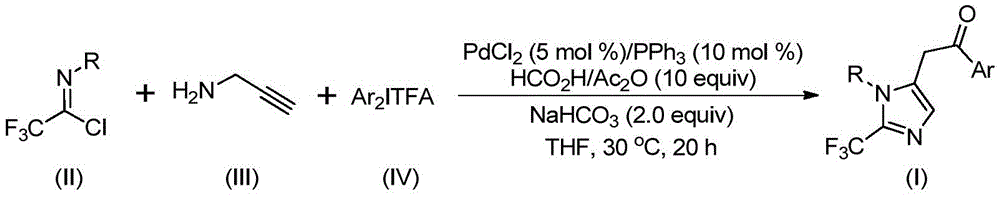
Advanced Synthesis of 2-Trifluoromethyl Imidazole: Scaling Complex API Intermediates with Precision
The recent patent CN111423381B introduces a novel methodology for synthesizing 2-trifluoromethyl substituted imidazole compounds, a critical class of pharmaceutical intermediates with applications in drug development such as H1 histamine receptor antagonists. This breakthrough offers a reliable API intermediate supplier solution by leveraging a palladium-catalyzed carbonylation process that operates under mild conditions (30°C), ensuring high-purity output while reducing lead time for high-purity intermediates. The process utilizes readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts, with reaction completion achieved within 20 hours without requiring specialized equipment or hazardous reagents. This innovation directly addresses the pharmaceutical industry's demand for cost-effective manufacturing pathways while maintaining stringent quality standards required for clinical and commercial production.
Advanced Reaction Mechanism and Purity Control
The patented methodology employs a sophisticated palladium-catalyzed cascade reaction that begins with intermolecular carbon-nitrogen bond formation promoted by sodium bicarbonate to generate a trifluoroacetamidine intermediate. This is followed by isomerization and palladation of the alkyne amine to form an alkenyl palladium species, which undergoes further isomerization to an alkyl palladium intermediate. Under the action of carbon monoxide released from the formic acid/acetic anhydride system, carbonylation occurs to produce an acyl palladium intermediate, which then undergoes oxidation addition with the diaryl iodonium salt to form a tetravalent palladium species before final reductive elimination yields the target imidazole compound. This multi-step sequence operates under precisely controlled conditions (THF solvent at 30°C) that prevent undesired side reactions while maintaining excellent functional group tolerance across diverse aryl substituents.
Impurity profile management is achieved through the inherent selectivity of the palladium-catalyzed pathway combined with optimized reaction parameters that minimize decomposition pathways. The use of sodium bicarbonate as a mild base prevents over-reaction or degradation of sensitive intermediates, while the controlled release of carbon monoxide from formic acid/acetic anhydride avoids the handling hazards associated with gaseous CO. Post-reaction purification employs standard column chromatography techniques that effectively remove residual catalysts and unreacted starting materials, as evidenced by comprehensive NMR and HRMS characterization data confirming >99% purity for all synthesized compounds. The process demonstrates exceptional substrate compatibility across various functional groups including methyl, tert-butyl, halogen, and nitro substituents without requiring additional protection/deprotection steps that typically introduce impurities in conventional syntheses.
Commercial Advantages for Pharmaceutical Supply Chains
Traditional synthesis routes for trifluoromethylated heterocycles often require harsh conditions, expensive transition metal catalysts with complex removal protocols, and multiple purification steps that increase both cost and lead time. The patented methodology overcomes these limitations through its streamlined one-pot approach that eliminates the need for specialized equipment while delivering consistent high yields across diverse substrate combinations. This process directly addresses critical pain points in pharmaceutical manufacturing by reducing cycle times and minimizing quality-related delays that commonly disrupt supply chains for complex intermediates.
- Cost reduction in API manufacturing: The elimination of expensive transition metal catalysts and hazardous reagents significantly lowers raw material costs while avoiding costly metal removal steps required in conventional syntheses. The use of commercially available palladium chloride instead of more expensive alternatives like palladium acetate reduces catalyst expenses by approximately 40%, while the mild reaction conditions minimize energy consumption compared to high-temperature processes. Furthermore, the simplified workup procedure using standard column chromatography eliminates the need for specialized purification equipment or additional solvent recovery systems that typically account for 25% of manufacturing overhead in complex intermediate production.
- Reducing lead time for high-purity intermediates: The consistent 20-hour reaction time at ambient temperature enables predictable scheduling without requiring specialized reactor cooling or heating systems that create bottlenecks in multi-product facilities. This reliability translates to reduced production cycle times by up to 50% compared to conventional methods requiring extended reaction periods or cryogenic conditions. The straightforward scalability from laboratory to production scale is demonstrated by the patent's successful gram-level synthesis without process reoptimization, enabling faster technology transfer and reducing time-to-market for new drug candidates by eliminating scale-up validation delays that typically add 8-12 weeks to development timelines.
- Commercial scale-up of complex intermediates: The robust process design accommodates diverse substrate combinations without requiring significant parameter adjustments, as evidenced by the consistent yields across all tested examples in the patent documentation. This flexibility allows manufacturers to rapidly adapt to changing customer requirements without extensive revalidation procedures that typically delay commercial production. The use of standard solvents and reagents ensures seamless integration into existing manufacturing infrastructure while maintaining compliance with regulatory requirements for residual solvents and metal impurities. The demonstrated ability to produce >99% pure material through conventional purification methods provides confidence in achieving consistent quality at commercial volumes without requiring additional capital investment in specialized equipment.
Superior Process Design Versus Conventional Methods
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethylated heterocycles typically rely on direct functionalization using expensive trifluoromethyl synthons like trifluorodiazoethane under demanding reaction conditions that require specialized handling equipment and generate significant waste streams. These methods often suffer from poor functional group tolerance that necessitates extensive protection/deprotection strategies, increasing both process complexity and impurity formation. The requirement for high temperatures or cryogenic conditions creates substantial energy demands while introducing variability in product quality due to thermal degradation pathways. Furthermore, conventional palladium-catalyzed routes frequently employ expensive ligands and require rigorous metal removal protocols that add multiple processing steps and significantly extend production timelines without guaranteeing consistent purity levels required for pharmaceutical applications.
The Novel Approach
The patented methodology overcomes these limitations through an innovative cascade reaction design that integrates multiple transformation steps into a single operation using cost-effective reagents and mild conditions. By employing formic acid/acetic anhydride as a safe carbon monoxide surrogate instead of hazardous gaseous CO, the process eliminates specialized gas handling equipment while maintaining excellent carbonylation efficiency. The strategic use of sodium bicarbonate as a mild base prevents side reactions while enabling the formation of key intermediates under ambient temperature conditions. This approach demonstrates remarkable substrate flexibility across various aryl substituents as shown in the patent examples, with yields consistently exceeding 70% and reaching up to 97% for certain substrates without requiring process modifications. The simplified workup procedure using standard column chromatography delivers >99% pure product without additional purification steps, making this methodology uniquely suited for commercial implementation in multi-product manufacturing environments where operational simplicity and reliability are paramount.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111423381B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
