Revolutionizing Amide Production: Green Catalysis for High-Purity Pharmaceutical Intermediates
Revolutionizing Amide Production: Green Catalysis for High-Purity Pharmaceutical Intermediates
The chemical manufacturing landscape is undergoing a profound transformation driven by the urgent need for sustainable processes that minimize environmental impact while maximizing efficiency. Patent CN101684076B introduces a groundbreaking methodology for the preparation of amides, specifically targeting the critical Beckmann rearrangement reaction which is fundamental to producing key intermediates like caprolactam for nylon and various pharmaceutical compounds. This innovation replaces the historically hazardous and corrosive oleum-based catalytic systems with task-specific amino acid ionic liquids that function simultaneously as both the reaction medium and the catalyst. By leveraging the unique physicochemical properties of these asymmetric ionic liquids, the process achieves exceptional ketoxime conversion rates and amide selectivity under remarkably mild thermal conditions. This technological leap not only addresses the severe equipment corrosion issues associated with concentrated sulfuric acid but also completely eliminates the generation of low-value ammonium sulfate by-products that plague traditional manufacturing lines. For global supply chain leaders and R&D directors, this represents a pivotal opportunity to adopt a greener, more cost-effective synthetic route that aligns with modern regulatory standards and sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industrial production of amides, particularly caprolactam, has been dominated by the liquid-phase Beckmann rearrangement utilizing fuming sulfuric acid or oleum as the primary catalyst. While this traditional approach boasts high conversion rates, it suffers from catastrophic downstream consequences that severely impact operational expenditure and environmental compliance. The most significant drawback is the stoichiometric generation of ammonium sulfate as a by-product during the neutralization step, which creates a massive waste stream of low economic value that requires costly disposal or finds limited application as fertilizer. Furthermore, the highly corrosive nature of concentrated sulfuric acid necessitates the use of specialized, expensive alloy-lined reactors and piping systems that require frequent maintenance and replacement due to degradation. The process also involves harsh reaction conditions and the handling of volatile organic compounds, posing significant safety risks to personnel and increasing the complexity of containment systems. These factors collectively result in a high carbon footprint and substantial hidden costs related to waste management, equipment downtime, and safety protocols, making the conventional route increasingly untenable in a competitive market focused on green chemistry principles.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes N-substituted amino acid ionic liquids to catalyze the rearrangement of ketoximes into amides with unprecedented efficiency and cleanliness. This method operates under mild temperatures ranging typically from 60°C to 150°C, drastically reducing energy consumption compared to the high-thermal demands of gas-phase alternatives or the exothermic management required for oleum. The ionic liquid acts as a dual-function agent, providing a polar environment that stabilizes the transition state of the rearrangement while the acidic protons on the amino acid backbone facilitate the migration of the alkyl group. Crucially, this system does not produce any volatile organic emissions or corrosive vapors, thereby preserving the integrity of standard stainless steel equipment and eliminating the need for exotic corrosion-resistant materials. The absence of ammonium sulfate formation simplifies the purification process immensely, as the product can be separated without complex neutralization and filtration steps, leading to a streamlined workflow that enhances overall throughput and reduces the physical footprint of the manufacturing facility.
Mechanistic Insights into Amino Acid Ionic Liquid Catalysis
The core of this technological advancement lies in the sophisticated molecular design of the N-substituted amino acid ionic liquid, which serves as a task-specific catalyst tailored for the Beckmann rearrangement. As illustrated in the structural formula below, the cation features an amino acid backbone where the nitrogen atom is substituted with alkyl groups, creating a chiral and asymmetric environment that can influence the stereochemical outcome of the reaction. The presence of functional groups such as carboxyl, hydroxyl, or amine moieties on the side chains allows for fine-tuning of the ionic liquid's acidity and solvation power, enabling it to interact specifically with the oxime substrate. The anionic component, often derived from inorganic acids like sulfuric acid or organic acids like methanesulfonic acid, provides the necessary proton source to initiate the rearrangement without the bulk acidity of free mineral acids. This synergistic interaction between the structured cation and the active anion creates a micro-environment that lowers the activation energy of the reaction, facilitating the migration of the substituent anti-to-the-hydroxyl group with high fidelity.
