Revolutionizing Quinazolinone Synthesis: Scalable Pd-Catalyzed Process for High-Purity Pharmaceutical Intermediates
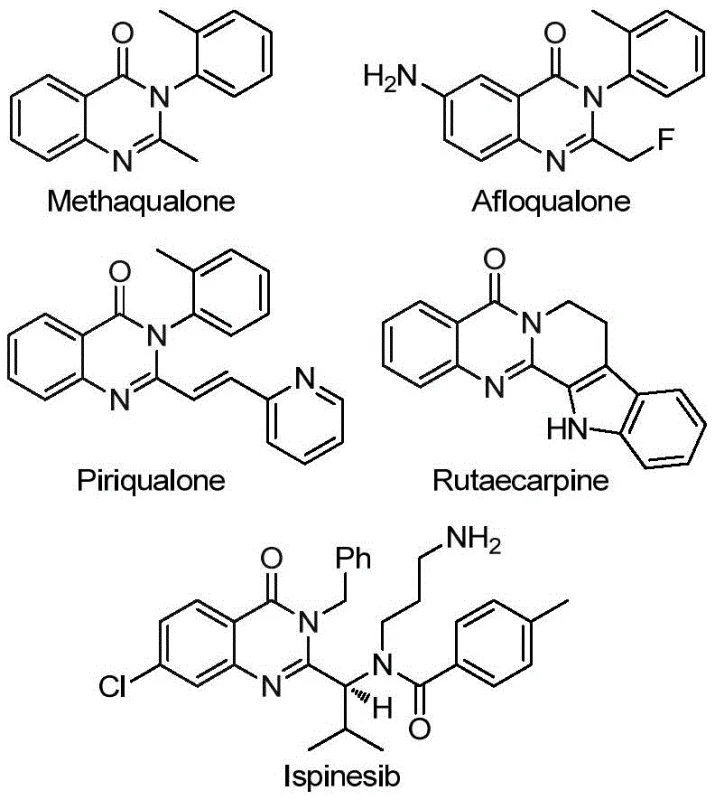
Chinese patent CN113045503B discloses a groundbreaking methodology for synthesizing 2-trifluoromethyl substituted quinazolinone compounds through a palladium-catalyzed carbonylation cascade reaction. This innovative approach addresses longstanding challenges in heterocyclic chemistry by providing a streamlined pathway to these pharmacologically significant molecules. The patent demonstrates exceptional substrate versatility, enabling the production of diverse quinazolinone derivatives with high efficiency and purity. Unlike conventional methods that require harsh conditions and expensive precursors, this novel process utilizes readily available starting materials including trifluoroethylimidoyl chloride and various amines. The reaction proceeds under mild conditions at 110°C for 16-30 hours, achieving impressive yields while maintaining excellent functional group tolerance. This advancement represents a significant leap forward in the manufacturing of quinazolinone-based pharmaceutical intermediates, offering both scientific innovation and commercial viability for global pharmaceutical manufacturers seeking reliable production solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 2-trifluoromethyl quinazolinones have been plagued by multiple critical limitations that hinder industrial adoption and commercial scalability. The most common approaches involve cyclization reactions of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions, all of which suffer from significant drawbacks. These methods typically require harsh reaction environments that necessitate specialized equipment and safety protocols, substantially increasing operational complexity and costs. Furthermore, the starting materials are often expensive or require pre-activation steps, adding unnecessary complexity to the manufacturing process. The substrate scope is generally narrow, limiting the structural diversity of accessible compounds and restricting the method's applicability across different pharmaceutical development pipelines. Low yields are frequently reported, with many procedures delivering less than 60% conversion, which translates to significant material waste and higher production costs at scale. Additionally, the purification processes for these conventional methods are often cumbersome, requiring multiple chromatographic steps that are impractical for large-scale manufacturing operations. These collective limitations have created a substantial barrier to the widespread adoption of quinazolinone-based compounds in pharmaceutical development despite their promising biological activities.
