Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Among these, quinazolinone derivatives represent a privileged structural motif found in numerous bioactive molecules exhibiting anti-inflammatory, antiviral, antifungal, and anticancer properties. A recent technological breakthrough disclosed in patent CN112125856A introduces a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, addressing critical limitations in existing synthetic routes. This innovation leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing readily available o-iodoaniline and trifluoroethylimidoyl chloride as starting materials. By replacing toxic gaseous carbon monoxide with a solid surrogate, this protocol not only enhances operational safety but also broadens the substrate scope, enabling the synthesis of diverse analogues essential for drug discovery and development pipelines.
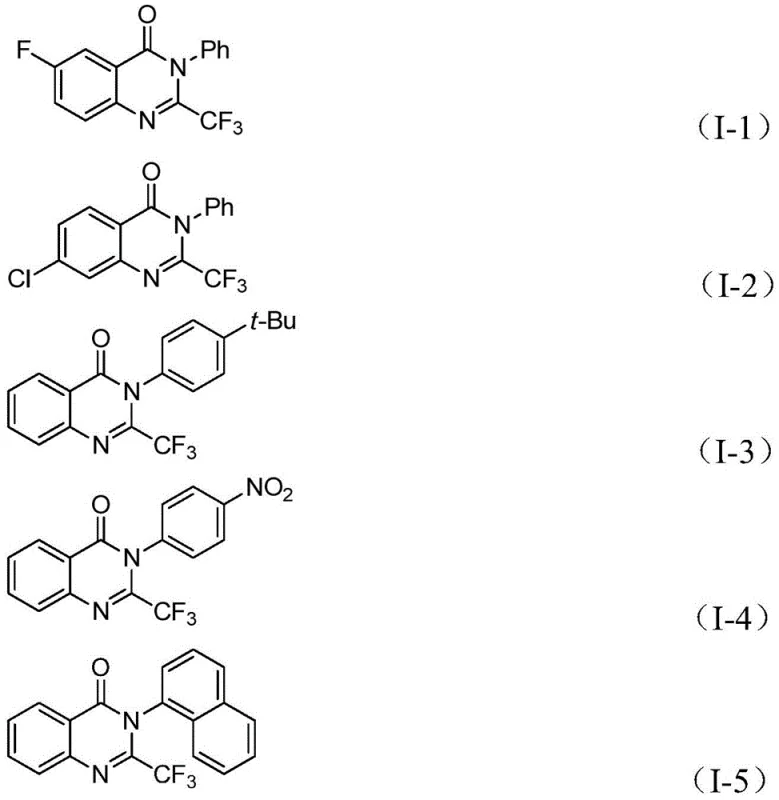
The strategic incorporation of the trifluoromethyl group into the quinazolinone core is of paramount importance due to the unique physicochemical properties imparted by fluorine atoms, such as improved metabolic stability, lipophilicity, and bioavailability. Traditional methods for accessing these valuable intermediates often suffer from harsh reaction conditions, the requirement for pre-activated expensive substrates, or the use of hazardous reagents. In contrast, the methodology outlined in the patent offers a streamlined approach that operates under relatively mild thermal conditions while maintaining high functional group tolerance. For R&D directors and process chemists, this represents a significant opportunity to optimize impurity profiles and reduce the environmental footprint of API intermediate manufacturing, aligning with modern green chemistry principles without compromising on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinones has relied on several distinct pathways, each carrying inherent drawbacks that hinder efficient commercial production. Common strategies include the cyclization of anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride, which often necessitates rigorous reaction conditions and generates stoichiometric amounts of acidic waste. Alternative routes involving the cyclization of anthranilic esters with unstable trifluoroacetamides or the use of isatoic anhydride with trifluoroacetic anhydride frequently encounter issues with low yields and narrow substrate applicability. Furthermore, methods employing T3P-promoted tandem reactions, while effective in some contexts, can be cost-prohibitive due to the expense of coupling reagents and the complexity of downstream purification. These conventional approaches often struggle to accommodate sensitive functional groups, limiting the chemical space available for medicinal chemists to explore during lead optimization phases.
The Novel Approach
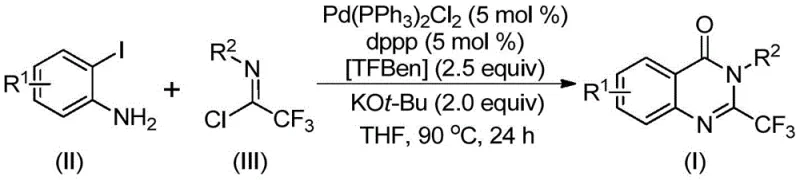
The novel palladium-catalyzed carbonylation strategy presented in the patent data fundamentally shifts the paradigm by utilizing a tandem reaction mechanism that constructs the quinazolinone ring and installs the trifluoromethyl group simultaneously. This approach employs 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, thereby circumventing the logistical and safety challenges associated with handling toxic carbon monoxide gas in a manufacturing setting. The reaction proceeds efficiently in common organic solvents such as tetrahydrofuran (THF) at moderate temperatures around 90°C. As illustrated in the reaction scheme below, the convergence of o-iodoaniline and trifluoroethylimidoyl chloride under these catalytic conditions allows for the rapid assembly of the target scaffold with excellent atom economy.