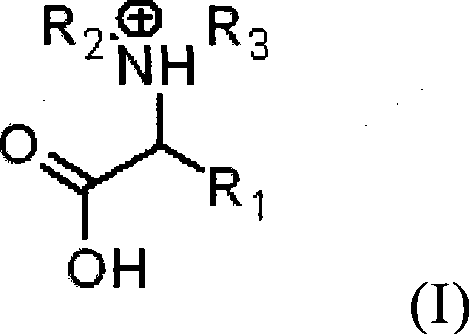
Beyond the primary catalytic cycle, the mechanism inherently supports superior impurity control, which is a critical parameter for pharmaceutical and high-performance polymer applications. Traditional acid-catalyzed routes often lead to oligomerization or degradation of the sensitive amide product due to the harsh acidic environment and elevated temperatures. However, the buffered acidity of the amino acid ionic liquid prevents over-protonation and subsequent decomposition of the formed amide, ensuring a cleaner crude product profile. The ionic nature of the solvent also suppresses side reactions that typically occur in non-polar organic solvents, such as radical formations or unwanted condensation reactions. Furthermore, the ability to recover and recycle the ionic liquid means that any trace impurities generated can be managed within the closed loop of the catalyst system rather than contaminating the final product stream. This intrinsic purity advantage reduces the burden on downstream purification units like distillation columns or crystallization tanks, allowing manufacturers to achieve stringent quality specifications with fewer processing steps and lower solvent consumption.
How to Synthesize Caprolactam Efficiently
The practical implementation of this synthesis route involves a straightforward yet highly controlled sequence of steps that begin with the preparation of the specialized ionic liquid catalyst itself. The process starts by reacting a naturally occurring or synthetic amino acid with an aldehyde compound, often formaldehyde or acetaldehyde, in the presence of a hydrogenation catalyst like Pd/C to form the N-substituted amino acid intermediate. This intermediate is subsequently treated with an acid to generate the final ionic liquid salt, which is then dried to remove residual moisture that could interfere with the rearrangement kinetics. Once the catalyst is prepared, it is mixed with the ketoxime substrate, such as cyclohexanone oxime, in a solvent like toluene at a specific molar ratio optimized for maximum conversion. The detailed standardized synthesis steps, including precise temperature ramps, stirring rates, and work-up procedures required to replicate this high-efficiency process, are outlined in the guide below.
- Prepare the N-substituted amino acid ionic liquid catalyst by reacting a specific amino acid with an aldehyde compound followed by acidification with an inorganic or organic acid.
- Mix the ketoxime substrate with the prepared ionic liquid catalyst in a suitable solvent such as toluene, maintaining a molar ratio between 1: 10 and 10:1.
- Heat the reaction mixture to a temperature between 60°C and 150°C for 0.1 to 10 hours to facilitate the Beckmann rearrangement, then isolate the amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-catalyzed process offers transformative economic benefits that extend far beyond simple yield improvements. The elimination of corrosive fuming sulfuric acid fundamentally alters the capital expenditure profile of a manufacturing plant, as it removes the necessity for expensive Hastelloy or glass-lined reactors, allowing for the use of standard carbon steel or stainless steel equipment which is significantly cheaper and easier to source. This shift also drastically reduces maintenance costs and unplanned downtime associated with acid corrosion, leading to higher asset utilization rates and more predictable production schedules. From a raw material perspective, the avoidance of ammonia for neutralization purposes cuts out a major commodity cost center, while the removal of ammonium sulfate waste disposal fees provides immediate bottom-line savings. These factors combine to create a robust cost structure that is less vulnerable to fluctuations in the prices of auxiliary chemicals and waste management services, ensuring long-term financial stability for the production of high-purity amide intermediates.
