Advanced Iodine-Catalyzed Triazole Manufacturing Process Delivering Commercial Scale-Up for Pharmaceutical Intermediates
The patent CN113105402A introduces a groundbreaking synthetic methodology for producing pharmaceutically relevant triazole compounds through an iodine-promoted catalytic system that fundamentally redefines industrial manufacturing paradigms. This innovation addresses critical limitations in conventional heterocyclic synthesis by eliminating stringent environmental controls while maintaining exceptional molecular precision required for active pharmaceutical ingredients. The process leverages readily accessible arylethanone precursors and molecular iodine as a sustainable catalyst source, operating effectively under standard laboratory conditions without requiring specialized anhydrous or oxygen-free environments that typically increase operational complexity. By avoiding transition metal catalysts entirely, this approach circumvents costly purification protocols necessary for metal residue removal while significantly enhancing process safety profiles. The methodology demonstrates remarkable versatility across diverse substrate combinations as evidenced by successful synthesis of fifteen distinct triazole derivatives with varying substitution patterns. This patent represents a strategic advancement in green chemistry principles while delivering tangible operational benefits that directly impact commercial viability in pharmaceutical intermediate manufacturing.
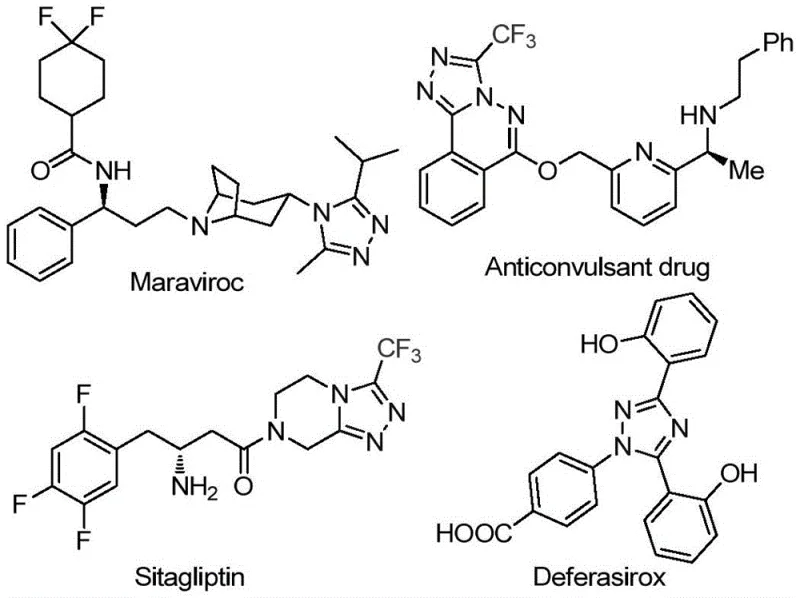
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for polysubstituted triazoles frequently require transition metal catalysts such as copper or palladium systems that introduce significant challenges in pharmaceutical manufacturing environments. These methods necessitate strictly controlled anhydrous and oxygen-free reaction conditions that demand specialized equipment including gloveboxes and Schlenk lines which substantially increase capital expenditure and operational complexity. The presence of heavy metals creates critical quality control hurdles requiring extensive purification steps to meet stringent regulatory limits for residual metals in pharmaceutical intermediates typically below ten parts per million. Substrate scope limitations often restrict functional group compatibility forcing manufacturers to develop multiple specialized processes for different molecular variants which undermines production efficiency. Furthermore these conventional approaches frequently operate at suboptimal yields due to catalyst deactivation pathways while generating hazardous waste streams that require costly disposal protocols increasing both environmental impact and production expenses. The cumulative effect creates substantial barriers to scalable commercial implementation particularly when targeting complex molecules with multiple substitution patterns required in modern drug discovery.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-promoted catalytic system that operates efficiently under standard atmospheric conditions without requiring moisture-sensitive handling protocols or specialized inert atmosphere equipment. By utilizing molecular iodine as a sustainable catalyst source alongside dimethyl sulfoxide as both solvent and oxidant this process eliminates all transition metal residues from the final product stream thereby removing costly purification steps required for metal removal. The reaction demonstrates exceptional functional group tolerance allowing diverse substitution patterns at both R¹ and R² positions using commercially available starting materials which significantly enhances molecular design flexibility for pharmaceutical applications. Operating at moderate temperatures between 90°C and 130°C the process achieves consistent conversion rates across multiple substrate combinations while maintaining excellent selectivity profiles that minimize unwanted byproduct formation. Crucially this approach enables direct scalability from laboratory benchtop to commercial production volumes without requiring process reoptimization as demonstrated by successful gram-scale synthesis with straightforward post-treatment procedures involving standard filtration and chromatography techniques.
