Advanced Catalytic Synthesis of Optically Active Tetrahydroquinoline Derivatives for Commercial Scale-Up
Advanced Catalytic Synthesis of Optically Active Tetrahydroquinoline Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign methodologies for constructing complex nitrogen-containing heterocycles, which serve as the structural backbone for countless bioactive molecules. A pivotal advancement in this domain is detailed in patent CN102766092A, which discloses a highly efficient method for synthesizing optically active tetrahydroquinoline derivatives using chiral spirocyclic phosphoric acids as organocatalysts. This technology represents a significant leap forward in asymmetric synthesis, addressing long-standing challenges regarding substrate scope and stereoselectivity that have plagued conventional routes. By leveraging a unique spiro-binaphthyl-derived phosphoric acid framework, this invention enables the construction of chiral tetrahydroquinoline cores with exceptional enantiomeric excess and diastereoselectivity under remarkably mild conditions. For R&D directors and process chemists, this patent offers a compelling alternative to transition-metal catalysis, promising a cleaner impurity profile and simplified downstream processing. The ability to utilize readily available starting materials—aldehydes, arylamines, and N-benzyloxycarbonyl ethylene amines—further underscores the practical utility of this method for the reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active tetrahydroquinoline derivatives has relied heavily on the asymmetric Povarov reaction or variations thereof, often employing chiral Lewis acids or transition metal complexes. While effective in specific contexts, these conventional methodologies frequently suffer from significant limitations that hinder their application in large-scale commercial manufacturing. A primary drawback is the narrow substrate scope; many established catalysts exhibit poor reactivity or diminished enantioselectivity when challenged with aliphatic aldehydes or electronically diverse aromatic substrates, necessitating extensive optimization for each new target molecule. Furthermore, traditional metal-catalyzed routes often require stringent anhydrous conditions, cryogenic temperatures, or prolonged reaction times, all of which escalate energy consumption and operational complexity. Perhaps most critically for the procurement manager, the use of heavy metal catalysts introduces the risk of metal contamination in the final API, mandating costly and time-consuming purification steps to meet rigorous regulatory limits for residual metals. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a chiral spirocyclic phosphoric acid catalyst to drive the three-component coupling reaction with unprecedented efficiency and selectivity. This organocatalytic strategy operates under mild reaction conditions, typically ranging from -30°C to 50°C, and achieves completion within a short timeframe of 1 to 5 hours. The core innovation lies in the rigid spiro-backbone of the catalyst, which creates a well-defined chiral environment that effectively discriminates between enantiotopic faces of the reacting imine intermediate. As illustrated in the reaction scheme below, this method accommodates a wide array of substrates, including those with electron-withdrawing or electron-donating groups, and notably maintains high performance even with aliphatic aldehydes, a class of substrates that traditionally poses significant challenges. The result is a process that delivers the desired cis-isomer with a diastereomeric ratio exceeding 20:1 and high enantiomeric excess, drastically simplifying the isolation of the target compound.
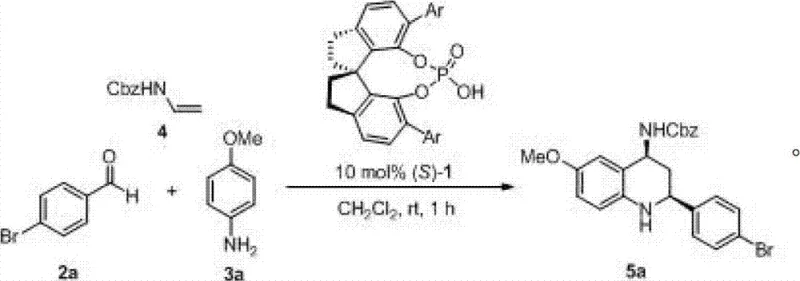
Mechanistic Insights into Chiral Spiro Phosphoric Acid Catalysis
To fully appreciate the technical superiority of this method, one must delve into the mechanistic nuances of the chiral spiro phosphoric acid catalysis. The catalyst, characterized by the general structure shown in Formula (1), functions as a chiral Brønsted acid. It activates the imine intermediate, formed in situ from the condensation of the aldehyde and the aromatic amine, through dual hydrogen bonding interactions. The phosphoryl oxygen and the hydroxyl proton of the catalyst engage simultaneously with the nitrogen atom of the imine, increasing its electrophilicity while locking it into a specific chiral conformation dictated by the bulky 3,3'-substituents on the spiro-binaphthyl skeleton. This precise spatial arrangement forces the nucleophilic attack of the N-benzyloxycarbonyl ethylene amine to occur from a single preferred trajectory, thereby establishing the two new stereocenters with high fidelity. The rigidity of the spiro-structure is paramount; unlike flexible binaphthyl analogs, the spiro-connection prevents conformational freedom that could lead to non-selective background reactions, ensuring that the chiral information is efficiently transferred to the product throughout the catalytic cycle.
