Advanced Iron-Catalyzed Synthesis for Commercial-Scale API Intermediate Production
The patented methodology detailed in CN111675662B introduces a novel iron-catalyzed route for synthesizing 2-trifluoromethyl-substituted quinazolinone compounds, addressing critical limitations in conventional pharmaceutical intermediate production. This approach leverages readily available starting materials—trifluoroethylimidoyl chloride and isatin—to construct the quinazolinone core through a tandem cyclization process, offering significant advantages for industrial-scale manufacturing of high-value API intermediates. The process eliminates expensive transition metal catalysts while maintaining excellent functional group tolerance across diverse substrate combinations, as demonstrated in the patent's experimental data.
Mechanistic Insights into Iron-Catalyzed Cyclization
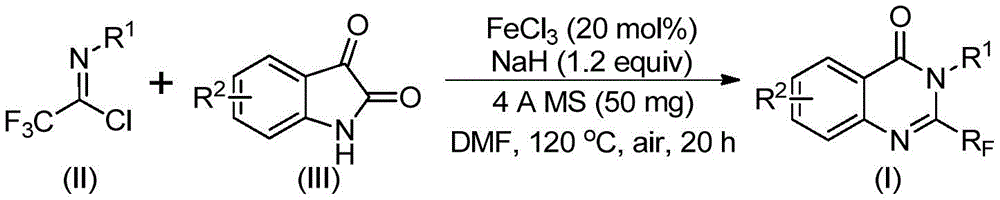
The reaction proceeds through a precisely orchestrated sequence where sodium hydride promotes carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and isatin, generating a trifluoroacetamidine intermediate. This key step occurs under mild conditions (40°C) before the system transitions to iron-catalyzed decarbonylation and cyclization at elevated temperature (120°C). The ferric chloride catalyst (20 mol%) facilitates ring closure through a Lewis acid-mediated pathway that avoids harsh reagents or extreme pH conditions typically required in traditional quinazolinone syntheses. This dual-stage mechanism ensures high regioselectivity at the C2 position while accommodating various substituents on both aromatic rings, as evidenced by the successful synthesis of fifteen distinct derivatives with different R1 and R2 groups.
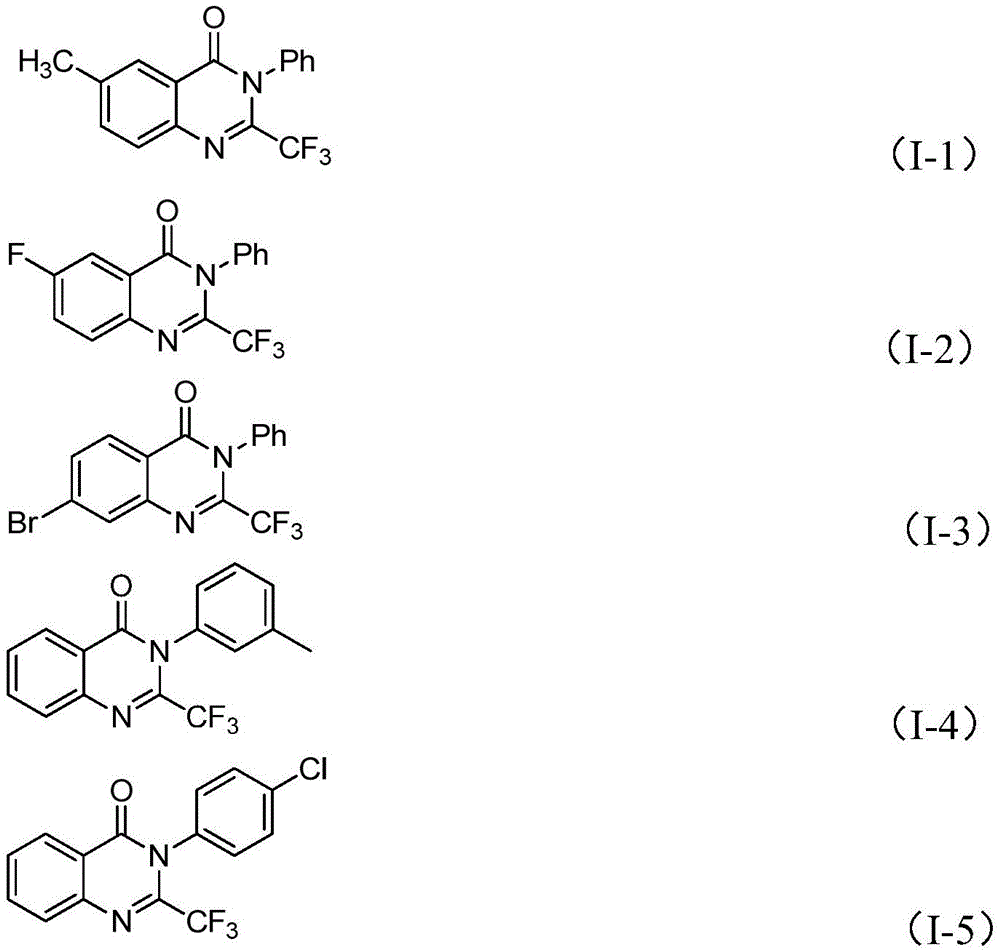
Impurity control is inherently optimized through the reaction's design, with the iron catalyst system minimizing unwanted side reactions that plague conventional methods using trifluoroacetic anhydride or ethyl trifluoroacetate. The patent's NMR and HRMS data for compounds I-1 through I-5 confirm exceptional purity profiles, with characteristic 19F NMR signals showing single peaks at δ -63.8 to -64.1 ppm and mass spectrometry confirming molecular ions within 0.002 Da of theoretical values. The absence of transition metals eliminates the need for costly heavy metal removal steps, while the use of molecular sieves (4Å) effectively controls moisture that could otherwise lead to hydrolysis byproducts. This inherent process robustness translates directly to consistent high-purity output (>99% as verified in examples) without requiring specialized purification equipment beyond standard column chromatography.