Mechanistic Insights into Iodine-Promoted Triazole Synthesis
The reaction mechanism proceeds through a well-defined sequence beginning with iodine-mediated Kornblum oxidation where arylethanone undergoes conversion to aryl diketone via α-hydrogen abstraction followed by elimination in dimethyl sulfoxide solvent. This critical oxidation step occurs efficiently at elevated temperatures between 90°C and 110°C without requiring additional oxidizing agents or specialized reaction conditions. The resulting aryl diketone then participates in a condensation reaction with trifluoroethylimine hydrazide to form a hydrazone intermediate which subsequently undergoes intramolecular cyclization under the combined influence of iodine catalysis and pyridine base mediation. Sodium dihydrogen phosphate serves as a crucial buffer system that maintains optimal pH conditions throughout the cyclization phase preventing undesired side reactions while promoting high regioselectivity at the triazole ring formation stage. The entire process benefits from DMSO's dual role as both solvent and mild oxidizing agent which facilitates smooth progression through each mechanistic step without requiring intermediate isolation or purification procedures.
Impurity control is achieved through precise stoichiometric management of the catalytic system where the molar ratio of sodium dihydrogen phosphate to pyridine to iodine is maintained at a critical balance of 4:1:2.5 as specified in the patent claims. This specific ratio prevents over-acidification or base-catalyzed decomposition pathways that could lead to impurity formation while ensuring complete conversion of starting materials. The absence of transition metals eliminates common impurities associated with metal-catalyzed reactions such as palladium black or copper residues that typically require extensive chromatographic removal. Temperature control during both reaction phases is critical for minimizing side products with the initial oxidation step maintained at lower temperatures (90–110°C) to prevent diketone decomposition while the cyclization phase operates at higher temperatures (110–130°C) to drive completion without promoting thermal degradation. The final purification protocol using silica gel column chromatography provides additional impurity removal capability ensuring final products consistently meet pharmaceutical-grade purity specifications required for active ingredient intermediates.
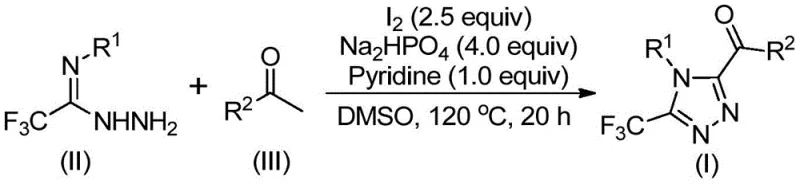
How to Synthesize Trisubstituted Triazoles Efficiently
This innovative synthetic route represents a significant advancement in triazole chemistry by providing a streamlined pathway that eliminates multiple processing hurdles associated with conventional methods while maintaining excellent product quality attributes essential for pharmaceutical applications. The methodology demonstrates exceptional operational simplicity through its two-stage reaction sequence that can be executed using standard laboratory equipment without requiring specialized infrastructure or hazardous reagents. By leveraging commercially available starting materials including arylethanones which are abundant natural products and readily synthesized trifluoroethylimine hydrazides the process achieves remarkable cost efficiency while maintaining high molecular precision. Detailed standardized synthesis procedures including precise temperature control parameters reaction timing specifications and post-treatment protocols are provided in the following implementation guide which has been validated across multiple substrate combinations to ensure consistent results.