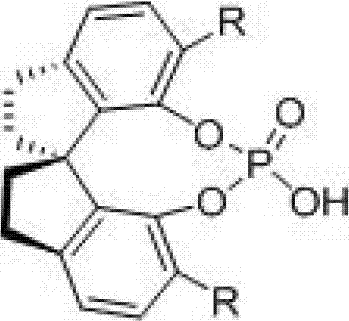
Furthermore, the mechanism inherently favors the formation of the cis-diastereomer due to the specific transition state geometry stabilized by the catalyst. The bulky substituents (R groups) at the 3,3'-positions of the catalyst, which can range from simple phenyl groups to complex naphthyl or anthracenyl moieties, act as steric gates. These gates not only enhance enantioselectivity by blocking unfavorable approach angles but also influence the diastereoselectivity by destabilizing the transition state leading to the trans-isomer. This level of control is critical for R&D teams focused on impurity profiling, as it minimizes the generation of hard-to-remove diastereomeric impurities. The ability to tune the electronic and steric properties of the R groups allows for fine-tuning the catalyst for specific substrate classes, providing a versatile platform for the synthesis of diverse tetrahydroquinoline libraries. This mechanistic robustness ensures that the process remains reliable even when scaling up, as the catalyst performance is less susceptible to minor fluctuations in reaction parameters compared to more sensitive metal-ligand systems.
How to Synthesize Optically Active Tetrahydroquinoline Derivatives Efficiently
The implementation of this catalytic system in a laboratory or pilot plant setting is straightforward and aligns well with standard organic synthesis protocols. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically nitrogen, to prevent moisture interference which could deactivate the phosphoric acid catalyst. The molar ratios are carefully controlled, with the aldehyde, arylamine, and vinyl carbamate usually employed in a 1:1:1 to 1:1:1.2 ratio, and the catalyst loading optimized between 1 to 15 mol% depending on the specific substrate reactivity. The choice of solvent is also flexible, with dichloromethane, chloroform, toluene, and acetonitrile all proving effective, allowing process engineers to select solvents based on cost, boiling point, and environmental regulations. Following the reaction period, the workup involves standard aqueous quenching and extraction, followed by purification via silica gel chromatography to yield the high-purity cis-isomer. For a detailed breakdown of the specific operational parameters and stoichiometry required for optimal results, please refer to the standardized synthesis guide below.
- Combine aldehyde, aromatic amine, and N-benzyloxycarbonyl ethylene amine in an organic solvent such as dichloromethane under nitrogen protection.
- Add the chiral spirocyclic phosphoric acid catalyst (1-15 mol%) and maintain the reaction temperature between -30°C and 50°C for 1 to 5 hours.