- Cost Reduction in Manufacturing: The economic argument for switching to this technology is compelling because it removes multiple cost-intensive unit operations from the value chain. By avoiding the neutralization step, manufacturers save on the purchase of ammonia and the energy required for pumping and mixing large volumes of neutralizing agents. Additionally, the recyclability of the ionic liquid catalyst means that the effective cost per kilogram of catalyst consumed approaches zero over multiple cycles, unlike homogeneous mineral acids which are consumed stoichiometrically. The reduction in waste treatment costs is equally significant, as the facility no longer needs to manage tons of solid ammonium sulfate by-product, freeing up logistics capacity and reducing environmental levies. These cumulative savings allow for a much more competitive pricing strategy for the final amide product, enhancing margin potential in price-sensitive markets like textiles and engineering plastics.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the simplified material requirements of this green synthesis route. Traditional processes rely on a steady supply of high-purity oleum and ammonia, both of which are subject to strict transportation regulations and potential supply disruptions due to their hazardous nature. In contrast, amino acid ionic liquids can be synthesized from readily available bulk chemicals like amino acids and formaldehyde, which have stable and diverse global supply bases. The non-volatile and non-flammable nature of the ionic liquid also simplifies storage and handling logistics, reducing insurance premiums and regulatory compliance burdens associated with hazardous material storage. This stability ensures consistent production output even during periods of market volatility for specialty chemicals, making the supply of critical intermediates like caprolactam more reliable for downstream customers in the pharmaceutical and polymer industries.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is inherently safer and more straightforward due to the mild reaction conditions and the absence of exothermic runaway risks associated with strong mineral acids. The liquid-phase nature of the reaction allows for easy adaptation to existing continuous flow reactors or batch vessels without major retrofitting, facilitating rapid capacity expansion to meet growing market demand. Environmentally, the process aligns perfectly with increasingly stringent global regulations regarding VOC emissions and hazardous waste generation, future-proofing the manufacturing site against tighter environmental legislation. The ability to market the final product as 'green' or 'sustainably produced' adds intangible value, appealing to end-consumers and brand owners who are prioritizing eco-friendly supply chains, thus opening up premium market segments that are inaccessible to conventionally produced commodities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of amino acid ionic liquid catalysis for amide synthesis, based on the data provided in the patent literature. These insights are designed to clarify the operational feasibility and strategic advantages of adopting this technology for large-scale manufacturing. Understanding these details is crucial for technical teams evaluating the transition from traditional acid-catalyzed processes to this advanced green chemistry platform. The answers reflect the specific performance metrics and mechanistic benefits observed in the experimental examples provided in the intellectual property documentation.
Q: What are the primary advantages of using amino acid ionic liquids over traditional oleum catalysts?
A: Unlike traditional oleum catalysts which produce large amounts of low-value ammonium sulfate by-products and cause severe equipment corrosion, amino acid ionic liquids operate under mild conditions, generate no volatile substances, and eliminate the need for neutralization steps, significantly reducing waste and maintenance costs.
Q: Can this ionic liquid catalyst be recycled for subsequent batches?
A: Yes, the patent highlights that ionic liquids are non-volatile and thermally stable, allowing them to be recovered and reused multiple times without significant loss of catalytic activity, which enhances the overall economic viability of the process.
Q: What types of ketoximes are compatible with this Beckmann rearrangement method?
A: The method demonstrates broad substrate scope, successfully converting various ketoximes including cyclohexanone oxime, acetone oxime, and benzophenone oxime into their corresponding amides with high conversion rates and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this ionic liquid-catalyzed pathway to redefine the production standards for amides and lactams in the global market. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for pharmaceutical and high-performance polymer applications, guaranteeing that every batch meets the highest international standards. We understand that adopting a new catalytic technology requires confidence in the supply partner, and our team is dedicated to providing the technical support and process optimization needed to maximize the benefits of this green synthesis route for your specific business needs.
We invite you to collaborate with us to explore how this advanced manufacturing method can optimize your supply chain and reduce your overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current volume requirements and quality targets. By engaging with us, you can access specific COA data and route feasibility assessments that demonstrate the tangible advantages of switching to our sustainably produced amide intermediates. Let us help you secure a reliable, cost-effective, and environmentally responsible supply of critical chemical building blocks for your next generation of products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