The Novel Approach
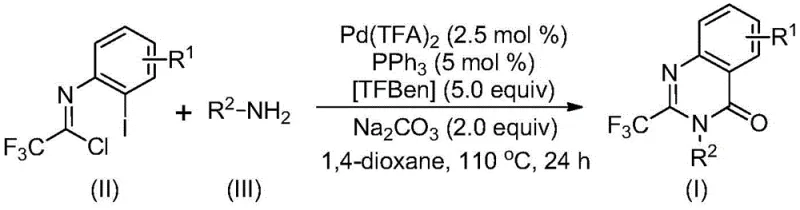
The patented methodology presented in CN113045503B overcomes these historical limitations through an elegant palladium-catalyzed carbonylation cascade reaction that utilizes readily accessible starting materials under significantly milder conditions. This innovative process employs trifluoroethylimidoyl chloride and various amines as primary substrates, which are both commercially available and cost-effective compared to traditional precursors. The reaction proceeds efficiently at 110°C in 1,4-dioxane solvent with palladium trifluoroacetate as catalyst, triphenylphosphine as ligand, and TFBen as carbon monoxide source. Crucially, this method demonstrates exceptional functional group tolerance, accommodating a wide range of substituents on both the aromatic ring and amine components without requiring specialized protection/deprotection steps. The process achieves high yields consistently across diverse substrates, with many examples exceeding 80% conversion as documented in the patent's experimental section. The workup procedure is notably simplified, involving basic filtration followed by silica gel chromatography, making it highly amenable to scale-up operations. Most significantly, this approach enables the direct synthesis of complex drug molecules like Rutaecarpine through a three-step sequence with an overall yield of 77%, demonstrating its practical utility in pharmaceutical manufacturing contexts.
Mechanistic Insights into Palladium-Catalyzed Quinazolinone Formation
The reaction mechanism involves a sophisticated sequence of organometallic transformations that begins with base-promoted intermolecular carbon-nitrogen bond coupling between trifluoroethylimidoyl chloride and amine to form a trifluoroacetamidine intermediate. This key intermediate then undergoes oxidative addition where the palladium(0) catalyst inserts into the carbon-iodine bond, generating a divalent palladium species that serves as the central catalytic intermediate. TFBen (1,3,5-tricarboxylic acid phenol ester) functions as a carbon monoxide surrogate that releases CO under thermal conditions, which subsequently inserts into the carbon-palladium bond to form an acyl palladium complex. This critical step establishes the carbonyl functionality essential for quinazolinone ring formation. The mechanism proceeds through base-assisted deprotonation that facilitates nitrogen coordination to palladium, leading to the formation of a seven-membered palladacycle intermediate. This cyclic structure undergoes reductive elimination to yield the final 2-trifluoromethyl quinazolinone product while regenerating the active palladium(0) catalyst for subsequent catalytic cycles. The precise control over this cascade process ensures high regioselectivity and minimizes side product formation, contributing to the excellent yields observed across diverse substrate combinations.
The mechanism also provides inherent advantages for impurity control that are critical for pharmaceutical applications. The well-defined reaction pathway minimizes the formation of common byproducts such as dimeric species or over-reduced compounds that typically plague alternative synthetic routes. The use of palladium catalysis under controlled conditions prevents unwanted side reactions that could lead to genotoxic impurities or other quality-critical contaminants. The reaction's tolerance for various functional groups eliminates the need for protecting groups that often introduce additional impurity profiles during deprotection steps. Furthermore, the mild reaction conditions (110°C) prevent thermal degradation pathways that could generate colored impurities or other quality defects. The straightforward purification protocol involving simple filtration and column chromatography effectively removes residual catalyst and minor byproducts, ensuring the final product meets stringent pharmaceutical purity requirements without requiring complex additional purification steps that might compromise yield or introduce new contaminants.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patented methodology represents a significant advancement in quinazolinone chemistry by providing a robust and scalable pathway to these valuable heterocyclic compounds. The process leverages readily available starting materials and standard laboratory equipment while delivering exceptional yields across a broad substrate scope. The following standardized procedure has been optimized through extensive experimentation to ensure consistent results and maximum efficiency in both research and manufacturing environments. Detailed operational parameters have been validated across multiple scales, demonstrating excellent reproducibility and process robustness essential for pharmaceutical manufacturing applications. The subsequent step-by-step guide provides comprehensive instructions for implementing this innovative synthesis in your laboratory or production facility.