- Upon completion, perform standard separation and purification processes, such as silica gel column chromatography, to isolate the optically active cis-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chiral spiro phosphoric acid catalyzed route offers transformative benefits that extend far beyond mere chemical elegance. The shift from metal-based catalysis to organocatalysis fundamentally alters the cost structure and risk profile of manufacturing these valuable intermediates. By eliminating the need for precious metals such as palladium, rhodium, or iridium, the process removes a major source of raw material cost volatility and supply chain vulnerability. Moreover, the absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for specialized scavenging resins or additional purification stages to reduce metal residues to ppm levels, which directly translates to reduced processing time and lower operational expenditures. The mild reaction conditions also imply significant energy savings, as the process does not require extreme heating or deep cryogenic cooling, further enhancing the sustainability profile of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the elimination of expensive transition metal catalysts and the associated purification costs. Traditional metal-catalyzed routes often incur hidden costs related to metal scavenging, waste disposal of metal-contaminated solvents, and the loss of yield during aggressive purification steps. In contrast, this organocatalytic approach utilizes stable, non-toxic phosphoric acid derivatives that are easier to handle and dispose of. The high stereoselectivity (>90% ee in many cases) means that less material is wasted as unwanted isomers, maximizing the yield of the valuable cis-product per batch. Additionally, the short reaction times (1-5 hours) increase reactor throughput, allowing facilities to produce more batches in the same timeframe without capital investment in new equipment, thereby driving down the unit cost of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity-grade starting materials. The key reagents—various substituted benzaldehydes, anilines, and N-Cbz-vinylamine—are widely available from multiple global suppliers, reducing the risk of single-source dependency. Unlike specialized chiral ligands for metal catalysis which may have long lead times or limited availability, the chiral spiro phosphoric acid catalysts can be synthesized in bulk or sourced reliably. The robustness of the reaction to different solvent choices (DCM, Toluene, ACN) also provides flexibility; if a specific solvent faces supply constraints or price spikes, the process can be adapted to use an alternative without compromising yield or selectivity. This flexibility ensures continuous production schedules and protects against market disruptions, making it a safer bet for long-term supply contracts.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory synthesis to multi-ton commercial production is facilitated by the simplicity of the reaction setup. The exotherm is manageable under the described mild conditions, reducing the need for complex cooling infrastructure often required for highly exothermic metal-catalyzed reactions. From an environmental perspective, the metal-free nature of the catalyst aligns perfectly with green chemistry principles, reducing the ecological footprint of the manufacturing process. The high atom economy and selectivity minimize waste generation, lowering the costs associated with waste treatment and disposal. For companies aiming to meet strict ESG (Environmental, Social, and Governance) goals, adopting this cleaner synthetic route demonstrates a commitment to sustainable manufacturing practices, which is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral spiro phosphoric acid catalyzed synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production workflows or for procurement specialists assessing the quality and consistency of the supply.
Q: What are the advantages of using chiral spiro phosphoric acid over traditional metal catalysts?
A: Unlike traditional transition metal catalysts which often require harsh conditions and leave toxic metal residues, chiral spiro phosphoric acids operate under mild temperatures (-30 to 50°C) and are metal-free. This eliminates the need for expensive and complex heavy metal removal steps, significantly simplifying the purification process and ensuring higher safety profiles for pharmaceutical intermediates.
Q: Does this catalytic method support a broad range of substrate structures?
A: Yes, the patented method demonstrates exceptional versatility. While many asymmetric Povarov reactions struggle with aliphatic aldehydes, this chiral spiro phosphoric acid system maintains high enantioselectivity and diastereoselectivity (cis:trans > 20:1) even when aliphatic aldehydes are used as substrates, alongside various aromatic aldehydes and amines with different electronic properties.
Q: How does this process impact the overall production timeline and cost?
A: The process significantly reduces production timelines by shortening reaction times to just 1-5 hours at ambient or near-ambient temperatures. Furthermore, the high stereoselectivity minimizes the formation of unwanted isomers, reducing waste and the need for extensive recrystallization, which translates to substantial cost reductions in raw material consumption and energy usage during manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Derivatives Supplier
The technological potential of chiral spiro phosphoric acid catalysis is immense, yet translating patent literature into a robust, GMP-compliant manufacturing process requires deep expertise and specialized infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this translation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert process chemists is adept at optimizing reaction parameters, such as catalyst loading and solvent recycling, to maximize efficiency while maintaining stringent purity specifications. We understand that for pharmaceutical intermediates, consistency is key; therefore, our rigorous QC labs employ advanced chiral HPLC and NMR techniques to ensure every batch meets the high enantiomeric excess and diastereomeric ratio standards demanded by the industry. We do not just supply chemicals; we deliver validated, scalable solutions that de-risk your supply chain.
We invite you to leverage our technical capabilities to accelerate your drug development programs. Whether you require custom synthesis of specific tetrahydroquinoline analogs or scale-up of the general protocol described in CN102766092A, our team is ready to assist. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced organocatalytic platforms can drive value and efficiency in your manufacturing operations.
