Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Metal-Free Strategy for Scalable API Intermediates
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Metal-Free Strategy for Scalable API Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for safer, more efficient, and environmentally benign synthetic routes. A groundbreaking development detailed in patent CN113121462B introduces a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, a structural motif ubiquitous in modern drug discovery and agrochemical design. This technology represents a paradigm shift away from traditional transition-metal catalysis, offering a robust, metal-free alternative that leverages inexpensive cesium carbonate as a promoter. For R&D directors and procurement strategists alike, this innovation addresses critical pain points regarding impurity profiles, specifically the removal of heavy metal residues, while simultaneously enhancing operational safety by circumventing the use of hazardous organic azides. The ability to construct these valuable scaffolds under mild conditions using readily available starting materials positions this methodology as a cornerstone for next-generation pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic variants involving trifluoromethyl ketones. As illustrated in the comparative reaction schemes below, these traditional pathways are fraught with significant drawbacks that hinder large-scale adoption. The primary concern lies in the reliance on organic azides, which are inherently unstable, toxic, and prone to explosive decomposition, posing severe safety risks in industrial settings. Furthermore, copper-catalyzed routes necessitate the use of stoichiometric or near-stoichiometric amounts of transition metals, which creates a downstream burden for purification. Removing trace copper to meet stringent pharmaceutical specifications often requires complex chelation workflows or extensive chromatography, driving up both production costs and lead times. Additionally, the requirement for specialized trifluoromethylating reagents in some legacy protocols adds another layer of expense and supply chain vulnerability.
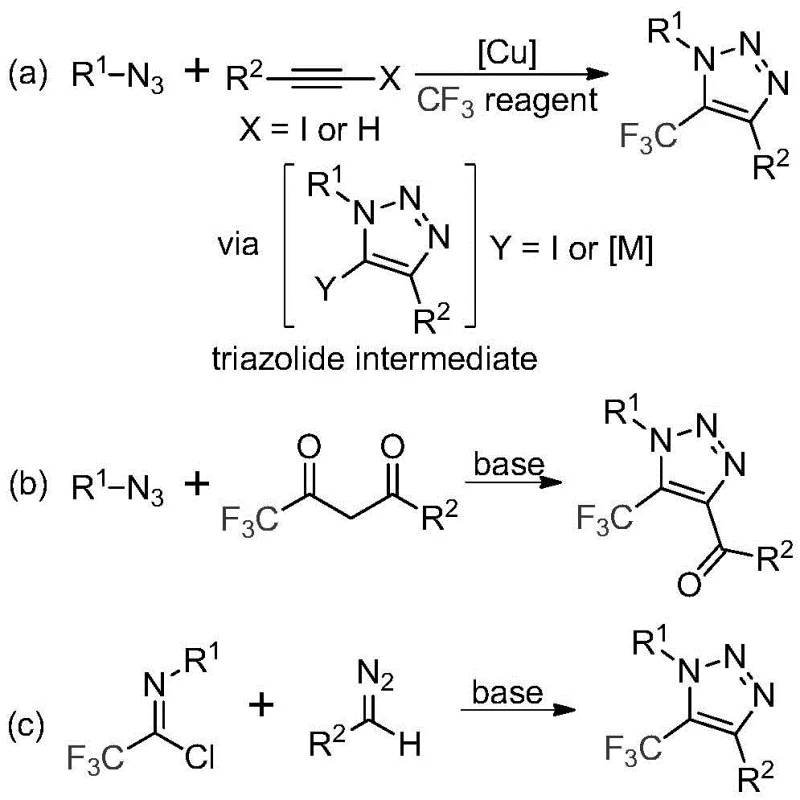
The Novel Approach
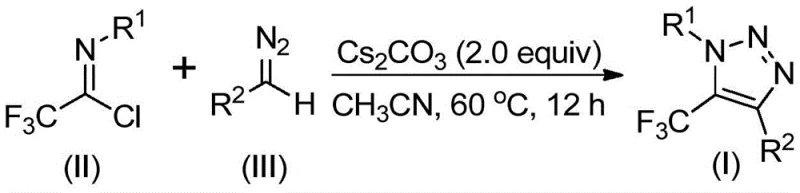
In stark contrast to these legacy methods, the technology disclosed in CN113121462B utilizes a base-promoted cyclization between trifluoroethylimidoyl chlorides and diazo compounds. This approach completely bypasses the need for explosive azides and toxic transition metal catalysts. The reaction proceeds smoothly in common aprotic solvents like acetonitrile at moderate temperatures ranging from 50°C to 70°C. By shifting the mechanistic paradigm to a nucleophilic addition-elimination sequence followed by cyclization, this method achieves high atom economy and exceptional functional group tolerance. The elimination of copper not only simplifies the workup procedure—often requiring only filtration and standard column chromatography—but also ensures a cleaner impurity profile, which is paramount for regulatory compliance in API synthesis. This streamlined process transforms a potentially hazardous multi-step operation into a safe, one-pot transformation suitable for continuous flow or batch processing.
