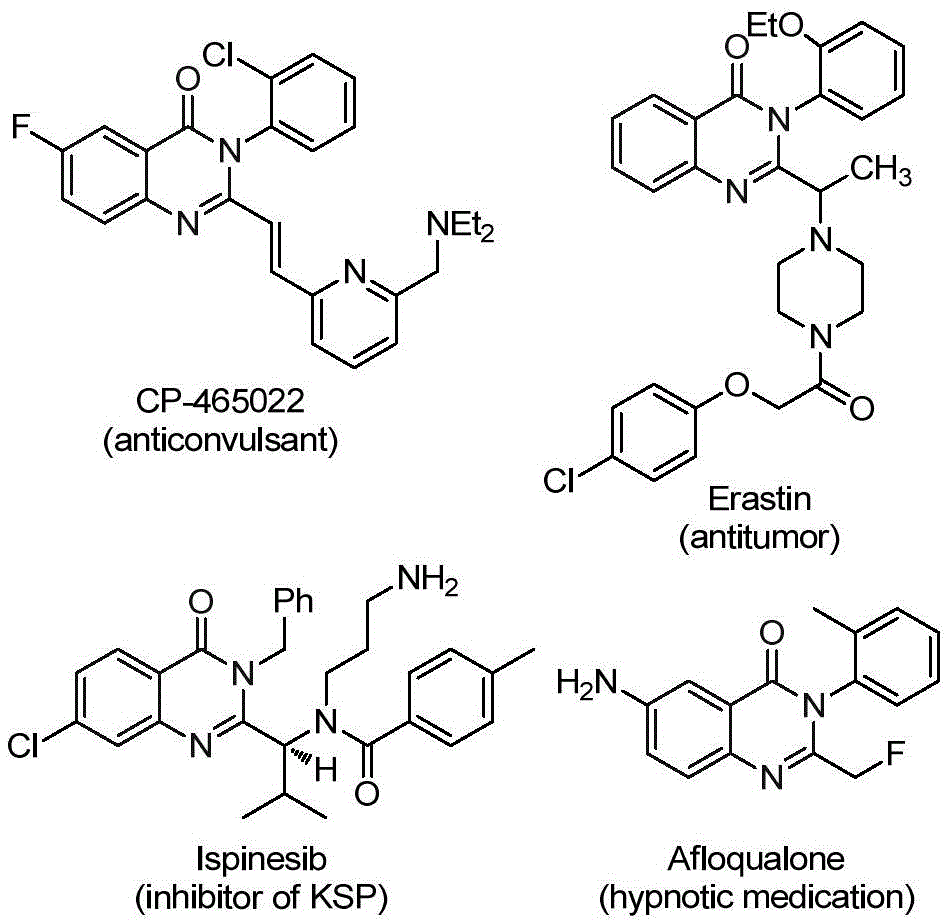
Advanced Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives: Bridging Innovation and Commercial Manufacturing
In the rapidly evolving landscape of pharmaceutical intermediate synthesis, patent CN112125856A presents a significant advancement in the preparation of 2-trifluoromethyl substituted quinazolinone derivatives. This innovative methodology addresses critical challenges in the production of these pharmacologically important compounds, which serve as key building blocks for various therapeutic agents including anticonvulsants, antitumor medications, and other bioactive molecules. The process eliminates the need for toxic carbon monoxide gas through the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid CO substitute, while maintaining excellent substrate compatibility and operational simplicity.
Advanced Reaction Mechanism and Purity Control
The patented methodology employs a sophisticated palladium-catalyzed carbonylation tandem reaction that begins with the intermolecular carbon-nitrogen bond coupling promoted by potassium tert-butoxide to form trifluoroacetamidine derivatives. This is followed by palladium catalyst insertion into the carbon-iodine bond to form a divalent palladium intermediate. Under heating conditions at 90°C, TFBen decomposes to release carbon monoxide, which inserts into the carbon-palladium bond to form an acyl palladium intermediate. The subsequent base-promoted formation of a palladium nitrogen base leads to a seven-membered ring palladium intermediate, culminating in reductive elimination to yield the final 2-trifluoromethyl substituted quinazolinone derivatives.


Purity control is inherently addressed through the reaction's design, as the use of well-defined starting materials and controlled reaction conditions minimizes side product formation. The process demonstrates excellent functional group tolerance across various substituents (R¹ = H, C₁-C₅ alkyl, halogen or trifluoromethyl; R² = substituted or unsubstituted aryl), enabling precise molecular design while maintaining high product quality. The post-processing protocol involving filtration, silica gel mixing, and column chromatography purification ensures removal of residual catalysts and impurities, resulting in products with confirmed high purity as evidenced by NMR and HRMS data from multiple examples in the patent documentation.
Commercial Advantages and Supply Chain Benefits
This innovative synthesis pathway addresses critical pain points in traditional manufacturing of quinazolinone derivatives, offering substantial commercial advantages that directly impact procurement decisions and supply chain efficiency. The elimination of hazardous carbon monoxide gas handling not only improves workplace safety but also removes significant regulatory and infrastructure burdens associated with toxic gas management in pharmaceutical manufacturing facilities.