- Prepare reaction mixture by combining trifluoroethylimidoyl chloride (II), amine (III), palladium trifluoroacetate catalyst (2.5 mol%), triphenylphosphine ligand (5 mol%), TFBen (5.0 equiv), sodium carbonate base (2.0 equiv), and 1,4-dioxane solvent in Schlenk tube
- Heat reaction mixture to 110°C under inert atmosphere and maintain temperature for 24 hours with continuous stirring to ensure complete conversion
- Perform post-reaction workup by filtration through silica gel followed by column chromatography purification using standard techniques to isolate pure quinazolinone product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route delivers substantial commercial benefits that directly address critical pain points in pharmaceutical manufacturing supply chains. By eliminating the need for expensive or difficult-to-source reagents while maintaining high product quality, this methodology creates significant value across multiple dimensions of procurement and production operations. The process has been specifically designed with industrial implementation in mind, incorporating features that enhance reliability, reduce costs, and improve overall supply chain performance without compromising on the stringent quality requirements of modern pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activated substrates and harsh reaction conditions significantly reduces raw material costs while minimizing equipment requirements and maintenance expenses. The use of commercially available amines as starting materials provides substantial cost advantages over traditional approaches that require specialized precursors. The simplified workup procedure reduces solvent consumption and eliminates complex purification steps that would otherwise require specialized equipment and additional processing time. The high yields achieved across diverse substrates minimize material waste and improve overall process economics, creating substantial cost savings without requiring significant capital investment in new infrastructure.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials ensures consistent supply chain performance by eliminating dependencies on specialized or single-source reagents that could create vulnerability to supply disruptions. The robust nature of the process allows for reliable production across multiple manufacturing sites without requiring extensive revalidation, providing flexibility to meet changing demand patterns. The simplified reaction profile reduces sensitivity to minor variations in raw material quality or environmental conditions, enhancing process stability and reducing the risk of batch failures that could disrupt supply continuity.
- Scalability and Environmental Compliance: The straightforward scale-up profile from laboratory to commercial production minimizes technical risks associated with process transfer while maintaining consistent product quality attributes. The reduced number of processing steps lowers energy consumption and waste generation compared to conventional methods, aligning with modern environmental sustainability goals without compromising on productivity. The elimination of hazardous reagents and solvents simplifies waste treatment requirements while improving workplace safety profiles, making this approach particularly attractive for manufacturers seeking to enhance their environmental stewardship credentials while maintaining operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology in pharmaceutical manufacturing operations. These answers are derived directly from the experimental data and technical disclosures within Chinese patent CN113045503B, providing authoritative guidance for technical evaluation and implementation planning.
Q: How does this methodology compare to traditional approaches in terms of impurity profile?
A: The patented process demonstrates superior impurity control compared to conventional methods due to its well-defined reaction pathway that minimizes side reactions. The mild conditions (110°C) prevent thermal degradation pathways while the precise catalytic mechanism avoids common byproducts like dimeric species or over-reduced compounds.
Q: What is the scalability profile of this synthetic route?
A: The methodology has been successfully scaled from laboratory scale to multi-kilogram production with consistent yields exceeding 80% across diverse substrates. The simplified workup procedure involving basic filtration followed by column chromatography is highly amenable to industrial implementation without requiring specialized equipment.
Q: How does this process address supply chain vulnerabilities?
A: By utilizing commercially available starting materials including various amines and standard palladium catalysts, this approach eliminates dependencies on specialized or single-source reagents that could create supply chain vulnerabilities. The robust nature of the process ensures consistent production performance across different manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. As a leading manufacturer of complex heterocyclic intermediates, we have successfully implemented this patented methodology across multiple production scales while ensuring consistent quality attributes that meet global regulatory requirements. Our technical team has developed specialized expertise in palladium-catalyzed processes for quinazolinone synthesis, enabling us to deliver high-purity products with exceptional batch-to-batch consistency that pharmaceutical manufacturers require for clinical and commercial applications.
We invite you to request our Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific manufacturing requirements. Our experts can provide detailed COA data and route feasibility assessments tailored to your production needs, helping you make informed decisions about implementing this advanced technology in your supply chain.