This methodology is particularly advantageous for procurement and supply chain teams because it relies on starting materials that are commercially available and inexpensive. The compatibility with a wide range of substituents, including halogens, alkyl groups, and nitro groups, means that a single robust platform can be used to generate a library of diverse derivatives. This versatility reduces the need for developing custom synthetic routes for each new analogue, significantly accelerating the timeline from bench-scale discovery to pilot plant production. The simplicity of the post-treatment process, involving filtration and standard column chromatography, further underscores the practical utility of this method for both laboratory research and potential industrial scale-up.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and troubleshooting during scale-up. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the o-iodoaniline and the trifluoroethylimidoyl chloride, facilitated by potassium tert-butoxide. This step generates a trifluoroacetamidine derivative in situ, which serves as the key intermediate for the subsequent cyclization. Following this initial coupling, the palladium catalyst, specifically bis(triphenylphosphine)palladium(II) dichloride in the presence of the dppp ligand, undergoes oxidative addition into the carbon-iodine bond of the aromatic ring. This forms a reactive divalent palladium species that is poised for carbonyl insertion.
Under the heated reaction conditions, the solid CO surrogate TFBen decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form an acyl palladium intermediate. This step is critical as it introduces the carbonyl functionality required for the lactam ring formation. Subsequently, the base promotes the formation of a palladium-nitrogen bond, leading to the generation of a seven-membered ring palladium intermediate. The catalytic cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active palladium catalyst. This detailed mechanistic pathway explains the high selectivity observed in the reaction, as the intramolecular cyclization is favored over potential intermolecular side reactions, ensuring a clean impurity profile that simplifies downstream purification efforts.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol described in the patent provides a clear and reproducible pathway for generating high-purity quinazolinone derivatives suitable for biological evaluation. The procedure involves charging a reaction vessel with the palladium catalyst system, the solid CO source, the base, and the two primary organic substrates in an anhydrous organic solvent. The mixture is then heated to facilitate the tandem coupling and cyclization events. While the specific molar ratios and reaction times can be adjusted based on the electronic nature of the substituents, the general conditions provided offer a reliable starting point for process development. For detailed operational parameters and specific stoichiometric recommendations tailored to your specific substrate, please refer to the standardized synthesis steps outlined below.
- Combine palladium catalyst, ligand, base, solid CO surrogate (TFBen), trifluoroethylimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed methodology offers substantial benefits that extend beyond mere chemical efficiency. For procurement managers, the reliance on widely available commodity chemicals such as o-iodoanilines and trifluoroethylimidoyl chlorides ensures a stable and cost-effective supply chain. Unlike specialized reagents that may have long lead times or volatile pricing, these starting materials are produced at scale by multiple suppliers globally, mitigating the risk of supply disruptions. Furthermore, the elimination of toxic carbon monoxide gas from the process removes the need for specialized high-pressure reactors and complex gas handling infrastructure, which translates directly into reduced capital expenditure and lower operational overheads for manufacturing facilities.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive catalysts and ligands that operate at low loading levels, minimizing the cost of goods sold. By avoiding the use of expensive coupling reagents like T3P and eliminating the need for cryogenic conditions or high-pressure equipment, the overall production cost is significantly lowered. Additionally, the high conversion rates and clean reaction profiles reduce the burden on purification processes, leading to higher overall yields and less solvent waste, which further contributes to cost efficiency in large-scale operations.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, a critical factor for maintaining supply continuity in the pharmaceutical sector. The tolerance for various functional groups means that the same process can be applied to synthesize a wide array of derivatives without extensive re-optimization, allowing for agile responses to changing market demands. The use of standard solvents like THF and common bases like potassium tert-butoxide simplifies logistics and inventory management, ensuring that production schedules are not delayed by the unavailability of niche reagents.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is straightforward due to the homogeneous nature of the catalytic system and the absence of hazardous gases. The solid CO surrogate simplifies dosing and control, making the process safer for operators and easier to validate under GMP conditions. Moreover, the reduced generation of hazardous waste and the potential for solvent recovery align with increasingly stringent environmental regulations, positioning this method as a sustainable choice for long-term manufacturing partnerships and reducing the environmental compliance burden on the supply chain.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the patent data. These inquiries address common concerns regarding safety, substrate scope, and scalability, providing clarity on how this method compares to traditional synthetic routes. Understanding these technical nuances is essential for integrating this process into existing manufacturing workflows and maximizing its potential value.
Q: What is the primary safety advantage of this synthesis method?
A: The method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, effectively eliminating the need for handling toxic and hazardous carbon monoxide gas directly.
Q: What types of substituents are compatible with this reaction?
A: The process demonstrates excellent substrate compatibility, tolerating various groups such as halogens (F, Cl, Br), alkyl groups, nitro groups, and naphthyl groups on both the aniline and imidoyl chloride components.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of commercially available raw materials, standard organic solvents like THF, and the avoidance of high-pressure gas equipment make this method highly scalable and operationally simple for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new pharmaceutical agents. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl quinazolinone derivative performs consistently in your downstream processes. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions safely and effectively, adhering to the highest standards of quality and safety.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in fine chemical synthesis can drive value and efficiency in your supply chain. Let us be your trusted partner in bringing innovative therapeutic solutions to market faster and more cost-effectively.
