intermediates and prevent the formation of side products such as homocoupling dimers or dehalogenated byproducts. Furthermore, the use of sodium carbonate as a mild base ensures that the reaction medium remains sufficiently alkaline to promote the cyclization step without causing hydrolysis of the sensitive imidoyl chloride or the final quinazolinone product. The compatibility of this system with various functional groups, including halogens and trifluoromethyl groups, suggests that the catalytic cycle is resilient against competitive coordination or poisoning, allowing for the synthesis of complex analogues with high fidelity. This level of mechanistic control translates directly into a cleaner crude reaction profile, simplifying downstream purification and ensuring that the final material meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, carbon monoxide source, and the two primary organic substrates in a suitable aprotic solvent such as 1,4-dioxane. The mixture is then heated to 120°C and stirred for a period ranging from 16 to 30 hours, depending on the specific electronic nature of the substrates involved. Upon completion, the reaction mixture is cooled, filtered to remove inorganic salts and metal residues, and the filtrate is concentrated. The crude product is typically purified using standard silica gel column chromatography to afford the target compound in high purity. For detailed standardized operating procedures and specific stoichiometric ratios tailored to your specific substrate requirements, please refer to the technical guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane solvent.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers profound advantages that resonate deeply with the strategic goals of procurement managers and supply chain heads. The primary driver of value is the substantial cost savings achieved through the utilization of commodity-grade starting materials. Nitro compounds are among the most abundant and inexpensive chemicals in the global market, and their direct use eliminates the need for costly pre-functionalization steps that characterize older synthetic routes. Furthermore, the replacement of gaseous carbon monoxide with a solid surrogate removes the logistical and safety costs associated with handling toxic gases, thereby reducing insurance premiums and facility compliance overheads. The high reaction efficiency and broad substrate tolerance mean that a single platform technology can be used to produce a diverse library of intermediates, allowing manufacturers to consolidate suppliers and reduce the complexity of their vendor management systems while ensuring a steady flow of materials for drug development pipelines.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive reagents and the simplification of the reaction setup. By avoiding high-pressure equipment and utilizing cheap nitro precursors, the overall cost of goods sold (COGS) for these intermediates is significantly lowered. The high yields observed across a range of substrates minimize raw material waste, further enhancing the economic efficiency of the process. Additionally, the use of a recyclable palladium catalyst system, although requiring initial investment, offers long-term savings compared to stoichiometric reagents, making the process economically sustainable for large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the key raw materials. Nitro compounds and trifluoroethylimidoyl chlorides are produced by numerous chemical manufacturers globally, reducing the risk of supply disruptions caused by single-source dependencies. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without significant re-validation, providing flexibility in sourcing strategies. This reliability ensures that downstream pharmaceutical clients can maintain their production schedules without interruption, mitigating the risk of drug shortages and supporting continuous commercial supply.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The use of 1,4-dioxane as a solvent, while requiring careful handling, is a well-understood industrial solvent with established recovery and recycling protocols. The reduction in waste generation, particularly the avoidance of heavy metal waste from stoichiometric reagents and the minimization of halogenated byproducts, aligns with modern green chemistry principles. This environmental compatibility simplifies regulatory compliance and waste disposal, reducing the environmental footprint of the manufacturing process and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent literature. These questions address common concerns regarding reaction scalability, substrate limitations, and purification requirements. The answers are derived directly from the empirical results of the study, ensuring that the information provided is accurate and reflective of the actual performance of the chemistry. We encourage potential partners to review these insights to understand how this methodology can be integrated into their existing workflows to enhance efficiency and reduce costs.
Q: What are the key advantages of this one-pot synthesis method over traditional routes?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates. It utilizes cheap nitro compounds and operates under atmospheric pressure with a solid CO source (Mo(CO)6), significantly simplifying the operational setup and improving safety profiles for industrial scale-up.
Q: What is the substrate scope for this palladium-catalyzed reaction?
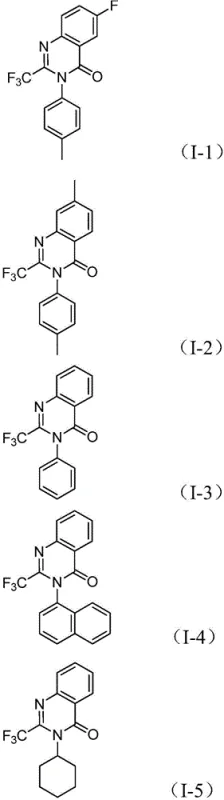
A: The reaction demonstrates excellent functional group tolerance. It accommodates various substituents on the nitro compound aromatic ring, including halogens (F, Cl, Br), alkyl groups (methyl), and electron-withdrawing groups (trifluoromethyl). Additionally, diverse amine precursors (aryl, alkyl, cycloalkyl) can be successfully incorporated into the final quinazolinone structure.
Q: How does this process impact the purity and isolation of the final API intermediate?
A: The process yields high-purity products with isolated yields ranging from 69% to 96% across different substrates. The post-treatment involves standard filtration and column chromatography, which are well-established purification techniques in the pharmaceutical industry, ensuring that impurity profiles are manageable and consistent with stringent QC requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into reliable commercial supplies. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging technologies such as the palladium-catalyzed one-pot synthesis described here, we can offer our clients a competitive edge through faster turnaround times and superior cost structures, enabling them to bring life-saving therapies to market more efficiently.
We invite you to collaborate with us to explore the full potential of this synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Together, we can build a resilient and efficient supply chain that supports your long-term growth and innovation goals in the global pharmaceutical market.