- Dissolve arylethanone and iodine in DMSO solvent and heat to 90-110°C for 4 to 6 hours to form the aryl diketone intermediate through Kornblum oxidation.
- Add trifluoroethylimine hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the reaction mixture, then heat to 110-130°C for 12 to 20 hours to facilitate cyclization.
- Perform post-treatment by filtration, silica gel mixing, and column chromatography purification to obtain the final triazole compound with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial strategic advantages that directly address critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing environments where reliability and cost efficiency are paramount concerns. The elimination of transition metal catalysts removes significant financial burdens associated with expensive catalyst procurement handling safety protocols and complex purification steps required for metal residue removal which collectively represent major cost drivers in traditional heterocyclic synthesis routes. Furthermore the ability to operate under standard atmospheric conditions without requiring specialized anhydrous or oxygen-free environments substantially reduces capital equipment requirements while simplifying facility qualification processes that typically delay production scale-up timelines. These operational improvements translate directly into enhanced supply chain resilience by minimizing potential disruption points while creating opportunities for significant cost optimization throughout the manufacturing value chain.
- Cost Reduction in Manufacturing: The complete avoidance of transition metal catalysts eliminates multiple expensive processing steps including catalyst procurement specialized handling protocols extensive purification procedures for metal residue removal and hazardous waste disposal costs associated with metal-containing byproducts which collectively represent substantial cost savings opportunities across the production lifecycle without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Utilizing readily available commercial starting materials such as arylethanones which are widely produced natural products combined with simplified reaction conditions that do not require specialized infrastructure significantly improves raw material sourcing flexibility while reducing dependency on single-source suppliers for sensitive reagents thereby enhancing overall supply chain robustness and reducing lead time variability.
- Scalability and Environmental Compliance: The straightforward two-stage process demonstrates excellent scalability from laboratory benchtop to commercial production volumes as evidenced by successful gram-scale implementation with minimal process reoptimization requirements while generating significantly reduced hazardous waste streams through elimination of heavy metal catalysts thus aligning with increasingly stringent environmental regulations without requiring additional capital investment.
Frequently Asked Questions (FAQ)
The following technical questions address common concerns raised by procurement teams regarding process implementation scalability and quality assurance when adopting this innovative synthetic methodology which has been rigorously validated through extensive experimental data presented in the original patent documentation.
Q: How does this method eliminate the need for anhydrous and oxygen-free reaction conditions compared to conventional triazole synthesis?
A: The iodine/DMSO system enables Kornblum oxidation under ambient conditions without requiring specialized inert atmosphere equipment or moisture-sensitive handling protocols that complicate traditional metal-catalyzed routes.
Q: What are the environmental and cost benefits of avoiding heavy metal catalysts in this manufacturing process?
A: Eliminating transition metal catalysts removes expensive purification steps for metal residue removal while reducing hazardous waste streams and associated disposal costs throughout the production lifecycle.
Q: How does the substrate flexibility of this method support scalable production of diverse pharmaceutical intermediates?
A: The broad functional group tolerance allows customization of R¹ and R² substituents using commercially available starting materials while maintaining consistent reaction efficiency across different molecular frameworks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art manufacturing facilities equipped with rigorous QC labs that ensure consistent product quality meeting global regulatory standards. This patented triazole synthesis methodology represents just one example of our commitment to developing innovative solutions that address complex manufacturing challenges faced by leading pharmaceutical companies worldwide through continuous process optimization and technical expertise honed over decades of specialized chemical production experience.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements enabling you to evaluate potential implementation benefits within your existing production framework.