Mechanistic Insights into Base-Promoted Cyclization
The elegance of this synthesis lies in its mechanistic simplicity and efficiency. The reaction initiates with the deprotonation of the diazo compound by cesium carbonate, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This intermolecular nucleophilic addition-elimination step is crucial as it establishes the carbon-carbon bond necessary for ring closure without the need for external oxidants or reductants. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific cyclization mode is thermodynamically favorable under the reaction conditions and leads directly to the formation of the 1,2,3-triazole core. The trifluoromethyl group, originating from the imidoyl chloride starting material, is retained at the 5-position of the triazole ring, ensuring precise regiocontrol that is often difficult to achieve in metal-catalyzed variants. The use of cesium carbonate is particularly strategic; its moderate basicity is sufficient to activate the diazo precursor without causing premature decomposition, while its solubility profile in acetonitrile facilitates homogeneous reaction kinetics.
From an impurity control perspective, this mechanism offers distinct advantages. Since no transition metals are involved, the risk of metal-catalyzed side reactions, such as homocoupling of alkynes or oxidative degradation, is entirely eliminated. The primary byproducts are typically inorganic salts derived from the base and the chloride leaving group, which are easily removed during the aqueous workup or filtration steps. This results in a crude product with high purity, reducing the load on downstream purification units. Furthermore, the broad substrate scope demonstrated in the patent data indicates that electron-withdrawing and electron-donating groups on both the imidoyl chloride and the diazo compound are well-tolerated. This versatility allows medicinal chemists to rapidly generate diverse libraries of trifluoromethyl-triazoles for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new analog.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and requires standard equipment. The process begins by charging a reaction vessel with the trifluoroethylimidoyl chloride, the corresponding diazo compound, and cesium carbonate in anhydrous acetonitrile. Molecular sieves (4 Å) are often added to scavenge trace moisture, which can hydrolyze the sensitive imidoyl chloride starting material. The mixture is then heated to approximately 60°C and stirred for 8 to 16 hours. Monitoring the reaction progress via TLC or HPLC ensures complete consumption of the starting materials before proceeding to isolation. The simplicity of the protocol means that specialized high-pressure reactors or inert atmosphere gloveboxes are generally not required, making it accessible for most synthetic facilities. For detailed standardized operating procedures and specific molar ratios optimized for different substrates, please refer to the technical guide below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to obtain the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible strategic benefits that extend beyond mere chemical yield. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. Traditional methods often rely on specialized, high-cost trifluoromethylating reagents or unstable azides that require cold-chain logistics and hazardous material handling certifications. In contrast, the starting materials for this novel process—trifluoroethylimidoyl chlorides and diazo compounds—are commercially available, stable, and can be sourced from multiple vendors, thereby mitigating single-source supply risks. This abundance of feedstock ensures consistent availability and stabilizes pricing, protecting manufacturing budgets from volatile market fluctuations associated with exotic reagents.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the elimination of expensive transition metal catalysts. Copper salts, ligands, and the associated scavengers required for their removal represent a significant line item in the bill of materials for traditional triazole synthesis. By replacing these with inexpensive cesium carbonate, the direct material costs are substantially lowered. Moreover, the simplified workup procedure reduces the consumption of solvents and silica gel during purification, further driving down operational expenditures. The absence of heavy metals also means that waste streams are easier and cheaper to treat, aligning with increasingly strict environmental regulations and reducing disposal fees.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity. The avoidance of explosive organic azides removes a major bottleneck in facility scheduling and insurance compliance. Facilities that are restricted from handling high-energy azides due to safety limits can now produce these valuable intermediates without requiring costly infrastructure upgrades or special permits. This flexibility allows for faster scale-up from pilot to commercial production, significantly reducing the lead time for bringing new drug candidates to market. The robustness of the reaction conditions also means that batch-to-batch variability is minimized, ensuring a reliable flow of high-quality intermediates to downstream formulation teams.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, this metal-free approach offers a clear pathway to sustainability. The reaction generates minimal hazardous waste, and the solvents used, such as acetonitrile, are easily recoverable and recyclable. The process is inherently scalable, having been demonstrated to work efficiently from milligram to gram scales in the patent literature, with clear indicators for successful tonnage production. This scalability ensures that as demand for trifluoromethyl-triazole containing drugs grows, the manufacturing capacity can be expanded seamlessly without encountering the engineering challenges often associated with exothermic metal-catalyzed processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: Why is this new synthesis method safer than traditional CuAAC routes?
A: Traditional methods often rely on toxic and explosive organic azides. This novel protocol utilizes stable diazo compounds and imidoyl chlorides, eliminating the safety hazards associated with handling high-energy azide intermediates.
Q: What are the cost advantages of using cesium carbonate over copper catalysts?
A: Using cesium carbonate eliminates the need for expensive transition metal catalysts and the subsequent costly purification steps required to remove trace heavy metals from pharmaceutical intermediates, significantly lowering production costs.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent explicitly demonstrates that the reaction conditions are mild and the procedure is simple, allowing for easy scale-up from gram-level laboratory synthesis to multi-ton commercial production without complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including rigorous moisture control and efficient solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,3-triazole meets the highest international standards for API intermediates, free from heavy metal contamination.
We invite you to collaborate with us to leverage this innovative technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your overall production budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