- Cost Reduction in Chemical Manufacturing: The use of readily available starting materials including commercially accessible aromatic amines and TFBen as a solid CO substitute significantly reduces raw material costs compared to conventional methods requiring specialized or hazardous reagents. The process operates under mild conditions (90°C) without high-pressure equipment typically needed for CO-based reactions, substantially lowering capital expenditure requirements and operational costs associated with specialized reactor systems. Furthermore, the elimination of complex CO gas handling infrastructure reduces maintenance costs and safety compliance expenses that would otherwise be required for traditional carbonylation processes.
- Reducing Lead Time for High-Purity Intermediates: The streamlined reaction protocol with simple workup procedures (filtration followed by column chromatography) significantly shortens production cycles compared to multi-step conventional approaches. The robust nature of the reaction across diverse substrates enables faster development timelines for new derivative compounds without extensive process reoptimization. This operational efficiency translates directly to reduced lead times for high-purity intermediates, allowing pharmaceutical manufacturers to accelerate their drug development pipelines while maintaining strict quality standards required for clinical and commercial production.
- Enhanced Supply Chain Resilience: The use of stable, non-hazardous solid reagents instead of toxic gases creates a more reliable supply chain by eliminating dependency on specialized gas suppliers and associated transportation challenges. The broad substrate compatibility allows for flexible production scheduling based on available raw materials without compromising product quality or yield consistency. This adaptability is particularly valuable in volatile market conditions where supply chain disruptions can significantly impact manufacturing continuity, providing pharmaceutical companies with greater assurance of uninterrupted access to critical intermediates.
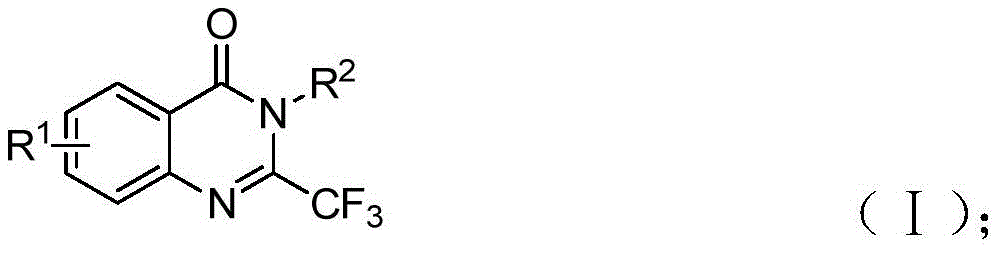
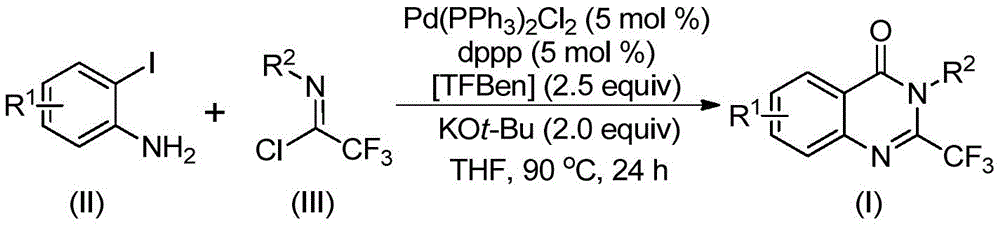
Traditional vs. Innovative Synthesis Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing 2-trifluoromethyl quinazolinone derivatives have been severely limited by multiple constraints that hinder both research and commercial production. Conventional methods typically involve harsh reaction conditions requiring high temperatures or pressures, expensive or difficult-to-handle reagents such as toxic carbon monoxide gas, and often suffer from narrow substrate scope that restricts molecular diversity. These limitations create significant barriers to efficient scale-up, with many approaches requiring pre-activated substrates or specialized equipment that increases both capital investment and operational complexity. Furthermore, traditional routes frequently produce lower yields due to competing side reactions or incomplete conversions, necessitating additional purification steps that further increase production costs and reduce overall process efficiency.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation tandem reaction that utilizes TFBen as a safe solid substitute for carbon monoxide. This innovation eliminates the need for specialized high-pressure equipment and hazardous gas handling systems while maintaining excellent reaction efficiency across diverse substrates. The process operates under mild conditions (90°C) with simple workup procedures that include standard filtration and column chromatography, making it readily adaptable to existing manufacturing infrastructure without significant capital investment. The broad functional group tolerance demonstrated in the patent documentation (with successful synthesis across various R¹ and R² substituents) enables pharmaceutical manufacturers to rapidly develop new derivatives without extensive process reoptimization, significantly accelerating time-to-market for novel therapeutic candidates while maintaining high product purity standards essential for pharmaceutical applications.
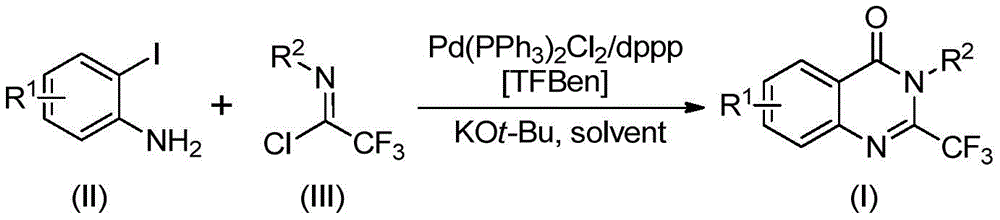

Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112125856A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