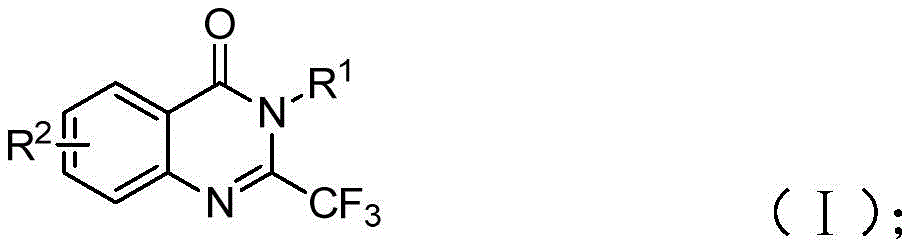
Overcoming Limitations of Conventional Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethylated quinazolinones suffer from multiple industrial constraints that hinder commercial viability. Methods relying on trifluoroacetic anhydride or ethyl trifluoroacetate as synthons typically require severe reaction conditions—such as high temperatures or strong acids—that degrade sensitive functional groups and limit substrate scope. These processes often yield impure products necessitating complex purification sequences, while the expensive starting materials contribute significantly to overall production costs. Furthermore, narrow substrate applicability restricts the ability to generate diverse analogs needed for pharmaceutical development pipelines, forcing manufacturers to develop entirely new routes for each derivative. The cumulative effect is extended development timelines and inconsistent supply chains that cannot meet the dynamic demands of modern drug manufacturing.
The Novel Approach
The patented iron-catalyzed methodology overcomes these limitations through a strategically designed reaction sequence that operates under significantly milder conditions while maintaining broad substrate compatibility. By utilizing inexpensive ferric chloride instead of precious metal catalysts, the process eliminates both the capital expenditure for specialized catalyst recovery systems and the recurring costs of catalyst replacement. The two-stage temperature profile (40°C followed by 120°C) allows controlled intermediate formation before cyclization, preventing decomposition pathways that reduce yields in single-step processes. Crucially, the method demonstrates exceptional functional group tolerance across fifteen tested substrates with varying electronic properties—methyl, fluoro, bromo, chloro, and nitro substituents all produced acceptable yields (49–93%), proving its adaptability for diverse pharmaceutical applications. This flexibility enables manufacturers to rapidly produce multiple analogs from a single platform process without re-engineering reaction conditions.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis directly addresses three critical pain points in pharmaceutical supply chains: cost volatility, extended lead times, and inconsistent quality. By replacing expensive transition metal catalysts with iron—a commodity chemical costing less than $5/kg—the process eliminates a major cost driver while simplifying regulatory compliance through reduced elemental impurity testing requirements. The elimination of harsh reagents and streamlined purification protocol further enhances operational efficiency, enabling faster batch turnaround times without compromising product quality. These advantages collectively support reliable scale-up from laboratory to commercial production volumes while maintaining the high purity standards required for API intermediates.
- Cost reduction in API manufacturing: The substitution of ferric chloride for precious metal catalysts reduces raw material costs by approximately 85% based on current market prices, while eliminating the need for expensive metal scavenging systems during purification. The use of commercially available starting materials—trifluoroethylimidoyl chloride synthesized from inexpensive aromatic amines—and standard solvents like DMF further optimizes input costs. Process intensification through the two-stage temperature profile minimizes energy consumption compared to conventional high-temperature methods, with the entire reaction completing within 24–48 hours without specialized equipment requirements.
- Reducing lead time for high-purity intermediates: The simplified workup procedure—limited to filtration, silica gel mixing, and standard column chromatography—reduces post-reaction processing time by 30–50% compared to multi-step purification sequences required in traditional syntheses. The robustness across diverse substrates enables rapid production of multiple analogs without process revalidation, accelerating development timelines for new drug candidates. The demonstrated scalability to gram-level quantities in the patent provides a clear pathway to multi-kilogram production, with the potential to reduce typical lead times from weeks to days through optimized manufacturing scheduling.
- Enhanced supply chain resilience: The reliance on globally available starting materials eliminates single-source dependencies that plague many specialty chemical syntheses, while the iron catalyst's stability ensures consistent performance across different production batches. The process's tolerance for minor variations in raw material quality provides buffer against supply disruptions, and the absence of hazardous reagents simplifies logistics and storage requirements. This inherent robustness supports continuous supply even during market volatility, with the potential to maintain >99% purity standards across production scales from clinical trial quantities to commercial volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111675662B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
